Abstract
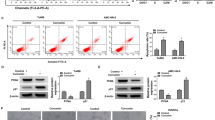
The medicinal properties of curcumin are well documented in Indian and Chinese systems of medicine, which refer to its wide use in the treatment of some diseases. It has shown to have anti-carcinogenic properties and is known to prevent tumor development in some cancers. In our study, we confirmed that the expression of miR-15a and miR-16 was upregulated and that of Bcl-2 was downregulated in curcumin-treated MCF-7 cells. Silencing miR-15a and miR-16 by specific inhibitors restored the expression of Bcl-2. Thus, we concluded that curcumin can reduce the expression of Bcl-2 by upregulating the expression of miR-15a and miR-16 in MCF-7 cells.



Similar content being viewed by others
References
Sarkar FH, Li Y, Wang Z, Kong D, et al. Cellular signaling perturbation by natural products. Cell Signal. 2009;21:1541–7.
Anand P, Sundaram C, Jhurani S, Kunnumakkara AB, Aggarwal BB. Curcumin and cancer: an “old-age” disease with an “age-old” solution. Cancer Lett. 2008;267:133–64.
Cimmino A, Calin GA, Fabbri M, Iorio MV. miR-15 and miR-16 induce apoptosis by targeting BCL-2. Proc Natl Acad Sci U S A. 2005;102:13944–9.
Woo JH, Kim YH, Choi YJ, Kim DG, Lee KS, Bae JH, et al. Molecular mechanisms of curcumininduced cytotoxicity: induction of apoptosis through generation of reactive oxygen species, down-regulation of Bcl- XL and IAP, the release of cytochrome c and inhibition of Akt. Carcinogenesis. 2003;24:1199–208.
Bhattacharyya S, Mandal D, Saha B, Sen GS, Das T, Sa G. Curcumin prevents tumor-induced T cell apoptosis through Stat-5a-mediated Bcl-2 induction. J Biol Chem. 2007;282:15954–64.
Garzon R, Calin GA, Croce CM. MicroRNAs in cancer. Annu Rev Med. 2009;60:167–79.
Wang V, Wu W. MicroRNA-based therapeutics for cancer. BioDrugs. 2009;23:15–23.
Davis CD, Ross SA. Evidence for dietary regulation of microRNA expression in cancer cells. Nutr Rev. 2008;66:477–82.
Sun M, Estrov Z, Ji Y, Coombes KR, Harris DH, Kurzrock R. Curcumin (diferuloylmethane) alters the expression profiles of microRNAs in human pancreatic cancer cells. Mol Cancer Ther. 2008;7:464–73.
Kluck RM, Bossy-Wetzel E, Green DR, Newmeyer DD. The release of cytochrome c from mitochondria: a primary site for Bcl-2 regulation of apoptosis. Science. 1997;275:1132–6.
Youle RJ, Strasser A. The Bcl-2 protein family: opposing activities that mediate cell death. Nat Rev Mol Cell Biol. 2008;9:47–59.
Frenzel A, Grespi F, Chmelewskij W, Villunger A. Bcl2 family proteins in carcinogenesis and the treatment of cancer. Apoptosis. 2009;14:584–96.
Lu Y, Xiao J, Lin H, Bai Y, Luo X, Wang Z, et al. A single anti-microRNA antisense oligodeoxyribonucleotide (AMO) targeting multiple microRNAs offers an improved approach for microRNA interference. Nucleic Acids Res. 2009;37:e24.
Esau CC. Inhibition of microRNA with antisense oligonucleotides. Methods. 2008;44:55–60.
Tamvakopoulos C, Dimas K, Sofianos ZD, Hatziantoniou S, Han Z, Liu ZL, et al. Metabolism and anticancer activity of the curcumin analogue, dimethoxycurcumin. Clin Cancer Res. 2007;13:1269–77.
Hammamieh R, Sumaida D, Zhang X, Das R, Jett M. Control of the growth of human breast cancer cells in culture by manipulation of arachidonate metabolism. BMC Cancer. 2007;7:138.
Shishodia S, Chaturvedi MM, Aggarwal BB. Role of curcumin in cancer therapy. Curr Probl Cancer. 2007;31:243–305.
Chekhun VF, Lukyanova NY, Kovalchuk O, Tryndyak VP, Pogribny IP. Epigenetic profiling of multidrug-resistant human MCF-7 breast adenocarcinoma cells reveals novel hyper- and hypomethylated targets. Mol Cancer Ther. 2007;6:1089–98.
Wang X, Belguise K, Kersual N, Kirsch KH, Mineva ND, Galtier F, et al. Oestrogen signalling inhibits invasive phenotype by repressing RelB and its target BCL2. Nat Cell Biol. 2007;9:470–8.
Garzon R, Pichiorri F, Palumbo T, Visentini M, Aqeilan R, Cimmino A, et al. MicroRNA gene expression during retinoic acid-induced differentiation of human acute promyelocytic leukemia. Oncogene. 2007;26:4148–57.
Acknowledgments
This work was supported by a grant from the National Natural Science Foundation of China (No.30872400).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Jie Yang, Yunxin Cao, and Jifeng Sun have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
12032_2009_9344_MOESM1_ESM.tif
Supplementary Fig. 1 The expression of Bcl-2 was downregulated in SKBR-3 and Bcap-37 cells treated with curcumin. SKBR-3 and Bcap-37 cells were treated with 60 μmol l−1 curcumin for 24 h. Bcl-2 level was assayed by western blot. Equal amounts of total cellular protein (50 μg) were resolved by 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS–PAGE), and β-actin was used as an internal control (TIFF 80 kb)
12032_2009_9344_MOESM2_ESM.tif
Supplementary Fig. 2 Both miR-15a and miR-16 were downregulated in SKBR-3 and Bcap-37 cells treated with curcumin. SKBR-3 and Bcap-37 cells were treated with 60 μmol l−1 curcumin for 24 h. The expression of miR-15a (a) and miR-16 (b) was tested by quantitative polymerase chain reaction (PCR). Relative expression levels of each mature miRNA were normalized to U6 expression levels (TIFF 1436 kb)
Rights and permissions
About this article
Cite this article
Yang, J., Cao, Y., Sun, J. et al. Curcumin reduces the expression of Bcl-2 by upregulating miR-15a and miR-16 in MCF-7 cells. Med Oncol 27, 1114–1118 (2010). https://doi.org/10.1007/s12032-009-9344-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12032-009-9344-3