Abstract
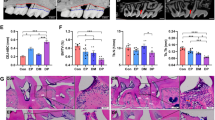
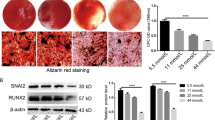
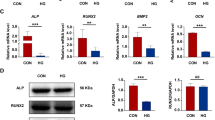
Periodontitis is one of the most common complications of diabetes, which seriously affects patients’ life quality. It is important to find the key factors and mechanisms to improve the treatment of periodontitis. In our study, high glucose (HG) and lipopolysaccharide (LPS) treated human periodontal ligament cells (hPDLCs) and LPS treated diabetic mice was used to establish the diabetic periodontitis model in vitro and in vivo. O-linked beta-N-acetylglucosamine glycosylation (O-GlcNAcylation) and O-linked N-acetylglucosamine transferase (OGT) protein levels were detected by western blot assay. Cell counting kit-8, alkaline phosphatase (ALP), and alizarin red staining (ARS) assays were used to observe the O-GlcNAcylation and OGT effects on cell viability and osteoblast differentiation. Co-immunoprecipitation (Co-IP) assay was used to detect the relationship between OGT and ALP. The results showed that the levels of OGT and O-GlcNAcylation were significantly increased in both cell and mouse models. ALP and ARS staining results showed that silencing of OGT or inhibition of O-glycosylation notably improved osteogenic differentiation, increased the osteoprotegerin (OPG) protein levels and decreased the receptor activator for nuclear factor-κB Ligand (RANKL) protein levels of the HG and LPS treated hPDLCs. In diabetic periodontitis mice, knockdown of OGT relieved the injury of gingival tissue, increased the ALP and OPG levels and decreased the RANKL levels. Besides, ALP interacted with OGT protein, and OGT protein was found to act on ALP serine 513 glycosylation. In conclusion, our study demonstrated that excessive O-GlcNAcylation could restrain osteoblast differentiation by O-glycosylation in ALP.




Similar content being viewed by others
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Grzeczka, A., Lech, M., Wozniak, G., Graczyk, S., Kordowitzki, P., Olejnik, M., Gehrke, M., & Jaśkowski, J. M. (2023). Periodontitis disease in farmed ruminants-current state of research. International Journal of Molecular Sciences, 24(11), 9763.
Li, B., Xin, Z., Gao, S., Li, Y., Guo, S., Fu, Y., Xu, R., Wang, D., Cheng, J., Liu, L., Zhang, P., & Jiang, H. (2023). SIRT6-regulated macrophage efferocytosis epigenetically controls inflammation resolution of diabetic periodontitis. Theranostics, 13(1), 231–249.
Shang, J., Liu, H., Zheng, Y., & Zhang, Z. (2023). Role of oxidative stress in the relationship between periodontitis and systemic diseases. Frontiers in Physiology, 14, 1210449.
Laursen, H. V. B., Jørgensen, E. P., Vestergaard, P., & Ehlers, L. H. (2023). A systematic review of cost-effectiveness studies of newer non-insulin antidiabetic drugs: Trends in decision-analytical models for modelling of type 2 diabetes mellitus. Pharmaco Economics. https://doi.org/10.1007/s40273-023-01268-5
Liu, N., Wang, G., Liu, C., Liu, J., Huang, S., Zhou, Y., & Xiao, E. (2023). Non-alcoholic fatty liver disease and complications in type 1 and type 2 diabetes: A Mendelian randomization study. Diabetes, Obesity & Metabolism, 25(2), 365–376.
Wu, C. Z., Yuan, Y. H., Liu, H. H., Li, S. S., Zhang, B. W., Chen, W., An, Z. J., Chen, S. Y., Wu, Y. Z., Han, B., Li, C. J., & Li, L. J. (2020). Epidemiologic relationship between periodontitis and type 2 diabetes mellitus. BMC Oral Health, 20(1), 204.
Simpson, T. C., Clarkson, J. E., Worthington, H. V., MacDonald, L., Weldon, J. C., Needleman, I., Iheozor-Ejiofor, Z., Wild, S. H., Qureshi, A., Walker, A., Patel, V. A., Boyers, D., & Twigg, J. (2022). Treatment of periodontitis for glycaemic control in people with diabetes mellitus. Cochrane Database System Review, 4(4), CD004714.
Lalla, E., & Papapanou, P. N. (2011). Diabetes mellitus and periodontitis: A tale of two common interrelated diseases. Nature Reviews Endocrinology, 7(12), 738–748.
Sørensen, D. M., Büll, C., Madsen, T. D., Lira-Navarrete, E., Clausen, T. M., Clark, A. E., Garretson, A. F., Karlsson, R., Pijnenborg, J. F. A., Yin, X., Miller, R. L., Chanda, S. K., Boltje, T. J., Schjoldager, K. T., Vakhrushev, S. Y., Halim, A., Esko, J. D., Carlin, A. F., Hurtado-Guerrero, R., … Narimatsu, Y. (2023). Identification of global inhibitors of cellular glycosylation. Nature Communications, 14(1), 948.
Eichler, J. (2019). Protein glycosylation. Current Biology, 29(7), R229–R231.
Macedo-da-Silva, J., Santiago, V. F., Rosa-Fernandes, L., Marinho, C. R. F., & Palmisano, G. (2021). Protein glycosylation in extracellular vesicles: Structural characterization and biological functions. Molecular Immunology, 135, 226–246.
Sanz-Martinez, I., Pereira, S., Merino, P., Corzana, F., & Hurtado-Guerrero, R. (2023). Molecular recognition of GalNAc in mucin-type O-glycosylation. Accounts of Chemical Research, 56(5), 548–560.
Li, X., Lei, C., Song, Q., Bai, L., Cheng, B., Qin, K., Li, X., Ma, B., Wang, B., Zhou, W., Chen, X., & Li, J. (2023). Chemoproteomic profiling of O-GlcNAcylated proteins and identification of O-GlcNAc transferases in rice. Plant Biotechnology Journal, 21(4), 742–753.
Chang, Y. H., Weng, C. L., & Lin, K. I. (2020). O-GlcNAcylation and its role in the immune system. Journal of Biomedical Science, 27(1), 57.
Li, Y., Xie, M., Men, L., & Du, J. (2019). O-GlcNAcylation in immunity and inflammation: An intricate system (Review). International Journal of Molecular Medicine, 44(2), 363–374.
Packer, M. (2023). Foetal recapitulation of nutrient surplus signalling by O-GlcNAcylation and the failing heart. European Journal of Heart Failure, 25(8), 1199–1212.
Zhu, Y., & Hart, G. W. (2021). Targeting O-GlcNAcylation to develop novel therapeutics. Molecular Aspects of Medicine, 79, 100885.
Zhang, L., Wang, P., Zhou, Y., Cheng, Y., Li, J., Xiao, X., Yin, C., Li, J., Meng, X., & Zhang, Y. (2023). Associations of ozone exposure with gestational diabetes mellitus and glucose homeostasis: Evidence from a birth cohort in Shanghai, China. The Science of the Total Environment, 857(Pt 1), 159184.
Zhao, M., Xie, Y., Gao, W., Li, C., Ye, Q., & Li, Y. (2023). Diabetes mellitus promotes susceptibility to periodontitis-novel insight into the molecular mechanisms. Frontiers in Endocrinology, 14, 1192625.
Zhang, X., Wang, M., Wang, X., Qu, H., Zhang, R., Gu, J., Wu, Y., Ni, T., Tang, W., & Li, Q. (2021). Relationship between periodontitis and microangiopathy in type 2 diabetes mellitus: A meta-analysis. Journal of Periodontal Research, 56(6), 1019–1027.
Cirelli, T., Nicchio, I. G., Bussaneli, D. G., Silva, B. R., Nepomuceno, R., Orrico, S. R. P., Cirelli, J. A., Theodoro, L. H., Barros, S. P., & Scarel-Caminaga, R. M. (2023). Evidence linking PPARG genetic variants with periodontitis and type 2 diabetes mellitus in a Brazilian population. International Journal of Molecular Sciences, 24(7), 6760.
Portes, J., Bullón, B., Quiles, J. L., Battino, M., & Bullón, P. (2021). Diabetes mellitus and periodontitis share intracellular disorders as the main meeting point. Cells, 10(9), 2411.
Xu, X. R., Xu, J. L., He, L., Wang, X. E., Lu, H. Y., & Meng, H. X. (2023). Comparison of the inflammatory states of serum and gingival crevicular fluid in periodontitis patients with or without type 2 diabetes mellitus. Journal of Dental Sciences, 18(3), 1125–1133.
Santos, V. R., Lima, J. A., Gonçalves, T. E., Bastos, M. F., Figueiredo, L. C., Shibli, J. A., & Duarte, P. M. (2010). Receptor activator of nuclear factor-kappa B ligand/osteoprotegerin ratio in sites of chronic periodontitis of subjects with poorly and well-controlled type 2 diabetes. Journal of Periodontology, 81(10), 1455–1465.
Saha, A., Bello, D., & Fernández-Tejada, A. (2021). Advances in chemical probing of protein O-GlcNAc glycosylation: Structural role and molecular mechanisms. Chemical Society Reviews, 50(18), 10451–10485.
Estevez, A., Zhu, D., Blankenship, C., & Jiang, J. (2020). Molecular interrogation to crack the case of O-GlcNAc. Chemistry, 26(53), 12086–12100.
Hanover, J. A., Chen, W., & Bond, M. R. (2018). O-GlcNAc in cancer: An oncometabolism-fueled vicious cycle. Journal of Bioenergetics and Biomembranes, 50(3), 155–173.
Cui, Y., Cruz, M., Palatnik, A., & Olivier-Van Stichelen, S. (2023). O-GlcNAc transferase contributes to sex-specific placental deregulation in gestational diabetes. Placenta, 131, 1–12.
Wang, H. F., Wang, Y. X., Zhou, Y. P., Wei, Y. P., Yan, Y., Zhang, Z. J., & Jing, Z. C. (2023). Protein O-GlcNAcylation in cardiovascular diseases. Acta Pharmacologica Sinica, 44(1), 8–18.
Lee, B. E., Suh, P. G., & Kim, J. I. (2021). O-GlcNAcylation in health and neurodegenerative diseases. Experimental & Molecular Medicine, 53(11), 1674–1682.
Shi, Q., Shen, Q., Liu, Y., Shi, Y., Huang, W., Wang, X., Li, Z., Chai, Y., Wang, H., Hu, X., Li, N., Zhang, Q., & Cao, X. (2022). Increased glucose metabolism in TAMs fuels O-GlcNAcylation of lysosomal cathepsin B to promote cancer metastasis and chemoresistance. Cancer Cell, 40(10), 1207–1222.
Xu, W., Zhang, X., Wu, J. L., Fu, L., Liu, K., Liu, D., Chen, G. G., Lai, P. B., Wong, N., & Yu, J. (2017). O-GlcNAc transferase promotes fatty liver-associated liver cancer through inducing palmitic acid and activating endoplasmic reticulum stress. Journal of Hepatology, 67(2), 310–320.
Llorente-Pelayo, S., Docio, P., Lavín-Gómez, B. A., García-Unzueta, M. T., de Las, C. I., de la Rubia, L., Cabero-Pérez, M. J., & González-Lamuño, D. (2020). Modified serum ALP values and timing of apparition of knee epiphyseal ossification centers in preterm infants with cholestasis and risk of concomitant metabolic bone disease of prematurity. Nutrients, 12(12), 3854.
Haarhaus, M., Brandenburg, V., Kalantar-Zadeh, K., Stenvinkel, P., & Magnusson, P. (2017). Alkaline phosphatase: A novel treatment target for cardiovascular disease in CKD. Nature Reviews Nephrology, 13(7), 429–442.
Al Nofal, A. A., Altayar, O., BenKhadra, K., Qasim Agha, O. Q., Asi, N., Nabhan, M., Prokop, L. J., Tebben, P., & Murad, M. H. (2015). Bone turnover markers in Paget’s disease of the bone: A systematic review and meta-analysis. Osteoporosis International, 26(7), 1875–1891.
Acknowledgements
Not applicable.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by LS. The first draft of the manuscript was written by WL and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical Approval
Animal experiments were operated according to the Institutional Animal Care and Use Committee of Beijing Hanhe Daguanying Dental Clinic. All animal experiments should comply with the ARRIVE guidelines. All methods were carried out in accordance with relevant guidelines and regulations.
Consent to Participate
Not applicable.
Consent to Publish
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Luo, W., Sun, L. O-Linked N-Acetylglucosamine Transferase Regulates Bone Homeostasis Through Alkaline Phosphatase Pathway in Diabetic Periodontitis. Mol Biotechnol (2023). https://doi.org/10.1007/s12033-023-00947-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12033-023-00947-0