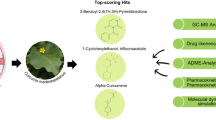
Abstract
This study explores the therapeutic potential of phytochemicals derived from Morus alba for colorectal cancer (CRC) treatment. Colorectal cancer is a global health concern with increasing mortality rates, necessitating innovative strategies for prevention and therapy. Employing in silico analysis, molecular docking techniques (MDT), and molecular dynamics simulations (MDS), the study investigates the interactions between Morus alba-derived phytochemicals and key proteins (AKT1, Src, STAT3, EGFR) implicated in CRC progression. ADME/T analysis screens 78 phytochemicals for drug-like and pharmacokinetic properties. The study integrates Lipinski’s Rule of Five and comprehensive bioactivity assessments, providing a nuanced understanding of Morus alba phytoconstituent’s potential as CRC therapeutic agents. Notably, 14 phytochemicals out of 78 emerge as potential candidates, demonstrating oral bioavailability and favorable bioactivity scores. Autodock 1.5.7 is employed for energy minimization followed by molecular docking with the highest binding energy observed to be − 11.7 kcal/mol exhibited by Kuwanon A against AKT1. Molecular dynamics simulations and trajectory path analysis were conducted between Kuwanon A and AKT1 at the Pleckstrin homology (PH) domain region (TRP80), revealing minimal deviations. In comparison to the standard drug Capivasertib, the phytochemical Kuwanon A emerges as a standout candidate based on computational analysis. This suggests its potential as an alternative to mitigate the limitations associated with the standard drug. The research aims to provide insights for future experimental validations and to stimulate the development of Kuwanon A as a novel, effective therapeutic agent for managing colorectal cancer.
Graphical abstract









Similar content being viewed by others
Data availability
Upon request, the data will be made accessible.
Code availability
Not applicable.
Abbreviations
- CRC:
-
Colorectal cancer
- PH:
-
Pleckstrin homology
- HDI:
-
Human development index
- AKT:
-
Serine/Threonine protein kinase
- STAT3:
-
Signal transducer and activator of transcription 3
- EGFR:
-
Epidermal Growth Factor Receptor
- Src:
-
Protooncogene Tyrosine Protein Kinase
- DNJ:
-
1-Deoxynojirimycin
- MEMA:
-
Morus alba L. root bark extract
- SREBF2:
-
Sterol regulatory element binding transcription factor 2
- FOXO3a:
-
Morusinol facilitated forkhead box O3
- MDT:
-
Molecular docking techniques
- MDS:
-
Molecular dynamics simulations
- RMSD:
-
Root mean square deviation
- MW:
-
Molecular weight
- NHA:
-
Number of hydrogen bond acceptor
- NHD:
-
Number of hydrogen bond donor
- NRB:
-
Number of rotatable bonds
- TPSA:
-
Total polar surface area
- NVT:
-
Number of molecules, volume, temperature
- NPT:
-
Number of molecules, pressure, temperature
- VMD:
-
Visual Molecular dynamics
References
Sawicki T, Ruszkowska M, Danielewicz A, Niedźwiedzka E, Arłukowicz T, Przybyłowicz KE. A review of colorectal cancer in terms of epidemiology, risk factors, development, symptoms and diagnosis. Cancers (Basel). 2021. https://doi.org/10.3390/cancers13092025.
Arnold M, Sierra MS, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global patterns and trends in colorectal cancer incidence and mortality. Gut. 2016. https://doi.org/10.1136/gutjnl-2015-310912.
Douaiher J, Ravipati A, Grams B, Chowdhury S, Alatise O, Are C. Colorectal cancer—global burden, trends, and geographical variations. J Surg Oncol. 2017;115:619–30.
Wong MCS, Huang J, Lok V, Wang J, Fung F, Ding H, et al. Differences in incidence and mortality trends of colorectal cancer worldwide based on sex, age, and anatomic location. Clin Gastroenterol Hepatol. 2021;19:955–66.
Hossain MS, Karuniawati H, Jairoun AA, Urbi Z, Ooi DJ, John A, et al. Colorectal cancer: a review of carcinogenesis, global epidemiology, current challenges, risk factors, preventive and treatment strategies. Cancers (Basel). 2022. https://doi.org/10.3390/cancers14071732.
Botteri E, Iodice S, Bagnardi V, Raimondi S, Lowenfels AB, Maisonneuve P. Smoking and colorectal cancer: a meta-analysis. JAMA. 2008;300:2765–78.
Bagnardi V, Rota M, Botteri E, Tramacere I, Islami F, Fedirko V, et al. Alcohol consumption and site-specific cancer risk: a comprehensive dose–response meta-analysis. Br J Cancer. 2015;112:580–93.
Freisling H, Arnold M, Soerjomataram I, O’Doherty MG, Ordóñez-Mena JM, Bamia C, et al. Comparison of general obesity and measures of body fat distribution in older adults in relation to cancer risk: meta-analysis of individual participant data of seven prospective cohorts in Europe. Br J Cancer. 2017;116:1486–97.
Vieira AR, Abar L, Chan DSM, Vingeliene S, Polemiti E, Stevens C, et al. Foods and beverages and colorectal cancer risk: a systematic review and meta-analysis of cohort studies, an update of the evidence of the WCRF-AICR Continuous Update Project. Ann Oncol. 2017;28:1788–02.
C NCG. National institute for health and care excellence: clinical guidelines. Chronic Kidney Dis. (Partial Updat. Early Identif. Manag. Chronic Kidney Dis. Adults Prim. Second. Care. National Institute for Health and Care Excellence (UK) Copyright{\copyright} National~…; 2014. p. 2014.
suspected cancer | Search results | NICE [Internet]. [cited 2024 Jan 29]. Available from: https://www.nice.org.uk/search?q=suspected+cancer&gst=Published
Schreuders EH, Ruco A, Rabeneck L, Schoen RE, Sung JJY, Young GP, et al. Colorectal cancer screening: a global overview of existing programmes. Gut. 2015. https://doi.org/10.1136/gutjnl-2014-309086.
Hua H, Zhang H, Chen J, Wang J, Liu J, Jiang Y. Targeting Akt in cancer for precision therapy. J Hematol Oncol. 2021;14:128. https://doi.org/10.1186/s13045-021-01137-8.
Jin W. Regulation of Src family kinases during colorectal cancer development and its clinical implications. Cancers (Basel). 2020. https://doi.org/10.3390/cancers12051339.
Lin Y, He Z, Ye J, Liu Z, She X, Gao X, et al. Progress in understanding the IL-6/STAT3 pathway in colorectal cancer. Onco Targets Ther. 2020;13:13023–32. https://doi.org/10.2147/OTT.S278013.
Lawal B, Wang Y-C, Wu ATH, Huang H-S. Pro-oncogenic c-Met/EGFR, biomarker signatures of the tumor microenvironment are clinical and therapy response prognosticators in colorectal cancer, and therapeutic targets of 3-Phenyl-2H-benzo[e][1,3]-Oxazine-2,4(3H)-Dione Derivatives. Front Pharmacol. 2021. https://doi.org/10.3389/fphar.2021.691234.
Atanasov AG, Waltenberger B, Pferschy-Wenzig E-M, Linder T, Wawrosch C, Uhrin P, et al. Discovery and resupply of pharmacologically active plant-derived natural products: a review. Biotechnol Adv. 2015;33:1582–614.
Hussain F, Rana Z, Shafique H, Malik A, Hussain Z. Phytopharmacological potential of different species of Morus alba and their bioactive phytochemicals: A review. Asian Pac J Trop Biomed. 2017;7:950–6.
Khan T, Ali M, Khan A, Nisar P, Jan SA, Afridi S, et al. Anticancer plants: a review of the active phytochemicals, applications in animal models, and regulatory aspects. Biomolecules. 2019. https://doi.org/10.3390/biom10010047.
Yamamoto K, Sakamoto Y, Mizowaki Y, Iwagaki Y, Kimura T, et al. Intake of mulberry 1-deoxynojirimycin prevents colorectal cancer in mice. J Clin Biochem Nutr. 2017;61:47–52.
Park H-J, Park S-H. Root bark of Morus alba L. Induced p53-independent apoptosis in human colorectal cancer cells by suppression of STAT3 activity. Nutr Cancer. 2022;74:1837–48. https://doi.org/10.1080/01635581.2021.1968444.
Zhang X, Dong Z, Yang Y, Liu C, Li J, Sun W, et al. Morusinol extracted from Morus alba inhibits cell proliferation and induces autophagy via FOXO3a nuclear accumulation-mediated cholesterol biosynthesis obstruction in colorectal cancer. J Agric Food Chem. 2023;71:16016–31.
Van Der Spoel D, Lindahl E, Hess B, Groenhof G, Mark AE, Berendsen HJC. GROMACS: Fast, flexible, and free. J Comput Chem. 2005;26:1701–18. https://doi.org/10.1002/jcc.20291.
Lipinski CA. Lead- and drug-like compounds: the rule-of-five revolution. Drug Discov Today Technol. 2004;1:337–41.
Daina A, Michielin O, Zoete V. SwissADME: a free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci Rep. 2017;7:42717. https://doi.org/10.1038/srep42717.
Daina A, Zoete V. A BOILED-egg to predict gastrointestinal absorption and brain penetration of small molecules. ChemMedChem. 2016;11:1117–21. https://doi.org/10.1002/cmdc.201600182.
Amin ML. P-glycoprotein inhibition for optimal drug delivery. Drug Target Insights. 2013. https://doi.org/10.33393/dti.2013.1349.
Rong S-B, Hu Y, Enyedy I, Powis G, Meuillet EJ, Wu X, et al. Molecular modeling Studies of the Akt PH Domain and Its Interaction with phosphoinositides. J Med Chem. 2001;44:898–908. https://doi.org/10.1021/jm000493i.
Banerji U, Dean EJ, Pérez-Fidalgo JA, Batist G, Bedard PL, You B, et al. A phase I open-label study to identify a dosing regimen of the pan-AKT inhibitor AZD5363 for evaluation in solid tumors and in PIK3CA-Mutated breast and gynecologic cancers. Clin Cancer Res. 2018;24:2050–9. https://doi.org/10.1158/1078-0432.CCR-17-2260.
Su J, Thakur A, Pan G, Yan J, Gaurav I, Thakur S, et al. Morus alba derived Kuwanon-A combined with 5-fluorouracil reduce tumor progression via synergistic activation of GADD153 in gastric cancer. MedComm – Oncol. 2023;2:e24. https://doi.org/10.1002/mog2.24.
Acknowledgements
The authors are thankful to Vellore Institute of Technology, Vellore for providing research facilities in School of Bio-Sciences and Technology.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
BS, SM, and KVBR conceptualized and orchestrated the research, contributing to its design. BS, SM, and KVBR conducted the experimental procedures and collaborated on drafting the manuscript. All authors meticulously reviewed and granted their approval for the final manuscript.
Corresponding author
Ethics declarations
Competing interest
The authors have no conflicts of interest to disclose.
Ethical approval
Not applicable.
Consent to participant
Not applicable.
Consent to publication
All authors meticulously reviewed.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Stany, B., Mishra, S. & Rao, K.V.B. Pharmacokinetic studies, molecular docking, and molecular dynamics simulations of phytochemicals from Morus alba: a multi receptor approach for potential therapeutic agents in colorectal cancer. Med Oncol 41, 156 (2024). https://doi.org/10.1007/s12032-024-02406-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-024-02406-5