Abstract
Background
Capecitabine and oxaliplatin are both synergistically active against metastatic colorectal cancer (MCRC). We evaluated our experience at two centers with capecitabine and oxaliplatin combination (XELOX) in previously untreated patients with MCRC.
Patients and methods
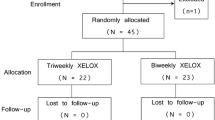
We reviewed medical records of 85 previously untreated patients with MCRC who received first-line XELOX regimen. Oxaliplatin was given at a dose of 130 mg/m2 on day 1 in combination with capecitabine 1500 mg/m2/day on days 1–14 every 3 weeks.
Results
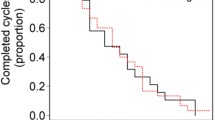
Seventy six of 85 patients were evaluated for response and toxicity. Patients with a follow up of less than 6 months were excluded from the study. Objective response rate was 46% including 8 complete responses (10.5%) and 27 partial responses (35.5%). Additionally, 20 patients (26.3%) had disease stabilization at least 3 months after the treatment. The patients were followed for a median 12.5 months (range 2–32). Median time to disease progression (TTP) was 11 months (range 2–27 months). Median overall survival (OS) time has not yet been reached. One-year survival rate was 66%. Toxicity was modest with infrequent grade 3–4 adverse effects.
Conclusion
XELOX is an active regimen against MCRC in the first-line setting with favorable toxicity profile. Our results appear to be comparable, if not superior, to the results of other reports of first-line XELOX therapy in respect to objective response rates, survival data, and safety profile. Convenience with oral administration of every 3-week schedule makes XELOX regimen a compelling therapeutic option in the treatment of first-line MCRC.


Similar content being viewed by others
References
Jemal A, Siegel R, Ward E, Murray T, Xu J, Thun MJ. Cancer statistics, 2007. CA Cancer J Clin 2007;57:43–66.
De Gramont A, Figer A, Seymour M, et al. Leucovorin and Fluorouracil with or without oxaliplatin as first-line treatment in advanced colorectal cancer. J Clin Oncol 2000;18:2938–47.
Douillard JY, Cunningham D, Roth AD, et al. Irinotecan combined with fluorouracil compared with fluorouracil alone as first line treatment for metastatic colorectal cancer: a multicentre randomized trial. Lancet 2000;355:1041–7.
Hoff PM, Ansari R, Batist G, et al. Comparison of oral capecitabine versus intravenous fluorouracil plus leucovorin as first-line treatment in 605 patients with metastatic colorectal cancer: Results of a randomized phase III study. J Clin Oncol 2001;19:2282–92.
Chang DZ, Abbruzzese JL. Capecitabine plus oxaliplatin vs infusional 5-fluorouracil plus oxaliplatin in the treatment of colorectal cancer. Pro: The CapeOx regimen is preferred over FOLFOX. Clin Adv Hematol Oncol 2005;3:400–4.
Therasse P, Arbuck SG, Eisenhauer EA, et al. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst 2000;3:205–16.
National Cancer Institute Common Toxicity Criteria (NCI-CTC) v2.0. http://www.ctep.info.nih.gov/reporting/ctc.html.
Kaplan EL, Meier P. Nonparametric estimation for incomplete observations. J Am Stat Assoc 1958;53:457–81.
Cassidy J, Tabernero J, Twelves C, et al. XELOX (capecitabine plus oxaliplatin): active first-line therapy for metastatic with colorectal cancer. J Clin Oncol 2004;22:2084–91.
Shields AF, Zalupski MM, Marshall JL, Meropol NJ. Treatment of advanced colorectal carcinoma with oxaliplatin and capecitabine: a phase II trial. Cancer 2004;100:531–7.
Hochster HS, Hart LL, Ramanathan RK, Hainsworth JD, Hedrick EE, Childs BH. Safety and efficacy of oxaliplatin/fluoropyrimidine regimens with or without bevacizumab as first-line treatment of metastatic colorectal cancer (mCRC): final analysis of the TREE-Study. J Clin Oncol 2006 ASCO Annual Meeting Proceedings Part I. Vol 24, No. 18S (June 20 Supplement), 2006:3510.
Martoni AA, Pinto C, Di Fabio F, et al. Capecitabine plus oxaliplatin (XELOX) versus protracted fluorouracil venous infusion plus oxaliplatin (pvifox) as first-line treatment in advanced colorectal cancer: a GOAM phase II randomized study (FOCA trial). Eur J Cancer 2006;42:3161–8.
Zeuli M, Nardoni C, Pino MS, et al. Phase II study of capecitabine and oxaliplatin as first line treatment in advanced colorectal cancer. Ann Oncol 2003,14:1378–82.
Makatsoris T, Kalofonos HP, Aravantinos G, et al. A phase II study of capecitabine plus oxaliplatin (XELOX): a new first line option in metastatic colorectal cancer. Int J Gastrointest Cancer 2005;35:103–9.
Cassidy J, Clarke S, Diaz-Rubio E, et al. XELOX vs. FOLFOX4: efficacy results from XELOX-1/NO16966, a randomized phase III trial in first-line metastatic colorectal cancer (MCRC). ASCO Gastrointestinal Cancers Symposium, January 19–21, 2007, Orlando FL: 270.
Feliu J, Salud A, Escudero P, et al. XELOX (capecitabine plus oxaliplatin) as first line treatment for elderly patients over 70 years of age with advanced colorectal cancer. Br J Cancer 2006;94:969–75.
Comella P, Natale D, Farris A, et al. Capecitabine plus oxaliplatin for the first line treatment of elderly patients with metastatic colorectal carcinoma: final results of the Southern Italy Cooperative Oncology Group Trial 0108. Cancer 2005;104:282–9.
Twelves CJ, Butts Ca, Cassidy J, et al. Capecitabine/oxaliplatin, a safe and active first line regimen for older patients with metastatic colorectal cancer: post hoc analysis of a large phase II study. Clin Colorectal Cancer 2005;5:101–7.
Author information
Authors and Affiliations
Consortia
Corresponding author
Rights and permissions
About this article
Cite this article
Yaman, E., Uner, A., Er, O. et al. Capecitabine plus oxaliplatin (xelox) in the treatment of chemotherapy-naive patients with metastatic colorectal cancer. Med Oncol 24, 431–435 (2007). https://doi.org/10.1007/s12032-007-0035-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12032-007-0035-7