Abstract
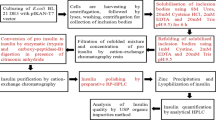
An extracellular phytase from Bacillus subtilis US417 (PHY US417) was purified and characterized. The purified enzyme of 41 kDa was calcium-dependent and optimally active at pH 7.5 and 55°C. The thermal stability of PHY US417 was drastically improved by calcium. Indeed, it recovered 77% of its original activity after denaturation for 10 min at 75°C in the presence of 5 mM CaCl2, while it retained only 22% of activity when incubated for 10 min at 60°C without calcium. In addition, PHY US417 was found to be highly specific for phytate and exhibited pH stability similar to Phyzyme, a commercial phytase with optimal activity at pH 5.5 and 60°C. The phytase gene was cloned by PCR from Bacillus subtilis US417. Sequence analysis of the encoded polypeptide revealed one residue difference from PhyC of Bacillus subtilis VTTE-68013 (substitution of arginine in position 257 by proline in PHY US417) which was reported to exhibit lower thermostability especially in the absence of calcium. With its neutral pH optimum as well as its great pH and thermal stability, the PHY US417 enzyme presumed to be predominantly active in the intestine has a high potential for use as feed additive.




Similar content being viewed by others
References
Mullaney, E. J., Daly, C. B., & Ullah, A. H. (2000). Advances in phytase research. Advances in Applied Microbiology, 47, 157–199. doi:10.1016/S0065-2164(00)47004-8.
Eeckhout, W., & De Paepe, M. (1994). Total phosphorus, phytate-phosphorus and phytase activity in plant feedstuffs. Animal Feed Science and Technology, 47, 19–29. doi:10.1016/0377-8401(94)90156-2.
Harland, B. F., & Oberleas, D. (1999). Phytic acid complex in feed ingredients. In M. B. Coelho & E. T. Kornegay (Eds.), Phytase in animal Nutrition and Waste management (pp. 69–75). Mount Olive, NJ: BASF Reference Manual, BASF Corporation.
Nelson, T. S. (1967). The utilization of phytate phosphorus by poultry—a review. Poultry Science, 46(4), 862–871.
Nys, Y., Frapin, D., & Pointillart, A. (1996). Occurence of phytase in plants, animals, and microorganisms. In M. B. Coelho & E. T. Kornegay (Eds.), Phytase in animal nutrition and waste management (pp. 213–236). Mount Olive, NJ: BASF Reference Manual, BASF Corporation.
Wodzinski, R. J., & Ullah, A. H. (1996). Phytase. Advances in Applied Microbiology, 42, 263–302. doi:10.1016/S0065-2164(08)70375-7.
Maga, J. A. (1982). Phytate: Its chemistry, occurrence, food interactions, nutritional significance, and methods of analysis. Journal of Agricultural and Food Chemistry, 30, 1–9. doi:10.1021/jf00109a001.
Bali, A., & Satyanarayana, T. (2001). Microbial phytases in nutrition and combating phosphorus pollution. Everyman’s Science, 4, 207–209.
Lassen, S. F., Breinholt, J., Ostergaard, P. R., Brugger, R., Bischoff, A., Wyss, M., et al. (2001). Expression, gene cloning, and characterization of five novel phytases from four basidiomycete fungi: Peniophora lycii, Agrocybe pediades, a Ceriporia sp., and Trametes pubescens. Applied and Environmental Microbiology, 67, 4701–4707. doi:10.1128/AEM.67.10.4701-4707.2001.
Leske, K. L., & Coon, C. N. (1999). A bioassay to determine the effect of phytase on phytate phosphorus hydrolysis and total phosphorus retention of feed ingredients as determined with broilers and laying hens. Poultry Science, 78(8), 1151–1157.
Maenz, D. D. (2001). Enzymatic characteristics of phytases as they relate to their use in animal feeds. In M. R. Bedford & G. G. Partridge (Eds.), Enzymes in farm animal nutrition (pp. 61–84). Wallingford, UK: CABI Publishing, CAB International.
Cowieson, A. J., Acamovic, T., & Bedford, M. R. (2006). Supplementation of corn-soy-based diets with an Escherichia coli-derived phytase: Effects on broiler chick performance and the digestibility of amino acids and metabolizability of minerals and energy. Poultry Science, 85(8), 1389–1397.
Jendza, J. A., Dilger, R. N., Sands, J. S., & Adeola, O. (2006). Efficacy and equivalency of an Escherichia coli-derived phytase for replacing inorganic phosphorus in the diets of broiler chickens and young pigs. Journal of Animal Science, 84, 3364–3374. doi:10.2527/jas.2006-212.
Wu, G., Liu, Z., Bryant, M. M., & Roland, D. A., Sr. (2006). Comparison of Natuphos and Phyzyme as phytase sources for commercial layers fed corn-soy diet. Poultry Science, 85, 64–69.
Elkhalil, E. A., Manner, K., Borriss, R., & Simon, O. (2007). In vitro and in vivo characteristics of bacterial phytases and their efficacy in broiler chickens. British Poultry Science, 48, 64–70. doi:10.1080/00071660601148195.
Pasamontes, L., Haiker, M., Wyss, M., Tessier, M., & van Loon, A. P. (1997). Gene cloning, purification, and characterization of a heat-stable phytase from the fungus Aspergillus fumigatus. Applied and Environmental Microbiology, 63, 1696–1700.
Kim, Y. O., Kim, H. K., Bae, K. S., Yu, J. H., & Oh, T. K. (1998). Purification and properties of a thermostable phytase from Bacillus sp. DS11. Enzyme and Microbial Technology, 22, 2–7. doi:10.1016/S0141-0229(97)00096-3.
Lehmann, M., Pasamontes, L., Lassen, S. F., & Wyss, M. (2000). The consensus concept for thermostability engineering of proteins. Biochimica et Biophysica Acta, 1543, 408–415.
Simon, O., & Igbasan, F. (2002). In vitro properties of phytases from various microbial origins. International Journal of Food Science and Technology, 37, 813–822. doi:10.1046/j.1365-2621.2002.00621.x.
Haefner, S., Knietsch, A., Scholten, E., Braun, J., Lohscheidt, M., & Zelder, O. (2005). Biotechnological production and applications of phytases. Applied Microbiology and Biotechnology, 68, 588–597. doi:10.1007/s00253-005-0005-y.
Vohra, A., & Satyanarayana, T. (2003). Phytases: microbial sources, production, purification, and potential biotechnological applications. Critical Reviews in Biotechnology, 23(1), 29–60. doi:10.1080/713609297.
Powar, V. K., & Jagannathan, V. (1982). Purification and properties of phytate-specific phosphatase from Bacillus subtilis. Journal of Bacteriology, 151, 1102–1108.
Shimizu, M. (1992). Purification and characterization of phytase from Bacillus subtilis (natto) N-77. Bioscience, Biotechnology, and Biochemistry, 56, 1266–1269.
Kerovuo, J., Lauraeus, M., Nurminen, P., Kalkkinen, N., & Apajalahti, J. (1998). Isolation, characterization, molecular gene cloning, and sequencing of a novel phytase from Bacillus subtilis. Applied and Environmental Microbiology, 64, 2079–2085.
Choi, Y. M., Suh, H. J., & Kim, J. M. (2001). Purification and properties of extracellular phytase from Bacillus sp. KHU-10. Journal of Protein Chemistry, 20, 287–292. doi:10.1023/A:1010945416862.
Gulati, H. K., Chadha, B. S., & Saini, H. S. (2007). Production and characterization of thermostable alkaline phytase from Bacillus laevolacticus isolated from rhizosphere soil. Journal of Industrial Microbiology and Biotechnology, 34, 91–98. doi:10.1007/s10295-006-0171-7.
Idriss, E. E., Makarewicz, O., Farouk, A., Rosner, K., Greiner, R., Bochow, H., et al. (2002). Extracellular phytase activity of Bacillus amyloliquefaciens FZB45 contributes to its plant-growth-promoting effect. Microbiology, 148, 2097–2109.
Bae, H. D., Yanke, L. J., Cheng, K. J., & Selinger, L. B. (1999). A novel staining method for detecting phytase activity. Journal of Microbiological Methods, 39, 17–22. doi:10.1016/S0167-7012(99)00096-2.
Sambrook, J., Fritsch, E. F., & Maniatis, T. (1989). Molecular cloning: A laboratory manual (2nd edn.). Cold Spring Harbor, NY: Cold Spring Harbor Laboratory Press.
Gurtler, V., & Stanisich, V. A. (1996). New approaches to typing and identification of bacteria using the 16S-23S rDNA spacer region. Microbiology, 142(1), 3–16.
Laemmli, U. K. (1970). Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature, 227, 680–685. doi:10.1038/227680a0.
Engelen, A. J., van der Heeft, F. C., Randsdorp, P. H., & Smit, E. L. (1994). Simple and rapid determination of phytase activity. Journal of AOAC International, 77, 760–764.
Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry, 72, 248–254. doi:10.1016/0003-2697(76)90527-3.
Wang, Y., Yao, B., Zeng, H., Shi, X., Cao, S., Yuan, T., et al. (2001). Purification and properties of neutral phytase form Bacillus subtilis. Wei sheng wu xue bao, 41, 198–203.
Nielsen, H., Engelbrecht, J., Brunak, S., & von Heijne, G. (1997). Identification of prokaryotic and eukaryotic signal peptides and prediction of their cleavage sites. Protein Engineering, 10, 1–6. doi:10.1093/protein/10.1.1.
Ha, N. C., Oh, B. C., Shin, S., Kim, H. J., Oh, T. K., Kim, Y. O., et al. (2000). Crystal structures of a novel, thermostable phytase in partially and fully calcium-loaded states. Nature Structural Biology, 7, 147–153. doi:10.1038/72421.
Oh, B. C., Chang, B. S., Park, K. H., Ha, N. C., Kim, H. K., Oh, B. H., et al. (2001). Calcium-dependent catalytic activity of a novel phytase from Bacillus amyloliquefaciens DS11. Biochemistry, 40, 9669–9676. doi:10.1021/bi010589u.
Tye, A. J., Siu, F. K., Leung, T. Y., & Lim, B. L. (2002). Molecular cloning and the biochemical characterization of two novel phytases from B. subtilis 168 and B. licheniformis. Applied Microbiology and Biotechnology, 59, 190–197. doi:10.1007/s00253-002-1033-5.
Park, S. C., Choi, Y. W., & Oh, T. K. (1999). Comparative enzymatic hydrolysis of phytate in various animal feedstuff with two different phytases. Journal of Veterinary Medical Science, 61, 1257–1259. doi:10.1292/jvms.61.1257.
Kim, Y. O., Lee, J. K., Kim, H. K., Yu, J. H., & Oh, T. K. (1998). Cloning of the thermostable phytase gene (phy) from Bacillus sp. DS11 and its overexpression in Escherichia coli. FEMS Microbiology Letters, 162, 185–191. doi:10.1111/j.1574-6968.1998.tb12997.x.
Kerovuo, J., Lappalainen, I., & Reinikainen, T. (2000). The metal dependence of Bacillus subtilis phytase. Biochemical and Biophysical Research Communications, 268, 365–369. doi:10.1006/bbrc.2000.2131.
Shin, S., Ha, N. C., Oh, B. C., Oh, T. K., & Oh, B. H. (2001). Enzyme mechanism and catalytic property of beta propeller phytase. Structure, 9, 851–858. doi:10.1016/S0969-2126(01)00637-2.
Acknowledgments
This research was funded by the Tunisian Government “Contrat Programme CBS-LEMP” and the local Company “Nutrisud” through a research agreement. Part of the work was also supported by the CMCU project (2007–2009) no. 07G0922 “CHOUAYEKH/MAGUIN”. The authors are grateful to Dr. Ahmed Rebai for his kind assistance in the statistical analysis of the pH data. We would like also to express our gratitude to Dr. Emmanuelle Maguin, Dr. Marie-Joëlle Virolle, and Dr. Mamdouh Ben Ali for useful discussion. Many thanks as well to Mr. Badr Abdelhedi, Mr. Amine Mrabet, and Mr. Dhiaeddine Hachicha for their permanent collaboration.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Farhat, A., Chouayekh, H., Ben Farhat, M. et al. Gene Cloning and Characterization of a Thermostable Phytase from Bacillus subtilis US417 and Assessment of its Potential as a Feed Additive in Comparison with a Commercial Enzyme. Mol Biotechnol 40, 127–135 (2008). https://doi.org/10.1007/s12033-008-9068-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12033-008-9068-1