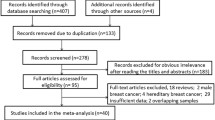
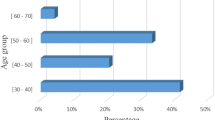
Abstract
The development of breast cancer is a multistep process associated with complex changes in host gene expression patterns including inactivation of tumor suppressor genes and activation of oncogenes. Critically, hereditary predisposition plays a significant role in cancer susceptibility. However, mutation of the BRCA1 gene is found only in the minority of hereditary breast cancer, which indicates that there might be alternative, novel mechanisms contributing to inactivation of the BRCA1 gene. Studies have shown that aberrant methylation of genomic DNA plays an important role in carcinogenesis. The aim of this study was to investigate whether DNA methylation may be an alternative mechanism for the inactivation of BRCA1 as an epigenetic modification of the genome and whether hereditary breast cancer has a different BRCA1 methylation phenotype pattern than sporadic breast cancer. The pattern of CpG island methylation within the promoter region of BRCA1 was assessed by bisulfite sequencing DNA from peripheral blood cells of 72 patients with hereditary predisposition but without BRCA1 mutations and 30 sporadic breast cancer controls. The overall methylation level in patients with hereditary predisposition was significantly lower than that in the sporadic control group. However, patients with hereditary predisposition showed a significantly higher methylation susceptibility for the sites −518 when compared to controls. These results suggest that there might be different BRCA1 promoter methylation levels and patterns in sporadic and hereditary breast cancer in peripheral blood DNA. These findings may facilitate the early diagnosis of hereditary breast cancer.



Similar content being viewed by others
References
Ahmedin J, Freddie B, Melissa M, Jacques F, Elizabeth W, David F. Global cancer statistics. CA Cancer J Clin. 2011;61(2):69–90.
James CR, Quinn JE, Mullan PB, Johnston PG, Harkin DP. BRCA1, a potential predictive biomarker in the treatment of breast cancer. Oncologist. 2007;12(2):142–50.
Pasche B. Recent advances in breast cancer genetics. Cancer Treat Res. 2008;141:1–10.
Miki Y, Swensen J, Shattuck-Eidens D, Futreal PA, Harshman K, Tavtigian S. A strong candidate for the breast and ovarian cancer susceptibility gene BRCA1. Science. 1994;266(5182):66–71.
Andrieu N, Goldgar DE, Easton DF, Rookus M, Brohet R, Antoniou AC. Pregnancies, breast-feeding, and breast cancer risk in the international BRCA1/2 carrier cohort study (IBCCS). J Natl Cancer Inst. 2006;98(8):535–44.
Gorski JJ, Kennedy RD, Hosey AM, Harkin DP. The complex relationship between BRCA1 and ERα in hereditary breast cancer. Clin Cancer Res. 2009;15(5):1514–8.
Xu X, Gammon MD, Zhang Y, et al. BRCA1 promoter methylation is associated with increased mortality among women with breast cancer. Breast Cancer Res Treat. 2009;115(2):397–404.
Jones PA, Baylin SB. The epigenomics of cancer. Cell. 2007;128(4):683–92.
Brooks J, Cairns P, Zeleniuch-Jacquotte A. Promoter methylation and the detection of breast cancer. Cancer Causes Control. 2009;20(9):1539–50.
Bird A. DNA methylation patterns and epigenetic memory. Genes Dev. 2002;16(1):6–21.
Rice JC, Ozcelik H, Maxeiner P, Andrulis I, Futscher BW. Methylation of the BRCA1 promoter is associated with decreased BRCA1 mRNA levels in clinical breast cancer specimens. Carcinogenesis. 2000;21(9):1761–5.
Attwood JT, Yung RL, Richardson BC. DNA methylation and the regulation of gene transcription. Cell Mol Life Sci. 2002;59(2):241–57.
Birgisdottir V, Stefansson OA, Bodvarsdottir SK, Hilmarsdottir H, Jonasson JG, Eyfjord JE. Epigenetic silencing and deletion of the BRCA1 gene in sporadic breast cancer. Breast Cancer Res. 2006;8(4):R38.
Suter CM, Martin DI, Ward RL. Germline epimutation of MLH1 in individuals with multiple cancers. Nat Genet. 2004;36(5):497–501.
Suijkerbuijk KP, Fackler MJ, Sukumar S, et al. Methylation is less abundant in BRCA1-associated compared with sporadic breast cancer. Ann Oncol. 2008;19(11):1870–4.
Rice JC, Massey-Brown KS, Futscher BW. Aberrant methylation of the BRCA1 CpG island promoter is associated with decreased BRCA1 mRNA in sporadic breast cancer cells. Oncogene. 1998;17(14):1807–12.
Honrado E, Osorio A, Milne RL, et al. Immunohistochemical classification of non-BRCA1/2 tumors identifies different groups that demonstrate the heterogeneity of BRCAX families. Mod Pathol. 2007;20(12):1298–306.
Ali AB, Iau PT, Sng JH. Cancer-specific methylation in the BRCA1 promoter in sporadic breast tumours. Med Oncol. 2010. [Epub ahead of print].
Tapia T, Smalley SV, Kohen P, Muñoz A, Solis LM, Corvalan A. Promoter hypermethylation of BRCA1 correlates with absence of expression in hereditary breast cancer tumors. Epigenetics. 2008;3(3):157–63.
Chen Y, Zhou J, Xu Y, Li Z, Wen X, Yao L, Xie Y, Deng D. BRCA1 promoter methylation associated with poor survival in Chinese patients with sporadic breast cancer. Cancer Sci. 2009;100:1663–7.
Matros E, Wang ZC, Lodeiro G, Miron A, Iglehart JD, Richardson AL. BRCA1 promoter methylation in sporadic breast tumors: relationship to gene expression profiles. Breast Cancer Res Treat. 2005;91:179–86.
Zhao Z, Han L. CpG islands: algorithms and applications in methylation studies. Biochem Biophys Res Commun. 2009;382(4):643–5.
Ehrlich M. DNA methylation in cancer: too much, but also too little. Oncogene. 2002;21(35):5400–13.
Baylin SB, Herman JG. DNA hypermethylation in tumorigenesis: epigenetics joins genetics. Trends Genet. 2000;16(4):168–74.
Zhu WG, Srinivasan K, Dai Z, Duan W, et al. Methylation of adjacent CpG sites affects Sp1/Sp3 binding and activity in the p21 (Cip1) promoter. Mol Cell Biol. 2003;23(12):4056–65.
Clark SJ, Harrison J, Molloy PL. Sp1 binding is inhibited by mCpmCpG methylation. Gene. 1997;195(1):67–71.
Watt F, Molloy PL. Cytosine methylation prevents binding to DNA of a HeLa cell transcription factor required for optimal expression of the adenovirus major late promoter. Genes Dev. 1988;2(9):1136–43.
Mancini DN, Rodenhiser DI, Ainsworth PJ, O’Malley FP, Singh SM, Xing W. CpG methylation within the 5′ regulatory region of the BRCA1 gene is tumor specific and includes a putative CREB binding site. Oncogene. 1998;16(9):1161–9.
Catteau A, Joanna R. Morris. BRCA1 methylation: a significant role in tumour development. Semin Cancer Biol. 2002;12(5):359–71.
Hitchins MP, Ward RL. Erasure of MLH1 methylation in spermatozoa- implications for epigenetic inheritance. Nat Genet. 2007;39:1289.
Dupont C, Armant DR, Brenner CA. Epigenetics: definition, mechanisms and clinical perspective. Semin Reprod Med. 2009;27(5):351–7.
Okano M, Xie S, Li E. Cloning and characterization of a family of novel mammalian DNA (cytosine-5) methyltransferases. Nat Genet. 1998;19(3):219–20.
Macis D, Maisonneuve P, Johansson H, Bonanni B, Botteri E, Iodice S. Methylenetetrahydrofolate reductase (MTHFR) and breast cancer risk: a nested-case-control study and a pooled meta-analysis. Breast Cancer Res Treat. 2007;106(2):263–71.
Abreu PA, Dellamora-Ortiz G, Leão-Ferreira LR, et al. DNA mrthylation: a promising target for the twenty-first century. Expert Opin Ther Targets. 2008;12(8):1035–47.
Chen Y, Toland AE, McLennan J, et al. Lack of germ-line promoter methylation in BRCA1-negative families with familial breast cancer. Genet Test. 2006;10(4):281–4.
Yazici H, Terry MB, Cho YH, et al. Aberrant methylation of RASSF1A in plasma DNA before breast cancer diagnosis in the breast cancer family registry. Cancer Epidemiol Biomarkers Prev. 2009;18(10):2723–5.
Acknowledgments
The authors are grateful to the insightful work provided by Dr. Yashuang Zhao and all of the fellows and students who gave me the possibility to complete this thesis. We thank Dr. Shihui Yu and Dr. QingBin Song for critical reading of the manuscript, graphics assistance and word processing assistance.
Conflict of interest
The authors declare that they have no competing financial interests.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Pang, D., Zhao, Y., Xue, W. et al. Methylation profiles of the BRCA1 promoter in hereditary and sporadic breast cancer among Han Chinese. Med Oncol 29, 1561–1568 (2012). https://doi.org/10.1007/s12032-011-0100-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12032-011-0100-0