Abstract
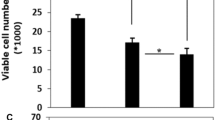
Chemoresistance is the leading cause of limiting long-term treatment success in cancer cells. Anticancer drugs usually kill cells through apoptosis induction and defects in this signaling pathway lead to chemoresistance. Apoptotic protease activating factor 1 regulates cellular stress evoked by chemotherapeutic agents through facilitating apoptosome assembling but can be degraded by proteasome. This study examined the role of proteasome inhibitor Bortezomib in the cytotoxic effects of Docetaxel on MCF7 cells response and its correlation with Apaf-1 expression level. MTT assay, caspase 3/7 activity assay, propidium iodide staining, adenosine triphosphate and reactive oxygen species amount measurements were utilized to demonstrate the role of Bortezomib in Docetaxel efficacy with and without Apaf-1 overexpressing. Meanwhile, two-dimensional cell migration assay was performed by scratch wound assay. The combination of Docetaxel with Bortezomib was significantly more cytotoxic compared single drug, more effectively delayed cell growth, reduced ATP level and increased ROS production. In Apaf-1 overexpressing, Docetaxel was more efficient in preventing cell migration, however, Docetaxel plus Bortezomib were not significantly effective; and fluorescence images supported the interpretation. Our findings demonstrated MCF7 resistance to Docetaxel is due in part to low Apaf-1 level and Apaf-1 overexpression resulted in the increase of cell susceptibility to Docetaxel stimulus. We assume that proteasome inhibitor may restore apoptotic proteins like Apaf-1 and prevent the degradation of cytosolic cytochrome c released by Docetaxel, consequently triggering intrinsic apoptosis and promoting cancer cell death. Collectively, treating MCF7 breast cells with proteasome inhibitor sensitizes cells to Docetaxel-induced apoptosis and possibly overcomes chemoresistance.




Similar content being viewed by others
References
Kerr F, Wyllie AH, Currie AR. Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br J Cancer. 1972;26:239–57. https://doi.org/10.1038/bjc.1972.33.
Yuan J, Yankner BA. Apoptosis in the nervous system. Nature. 2000;407:802–9. https://doi.org/10.1046/j.1471-4159.2000.0740001.x.
Brenner D, Mak TW. Mitochondrial cell death effectors. Curr Opin Cell Biol. 2009;21:871–7. https://doi.org/10.1016/j.ceb.2009.09.004.
Li P, Nijhawan D, Budihardjo I, et al. Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell. 1997;91:479–89. https://doi.org/10.1016/s0092-8674(00)80434-1.
Zou H, Henzel WJ, Liu X, Lutschg A, Wang X. Apaf-1, a human protein homologous to C. elegans CED-4, participates in cytochrome c-dependent activation of caspase-3. Cell. 1997;90:405–13. https://doi.org/10.1016/s0092-8674(00)80501-2.
Green DR, Kroemer G. Pharmacological manipulation of cell death: clinical applications in sight? J Clin Invest. 2005;15:2610–7. https://doi.org/10.1172/JCI26321.
Fox F, MacFarlane M. Targeting cell death signalling in cancer: minimising ‘Collateral damage.’ Br J Cancer. 2016;115:5–11. https://doi.org/10.1038/bjc.2016.111.Epub2016May3.
Jia L, Macey MG, Yin Y, Newland AC, Kelsey SM. Subcellular distribution and redistribution of Bcl-2 family proteins in human leukemia cells undergoing apoptosis. Blood. 1999;93:2353–9.
El-Khattouti A, Selimovic D, Haikel Y, Hassan M. Crosstalk between apoptosis and autophagy: molecular mechanisms and therapeutic strategies in cancer. J Cell Death. 2013;6:37–55. https://doi.org/10.4137/JCD.S11034.
Long JS, Ryan KM. New frontiers in promoting tumor cell death: targeting apoptosis, necroptosis and autophagy. Oncogene. 2012;31:5045–60. https://doi.org/10.1038/onc.2012.7.Epub2012Feb6.
Zhang HG, Wang J, Yang X, Hsu HC, Mountz JD. Regulation of apoptosis proteins in cancer cells by ubiquitin. Oncogene. 2004;23:2009–15. https://doi.org/10.1038/sj.onc.1207373.
Frankland-Searby S, Bhaumik SR. The 26S proteasome complex: an attractive target for cancer therapy. Biochem Biophys Acta. 2012;1825:64–76. https://doi.org/10.1016/j.bbcan.2011.10.003.
Rock KL, Gramm C, Rothstein L, Clark K, Stein R, Dick L, Hwang D, Goldberg AL. Inhibitors of the proteasome block the degradation of most cell proteins and the generation of peptides presented on MHC class I molecules. Cell. 1994;78:761. https://doi.org/10.1016/S0092-8674(94)90462-6.
Kisselev AF, Goldberg AL. Proteasome inhibitors: from research tools to drug candidates. Chem Biol. 2001;8:739–58. https://doi.org/10.1016/S1074-5521(01)00056-4.
Adams J, Kauffman M. Development of the proteasome inhibitor Velcade (Bortezomib). Cancer Invest. 2004;22:304–11. https://doi.org/10.1081/cnv-120030218.
Shah SA, Potter MW, McDade TP, Ricciardi R, Perugini RA, Elliott PJ, Adams J, Callery MP. 26S proteasome inhibition induces apoptosis and limits growth of human pancreatic cancer. J Cell Biochem. 2001;82:110–22. https://doi.org/10.1002/jcb.1150.
Jones SE, Erban J, Overmoyer B, et al. Randomized phase III study of docetaxel compared with paclitaxel in metastatic breast cancer. J Clin Oncol. 2005;23:5542–51. https://doi.org/10.1200/JCO.2005.02.027.
Eisenhauer EA, Vermorken JB. The toxoids. Comparative clinical pharmacology and therapeutic potential. Drugs. 1998;55:5–30. https://doi.org/10.2165/00003495-199855010-00002.
Karimzadeh S, Hosseinkhani S, Fathi A, Ataei F, Baharvand H. Insufficient Apaf-1 expression in early stages of neural differentiation of human embryonic stem cells might protect them from apoptosis. Eur J Cell Biol. 2018;97:126–35. https://doi.org/10.1016/J.EJCB.2018.01.005.
Amroudie MN, Ataei F. Experimental and theoretical study of IBC domain from human IP3R2; molecular cloning, bacterial expression and protein purification. Int J Biol Macromol. 2019;124:1321–7. https://doi.org/10.1016/j.ijbiomac.2018.09.117.
Ataei F, Torkzadeh-Mahani M, Hosseinkhani S. A novel luminescent biosensor for rapid monitoring of IP3 by split-luciferase complementary assay. Biosen Bioelectron. 2013;41:642–8. https://doi.org/10.1016/J.BIOS.2012.09.037.
Abazari R, Mahjoub AR, Ataei F, Morsali A, Carpenter-Warren CL, Mehdizadeh K, Slawin AMZ. Chitosan immobilization on Bio-MOF nanostructures: a biocompatible pH-responsive nanocarrier for doxorubicin release on MCF-7 cell lines of human breast cancer. Inorg Chem. 2018;57:13364–79. https://doi.org/10.1021/acs.inorgchem.8b01955.
Mehdizadeh K, Ataei F, Hosseinkhani S. Effects of doxorubicin and docetaxel on susceptibility to apoptosis in high expression level of survivin in HEK and HEK-S cell lines as in vitro models. Biochem Biophys Res Commun. 2020;532:139–44. https://doi.org/10.1016/j.bbrc.2020.08.028.
Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248–54. https://doi.org/10.1006/abio.1976.9999.
Pakravan K, Babashah S, Mowla SJ, Mossahebi-Mohammad M, Ataei F, Dana N, Javan M. MicroRNA-100 shuttled by mesenchymal stem cell-derived exosomes suppresses in vitro angiogenesis through modulating the mTOR/HIF-1α/VEGF signaling axis in breast cancer cells. Cell Oncol. 2017;40:457–70. https://doi.org/10.1007/s13402-017-0335-7.
Wang X, Luo H, Chen H, Duguid W, Wu J. Role of proteasomes in T cell activation and proliferation. J Immunol. 1998;160:788–801.
Marshansky V, Wang X, Bertrand R, Luo H, Duguid W, Chinnadurai G, Kanaan N, Diem VM, Wu J. Proteasomes modulate balance among proapoptotic and antiapoptotic Bcl-2 family members and compromise functioning of the electron transport chain in leukemic cells. J Immunol. 2001;166:3130–42. https://doi.org/10.4049/jimmunol.166.5.3130.
Zamaraeva MV, Sabirov RZ, Maeno E, Ando-Akatsuka Y, Bessonova SV, Okada Y. Cells die with increased cytosolic ATP during apoptosis: a bioluminescence study with intracellular luciferase. Cell Death Differ. 2005;12:1390–7. https://doi.org/10.1038/sj.cdd.4401661.
Le Bras M, Clément MV, Pervaiz S, Brenner C. Reactive oxygen species and the mitochondrial signaling pathway of cell death. Histol Histopathol. 2005;20:205–19. https://doi.org/10.14670/HH-20.205.
Liang CC, Park AY, Guan JL. In vitro scratch assay: a convenient and inexpensive method for analysis of cell migration in vitro. Nat Protoc. 2007;2:329–33. https://doi.org/10.1038/nprot.2007.30.
Houshdarpour R, Ataei F, Hosseinkhani S. Efficient stable cell line generation of survivin as an in vitro model for specific functional analysis in apoptosis and drug screening. Mol Biotechnol. 2021. https://doi.org/10.1007/s12033-021-00313-y.
Richardson PG, Barlogie B, Berenson J, et al. A phase 2 study of bortezomib in relapsed, refractory myeloma. N Engl J Med. 2003;348:2609–17. https://doi.org/10.1056/NEJMoa030288.
Adams J, Palombella VJ, Elliott PJ. Proteasome inhibition: a new strategy in cancer treatment. Invest New Drugs. 2000;18:109–21. https://doi.org/10.1023/a:1006321828515.
Messersmith WA, Baker SD, Lassiter L, Sullivan RA, Dinh K, Almuete VI, Wright JJ, Donehower RC, Carducci MA, Armstrong DK. Phase I trial of bortezomib in combination with docetaxel in patients with advanced solid tumors. Clin Cancer Res. 2006;12:1270–5. https://doi.org/10.1158/1078-0432.CCR-05-1942.
Tsujimoto Y. Apoptosis and necrosis: intracellular ATP level as a determinant for cell death modes. Cell Death Differ. 2007;4:429–34. https://doi.org/10.1038/sj.cdd.4400262.
Panieri E, Santoro MM. ROS homeostasis and metabolism: a dangerous liason in cancer cells. Cell Death Dis. 2016;9:e2253. https://doi.org/10.1038/cddis.2016.105.
Brentnall M, Rodriguez-Menocal L, De Guevara RL, Cepero E, Boise LH. Caspase-9, caspase-3 and caspase-7 have distinct roles during intrinsic apoptosis. BMC Cell Biol. 2013;9:32. https://doi.org/10.1186/1471-2121-14-32.
Essmann F, Engels IH, Totzke G, Schulze-Osthoff K, Janicke RU. Apoptosis resistance of MCF-7 breast carcinoma cells to ionizing radiation is independent of p53 and cell cycle control but caused by the lack of caspase-3 and a caffeine-inhibitable event. Cancer Res. 2004;64:7065–72. https://doi.org/10.1158/0008-5472.CAN-04-1082.
Janick RU. MCF-7 breast cancer cells do not express caspase-3. Breast Cancer Res Treat. 2009;117:219–21. https://doi.org/10.1007/s10549-008-0217-9.
Liang Y, Yan C, Schor NF. Apoptosis the in the absence of caspase 3. Oncogene. 2001;20:6570–8. https://doi.org/10.1038/sj.onc.1204815.
Anichini A, Mortarini R, Sensi M, Zanon M. APAF-1 signaling in human melanoma. Cancer Lett. 2006;238:168–79. https://doi.org/10.1016/j.canlet.2005.06.034.
Gama V, et al. PARC/CUL9 mediates the degradation of mitochondrial-released cytochrome c and promotes survival in neurons and cancer cells. Sci Signal. 2014;7:ra67. https://doi.org/10.1126/scisignal.2005309.
Acknowledgements
We would like to thank the research council of Tarbiat Modares University through a grant (#D/2003) to cell death and differentiation research program, and Iranian National Science Foundation (INSF, project 93043373) for the financial support of this investigation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There is no conflict of interest to disclose.
Informed consent
All authors agree to submit this article for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mehdizadeh, K., Ataei, F. & Hosseinkhani, S. Treating MCF7 breast cancer cell with proteasome inhibitor Bortezomib restores apoptotic factors and sensitizes cell to Docetaxel. Med Oncol 38, 64 (2021). https://doi.org/10.1007/s12032-021-01509-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-021-01509-7