Abstract
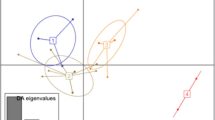
Chloroplast DNA has been used extensively to analyze plant phylogenies at different taxonomic levels because of its size, organization and sequence conservation. In the present research, two chloroplastic regions, petA–psaJ, trnC–trnD and four DNA barcodes (trnH–psbA, ITS, rbcL, matK), were used to introduce suitable regions for the assessment of genetic diversity among P. granatum L. genotypes. Analysis of psbE–petL in petA–psaJ region revealed 1,300 nucleotides with 4.29 % genetic diversity among genotypes, while trnC–petN in trnC–trnD region showed 1.8 % genetic diversity. Therefore, despite the results obtained from the study of other plants, the trnC–trnD region had a low potential for the evaluation of diversity among pomegranate genotypes. Analysis of DNA barcodes in pomegranate showed that trnH–psbA (genetic diversity 2.91 %) provides the highest intra-species variation, followed by ITS (genetic diversity 0.44 %). Eighteen genotypes from different geographical origins of Iran were used to investigate psbE–petL and trnH–psbA potential as novel barcodes to determine genetic polymorphism and characterize pomegranate genotypes. The results suggested that two regions, psbE–petL and trnH–psbA, were more suitable for determining intra-species relationships of pomegranate.





Similar content being viewed by others
References
H. Behzadi-Shahrbabaki, Genetic diversity of pomegranate genotypes in Iran. Nashr Amoozesh Keshavarzi, Iran (1998).
Mars, M. (2000). Pomegranate plant material: genetic resources and breeding, a review. Options Méditerranéennes Serie A, 42, 55–62.
Hasnoui, N., Buonamici, A., Sebastiani, F., Mars, M., Zhang, D., & Vendramin, G. (2011). Molecular genetic diversity of Punica granatum L (pomegranate) as revealed by microsatellite DNA markers (SSR). Gene, 493, 105–112.
Koehne, E. (1881). Lythraceae monographice describuntur. Botanische Jahrbücher für Systematik, 1, 142–157.
Morris, J. (2007). A molecular phylogeny of the Lythraceae and inference of the evolution of heterostyly. PhD thesis, Kent State University, Ohio.
Conti, E., Lit, A., Wilson, P. G., Graham, S. A., Brigges, B. G., Johnson, L. A. S., et al. (1997). Interfamilar relationships in Myrtales molecular phylogeny and pattern of morphological evolution. Systematic Botany, 22, 629–647.
Ozkan, Y. (2005). Investigation on physical and chemical characteristics of some pomegranate genotypes (Punica granatum L) of Tokat province in Turkey. Asian Journal of Chemistry, 17, 939–942.
Ercisli, S., Agar, G., Orhan, E., Yildirim, N., & Hizarci, Y. (2007). Interspecific variability of RAPD and fatty acid composition of some pomegranate cultivars (Punica granatum L) growing in southern Anatolia region in Turkey. Biochemical Systematics and Ecology, 35, 764–769.
Talebi, M., Bahar, M., Sharifnabi, B., & Yamchi, A. (2011). Evaluation of genetic diversity among Iranian pomegranate (Punica granatum L) cultivars, using ISSR and RAPD markers. Taxonomics Biosystematics, 8, 35–44.
Pirseyedi, S. M., Valizadehghan, S., Mardi, M., Ghaffari, M. H., Mahmoodi, P., Zahravi, M., et al. (2010). Isolation and characterization of novel microsatellite markers in pomegranate (Punica granatum L.). International Journal of Molecular Sciences, 11, 2010–2016.
Soriano, J., Zuriaga, E., Rubio, P., Llácer, G., Infante, R., & Badenes, M. (2011). Development and characterization of microsatellite markers in pomegranate (Punica granatum L). Molecular Breeding, 27, 119–128.
Parvaresh, M., Talebi, M., & Sayed-Tabatabaei, B. E. (2012). Molecular diversity and genetic relationship of pomegranate (Punica granatum L) genotypes using microsatellite markers. Scientia Horticulturae, 138, 244–252.
Jbir, R., Hasnaoui, N., Mars, M., Marrakchi, M., & Trifi, M. (2008). Characterization of Tunisian pomegranate (Punica granatum L.) cultivars using amplified fragment length polymorphism analysis. Scientia Horticulturae, 115, 231–237.
Ercisli, S., Kafkas, E., Orphan, E., Kafkas, S., Dogan, Y., & Esitken, A. (2011). Genetic characterization of pomegranate (Punica granatum L.) genotypes by AFLP markers. Biological Research, 44, 345–350.
Soleimani, M. H., Talebi, M., & Sayed-Tabatabaei, B. E. (2012). Use of SRAP markers to assess genetic diversity and population structure of wild, cultivated, and ornamental pomegranates (Punica granatum L) in different regions of Iran. Plant Systematics and Evolution, 298(6), 1141–1149.
Norouzi, M., Talebi, M., & Sayed-Tabatabaei, B. E. (2012). Chloroplast microsatellite diversity and population genetic structure of Iranian pomegranate (Punica granatum L) genotypes. Scientia Horticulturae, 137, 114–120.
Clegg, M. T., Gaut, B. S., Learn, G. H., & Morton, B. R. (1994). Rates and patterns of chloroplast DNA evolution. Proceedings of the National Academy of Sciences of the United States of America, 91, 6797–6801.
Lee, C., & Wen, J. (2004). Phylogeny of Panax using chloroplast trnC–trnD intergenic region and the utility of trnC–trnD in interspecific studies of plants. Molecular Phylogenetics and Evolution, 31, 894–903.
Kress, W. J., Wurdack, K. J., Zimmer, E. A., Weigt, L. A., & Janzen, D. H. (2005). Use of DNA barcodes to identify flowering plants. Proceedings of the National Academy of Sciences of the United States of America, 102, 8369–8374.
Ngamriabsakul, C., & Techaprasan, J. (2006). The phylogeny of Thai Boesenbergia (Zingiberaceae) based on petA–psbJ spacer (chloroplast DNA). Journal of Science and Technology, 28(1), 49–57.
Steele, P. R., Friar, L. M., Gilbert, L. E., & Jansen, R. K. (2010). Molecular systematics of the neotropical genus Psiguria (Cucurbitaceae): implication for phylogeny and species identification. American Journal of Botany, 97(1), 156–173.
Hebert, P. D. N., Ratnasingham, S., & de Waard, J. R. (2003). Barcoding animal life: cytochrome c oxidase subunit 1 divergences among closely related species. Proceedings of the Royal Society B: Biological Sciences, 270, 96–99.
Kress, W. J., & Erickson, D. L. (2007). A two-locus global DNA barcode for land plants: the coding rbcL gene complements the non-coding trnH–psbA spacer region. PLoS ONE, 2, 508.
Lahaye, R., van der Bank, M., Bogarin, D., Warner, J., & Pupulin, F. (2008). DNA barcoding the floras of biodiversity hotspots. Proceedings of the National Academy of Sciences of the United States of America, 105, 2923–2928.
Newmaster, S. G., Fazekas, A. J., Steeves, R. A. D., & Janovec, J. (2008). Testing candidate plant barcode regions in the Myristicaceae. Molecular Ecology Resources, 8, 480–490.
Thomas, C. (2009). Plant barcode soon to become reality. Science, 325, 526.
Huang, Y. L., & Shi, S. H. (2002). Phylogenetic of Lythraceae sensu lato: a preliminary analysis based on chloroplast rbcL gene, psaA–ycf3 spacer, and nuclear rDNA internal transcribed spacer sequences. International Journal of Plant Sciences, 163, 215–225.
Graham, S. W., & Olmstead, R. G. (2000). Utility of 17 chloroplast genes for inferring the phylogeny of the basal angiosperms. American Journal of Botany, 87, 1712–1730.
Murray, M. G., & Thompson, W. F. (1980). Rapid isolation of high molecular weight plant DNA. Nucleic Acids Research, 8, 4321–4325.
Melgarejo, P., Marti′nez, J. J., Herna′ndez, F., Marti′nez, R., Legua, P., Oncina, R., et al. (2009). Cultivar identification using 18S–28S rDNA intergenic spacer-RFLP in pomegranate (Punica granatum L). Scientia Horticulturae, 120, 500–503.
Cuenoud, P., Savolainen, V., & Chatrou, L. W. (2002). Molecular phylogenetics of Caryophyllales based on nuclear 18S rDNA and plastid rbcL, atpB, and matK DNA sequences. American Journal of Botany, 89, 132–144.
Technelysium Pty Ltd. (2004). Chromas. http://www.technelysium.com.au/chromas.html.
Altschul, S. F., Gish, W., Miller, W., Myers, E. W., & Lipman, D. J. (1990). Basic local alignment search tool. Journal of Molecular Biology, 215, 403–410.
Tamura, K., Peterson, N., Peterson, D., Stecher, G., Nei, M., & Kumar, S. (2011). MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Molecular Biology and Evolution, 28, 2731–2739.
Swofford, D. L. (2002). PAUP* phylogenetic analysis using parsimony (* and other methods), version 40b10. Sunderland: Sinauer.
Excoffier, L., Laval, G., & Schneider, S. (2005). Arlequin ver. 3.0: an integrated software package for population genetics data analysis. Journal of Evolutionary Bioinformatics, 1, 47–50.
Felsenstein, J. (1985). Confidence limits on phylogenies: an approach using the bootstrap. Evolution, 39, 783–791.
Brown, T. A. (2006). Gene cloning and DNA analysis, 5th edn. UK: Blackwell.
Marechal, L., Guillemaut, P., Grienenberger, J. M., & Weil, H. (1985). Sequence and codon recognition of bean mitochondria and chloroplast tRNAtrp: evidence for a high degree of homology. Nucleic Acids Research, 13(12), 4411–4416.
Hande, S. (1997). Nucleotide sequence of a cucumber chloroplast proline tRNA. Journal of Biosciences, 22, 143–147.
Marechal-Drouard, L., Weil, J. Η., & Dietrich, A. (1993). Transfer RNAs and transfer RNA genes in plant. Annual Review of Plant Physiology and Molecular Biology, 44, 13–32.
Shaw, J., Lickey, E. B., Beck, J. T., Farmer, S. B., Liu, W., Miller, J., et al. (2005). The tortoise and the hare II: Relative utility of 21 noncoding chloroplast DNA sequences for phylogenetic analysis. American Journal of Botany, 92, 142–166.
Shaw, J., Lickey, E. B., Schilling, E. E., & Small, R. L. (2007). Comparison of whole chloroplast genome sequences to choose noncoding region for phylogenetic studies in angiosperms: The tortoise and the hare III. American Journal of Botany, 94(3), 275–288.
Yan, H. F., Hao, G., Hu, C. M., & Ge, X. J. (2011). DNA barcoding in closely related species: a case study of Primula L sect Proliferae Pax (Primulaceae) in China. Journal of Systematics and Evolution, 49(3), 225–236.
Edwards, D., Horn, A., Taylor, D., Savolainen, V., & Hawkins, J. A. (2008). DNA barcoding of a large genus, Aspalathus L (Fabaceae). Taxon, 57, 1317–1327.
Ackerfield, J., & Wen, J. (2003). A morphometric analysis of Hedera L (the ivy genus, Araliaceae) and its taxonomic implications. International Journal of Plant Sciences, 164, 593–602.
Yamashiro, T., Fukuda, T., Yokoyama, J., & Maki, M. (2004). Molecular phylogeny of Vincetoxicum (Apocynaceae–Asclepiadoideae) based on the nucleotide sequences of cpDNA and nrDNAs. Molecular Phylogenetics and Evolution, 31(2), 689–7006.
Newmaster, S. G., & Ragupathy, S. (2009). Testing plant barcoding in a sister species complex of pantropical Acacia (Mimosoideae, Fabaceae). Molecular Ecology Resources, 9, 172–180.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
12033_2013_9676_MOESM2_ESM.doc
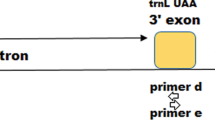
Fig. S1 Determination of gene location using BLASTn tool. petA–psaJ region (A), trnC–trnD (B), ITS (C) and trnH–psbA (D) (DOC 143 kb)
Rights and permissions
About this article
Cite this article
Hajiahmadi, Z., Talebi, M. & Sayed-Tabatabaei, B.E. Studying Genetic Variability of Pomegranate (Punica granatum L.) Based on Chloroplast DNA and Barcode Genes. Mol Biotechnol 55, 249–259 (2013). https://doi.org/10.1007/s12033-013-9676-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12033-013-9676-2