Abstract
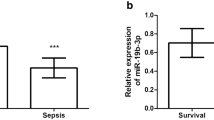
Severe pneumonia in children is a group of inflammatory diseases of respiratory tract caused by pathogenic microorganisms. Increasing evidence suggested the crucial effects of microRNA on inflammatory diseases. This study aimed to reveal the expression and role of miR-483-3p in the serum of children with severe pneumonia, and to explore the effect of miR-483-3p on the biological function of lipopolysaccharide (LPS)-induced MRC-5 cells. MRC-5 cells were disposed with LPS to construct an in vitro pneumonia cell model. The relative expression level of miR-483-3p was measured by qRT-PCR. ROC curve was used to evaluate the diagnostic value of miR-483-3p in severe pneumonia. The Kaplan–Meier curve was performed to test the characteristics of survival distribution of different miRNA classifications. Cell viability and apoptosis were performed by CCK-8 assay and flow cytometry. IL-1β, TNF-α, and IL-6 were detected by ELISA. Luciferase reporter gene assay and western blot analysis were performed to detect the interaction between miR-483-3p and IGF-1. The expression of serum miR-483-3p in severe pneumonia patients was higher than in controls. The AUC value of the ROC curve was 0.919, indicating that miR-483-3p had diagnostic value for severe pneumonia. The survival curve showed that patients with high expression of miR-483-3p had higher mortality. Cell viability and apoptosis assay showed that overexpression of miR-483-3p suppressed cell proliferation and promoted apoptosis. And upregulation of miR-483-3p promoted generation of inflammatory cytokines. Luciferase report gene assay and western blot assay both illustrated that IGF-1 might be the target gene of miR-483-3p. Serum miR-483-3p can be used as a biomarker for the diagnosis of severe pneumonia. High expression of miR-483-3p promoted the development of severe pneumonia.





Similar content being viewed by others
References
Waites, K. B. (2003). New concepts of Mycoplasma pneumoniae infections in children. Pediatric Pulmonology, 36, 267–278.
Levine, O. S., O’Brien, K. L., Deloria-Knoll, M., Murdoch, D. R., Feikin, D. R., DeLuca, A. N., et al. (2012). The pneumonia etiology research for child health project: A 21st century childhood pneumonia etiology study. Clinical Infectious Diseases, 54(Suppl 2), S93-101.
Chen, Z. R., Zhang, G. B., Wang, Y. Q., Yan, Y. D., Zhou, W. F., Zhu, C. H., et al. (2013). Soluble B7–H3 elevations in hospitalized children with Mycoplasma pneumoniae pneumonia. Diagnostic Microbiology and Infectious Disease, 77, 362–366.
Ebrahimi, A., & Sadroddiny, E. (2015). MicroRNAs in lung diseases: Recent findings and their pathophysiological implications. Pulmonary Pharmacology & Therapeutics, 34, 55–63.
Bartel, D. P. (2004). MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell, 116, 281–297.
Rupaimoole, R., & Slack, F. J. (2017). MicroRNA therapeutics: Towards a new era for the management of cancer and other diseases. Nature Reviews Drug Discovery, 16, 203–222.
Jin, F., Hu, H., Xu, M., Zhan, S., Wang, Y., Zhang, H., et al. (2018). Serum microRNA profiles serve as novel biomarkers for autoimmune diseases. Frontiers in Immunology, 9, 2381.
Kumar, S., Vijayan, M., Bhatti, J. S., & Reddy, P. H. (2017). MicroRNAs as peripheral biomarkers in aging and age-related diseases. Progress in Molecular Biology and Translational Science, 146, 47–94.
Wojciechowska, A., Braniewska, A., & Kozar-Kaminska, K. (2017). MicroRNA in cardiovascular biology and disease. Advances in Clinical and Experimental Medicine, 26, 865–874.
Abd-El-Fattah, A. A., Sadik, N. A., Shaker, O. G., & Aboulftouh, M. L. (2013). Differential microRNAs expression in serum of patients with lung cancer, pulmonary tuberculosis, and pneumonia. Cell Biochemistry and Biophysics, 67, 875–884.
Fei, S., Cao, L., & Pan, L. (2018). microRNA3941 targets IGF2 to control LPSinduced acute pneumonia in A549 cells. Molecular Medicine Reports, 17, 4019–4026.
Ying, H., Kang, Y., Zhang, H., Zhao, D., Xia, J., Lu, Z., et al. (2015). MiR-127 modulates macrophage polarization and promotes lung inflammation and injury by activating the JNK pathway. The Journal of Immunology, 194, 1239–1251.
Fu, H., Tie, Y., Xu, C., Zhang, Z., Zhu, J., Shi, Y., et al. (2005). Identification of human fetal liver miRNAs by a novel method. FEBS Letters, 579, 3849–3854.
Liu, P., Yang, X., Zhang, H., Pu, J., & Wei, K. (2018). Analysis of change in microRNA expression profiles of lung cancer A549 cells treated with radix tetrastigma hemsleyani flavonoids. Oncotargets and Therapy, 11, 4283–4300.
Huang, S., Feng, C., Zhai, Y. Z., Zhou, X., Li, B., Wang, L. L., et al. (2017). Identification of miRNA biomarkers of pneumonia using RNA-sequencing and bioinformatics analysis. Experimental and Therapeutic Medicine, 13, 1235–1244.
Dean, P., & Florin, T. A. (2018). Factors associated with pneumonia severity in children: A systematic review. Journal of Pediatric Infectious Diseases Society, 7, 323–334.
Ruuskanen, O., Lahti, E., Jennings, L. C., & Murdoch, D. R. (2011). Viral pneumonia. Lancet, 377, 1264–1275.
Boyd, K. (2017). Back to the basics: Community-acquired pneumonia in children. Pediatric Annals, 46, e257–e261.
le Roux, D. M., & Zar, H. J. (2017). Community-acquired pneumonia in children—A changing spectrum of disease. Pediatric Radiology, 47, 1392–1398.
Hirsch, A. W., Monuteaux, M. C., Fruchtman, G., Bachur, R. G., & Neuman, M. I. (2016). Characteristics of children hospitalized with aspiration pneumonia. Hospital Pediatrics, 6, 659–666.
Polikepahad, S., Knight, J. M., Naghavi, A. O., Oplt, T., Creighton, C. J., Shaw, C., et al. (2010). Proinflammatory role for let-7 microRNAS in experimental asthma. Journal of Biological Chemistry, 285, 30139–30149.
Maemura, T., Fukuyama, S., & Kawaoka, Y. (2020). High levels of miR-483-3p are present in serum exosomes upon infection of mice with highly pathogenic avian influenza virus. Frontiers in Microbiology, 11, 144.
Stiles, A. D., & D’Ercole, A. J. (1990). The insulin-like growth factors and the lung. American Journal of Respiratory Cell and Molecular Biology, 3, 93–100.
Trueba-Saiz, A., Fernandez, A. M., Nishijima, T., Mecha, M., Santi, A., Munive, V., et al. (2017). Circulating insulin-like growth factor I regulates its receptor in the brain of male mice. Endocrinology, 158, 349–355.
Oherle, K., Acker, E., Bonfield, M., Wang, T., Gray, J., Lang, I., et al. (2020). Insulin-like growth factor 1 supports a pulmonary niche that promotes type 3 innate lymphoid cell development in newborn lungs. Immunity, 52, 275–294.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declared that they have no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhou, J., Lin, J., Zhao, Y. et al. Deregulated Expression of miR-483-3p Serves as a Diagnostic Biomarker in Severe Pneumonia Children with Respiratory Failure and Its Predictive Value for the Clinical Outcome of Patients. Mol Biotechnol 64, 311–319 (2022). https://doi.org/10.1007/s12033-021-00415-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12033-021-00415-7