Abstract
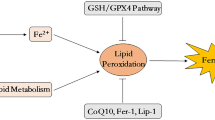
Propofol is a short-acting intravenous anesthetic that is widely used in clinical treatment. Previous articles have indicated that propofol is a therapeutic target for anti-apoptosis, anti-inflammation, anti-lipid peroxidation, and anti-reactive oxygen species (ROS). Moreover, cell ferroptosis is strongly correlated with cellular ROS, inflammatory responses, and lipid peroxidation. However, the mechanisms by which propofol attenuates neuronal injury by reducing ferroptosis remain unknown. Hence, we hypothesized that propofol could protect neurons by reducing ferroptosis. To test this hypothesis, HT-22 cells were treated with a specific ferroptosis activator (erastin) in the presence of propofol (50 μM). We found that propofol reduced erastin-induced high Fe2+ concentrations, lipid peroxides, and excess ROS. Western blotting results also suggested that propofol could rescue erastin-induced low expression of GXP4 and system Xc−. Further experiments indicated that propofol attenuated p-ALOX5 expression at Ser663 independent of ERK. In addition, we built two transient transfection cell lines, ALOX5 OE and Ser663Ala-ALOX5 OE, to confirm the target of propofol. We found that the Ser663 point is the critical role of propofol in rescuing erastin-induced cell injury/lipid peroxidation. In conclusion, propofol may help attenuate ferroptosis, which may provide a new therapeutic method to treat neuronal injury or the brain inflammatory response.




Similar content being viewed by others
Availability of Data and Materials
All data generated or analyzed during this study are included in this published article. Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
Abbreviations
- 4-HNE:
-
4-Hydroxy-2E-nonenal
- CCK:
-
Cell counting kit
- COX:
-
Cyclooxygenases
- DHE:
-
Dihydroethidium
- Era:
-
Erastin
- EM:
-
Electronic microscopy
- ERK:
-
Extracellular regulated protein kinases
- Fer-1:
-
Ferrostain-1
- GPX4:
-
Glutathione peroxidase 4
- IF:
-
Immunofluorescences
- MK2:
-
MAPK-activated protein kinase 2;
- PPF:
-
Propofol
- PKA:
-
Protein kinase A
- qRT-PCR:
-
Quantitative real-time PCR
- PI:
-
Propidium iodide
- PTGS2:
-
Prostaglandin-endoperoxide synthase 2
- RSL:
-
RAS selective lethal
- ROS:
-
Reactive oxygen species
- mtROS:
-
Mitochondrial ROS
- xCT:
-
System Xc−
References
Angelova P, Abramov A (2018) Role of mitochondrial ROS in the brain: from physiology to neurodegeneration. FEBS Lett 592(5):692–702. https://doi.org/10.1002/1873-3468.12964
Brohan J, Goudra BG (2017) The Role of GABA Receptor agonists in anesthesia and sedation. CNS Drugs 31(10):845–856. https://doi.org/10.1007/s40263-017-0463-7
Cheloni G, Vi S (2013) Optimization of the C11-BODIPY(581/591) dye for the determination of lipid oxidation in Chlamydomonas reinhardtii by flow cytometry. Cytometry A 83(10):952–961. https://doi.org/10.1002/cyto.a.22338 (Epub 2013 Aug 13)
Chen C, Zhou X, He J, Xie Z, Xia S, Lu G (2019) The roles of GABA in ischemia-reperfusion injury in the central nervous system and peripheral organs. Oxid Med Cell Longev. https://doi.org/10.1155/2019/4028394
Chen D, Fan Z, Rauh M, Buchfelder M, Eyupoglu IY, Savaskan N (2017) ATF4 promotes angiogenesis and neuronal cell death and confers ferroptosis in a xCT-dependent manner. Oncogene 36(40):5593–5608. https://doi.org/10.1038/onc.2017.146
Chu J, Liu X, Song R, Li Q (2020) Ferrostatin-1 protects HT-22 cells from oxidative toxicity. Neural Regen Res 15(3):528–536. https://doi.org/10.4103/1673-5374.266060
Clemente S, Martinez O, Monsalve M, Samhan A (2020) Targeting lipid peroxidation for cancer treatment. Molecules 25(21):5144. https://doi.org/10.3390/molecules25215144
Dinis R (2018) Metabolic profiles of propofol and fospropofol: clinical and forensic interpretative aspects. Biomed Res Int 2018:6852857. https://doi.org/10.1155/2018/6852857
Dixon S, Lemberg K, Lamprecht M, Skouta R, Zaitsev E, Gleason C et al (2012) Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell 149(5):1060–1072. https://doi.org/10.1016/j.cell.2012.03.042
Erkkinen M, Kim M, Geschwind M (2018) Clinical neurology and epidemiology of the major neurodegenerative diseases. Cold Spring Harb Perspect Biol 10(4):a033118. https://doi.org/10.1101/cshperspect.a033118
Fan W, Zhu X, Wu L, Wu Z, Li D, Huang F et al (2015) Propofol: an anesthetic possessing neuroprotective effects. Eur Rev Med Pharmacol Sci 19(8):1520–1529
Fang D, Maldonado E (2018) VDAC regulation: a mitochondrial target to stop cell proliferation. Adv Cancer Res 138:41–69. https://doi.org/10.1016/bs.acr.2018.02.002
Gan B (2021) Mitochondrial regulation of ferroptosis. J Cell Biol 220(9):e202105043. https://doi.org/10.1083/jcb.202105043
Hu F, Jiang J, Yu G, Zang H, Sun H (2020) Propofol pretreatment prevents oxygen-glucose deprivation/reoxygenation (OGD/R)-induced inflammation through nuclear transcription factor kappaB (NF-kappaB) pathway in neuroblastoma cells. Curr Neurovasc Res 17(1):27–34. https://doi.org/10.2174/1567202617666191227110158
Imai H, Matsuoka M, Kumagai T, Sakamoto T, Koumura T (2017) Lipid peroxidation-dependent cell death regulated by GPx4 and ferroptosis. Curr Top Microbiol Immunol 403:143–170. https://doi.org/10.1007/82_2016_508
Jiang L, Kon N, Li T, Wang S, Su T, Hibshoosh H et al (2015) Ferroptosis as a p53-mediated activity during tumour suppression. Nature 520(7545):57–62. https://doi.org/10.1038/nature14344
Karuppagounder S, Alin L, Chen Y, Brand D, Bourassa M, Dietrich K (2018) N-acetylcysteine targets 5 lipoxygenase-derived, toxic lipids and can synergize with prostaglandin E2 to inhibit ferroptosis and improve outcomes following hemorrhagic stroke in mice. Ann Neurol 84(6):854–872. https://doi.org/10.1002/ana.25356
Korczak M, Kurowski P, Lesniak A, Gronbladh A, Filipowska A, Bujalska M (2020) GABAB receptor intracellular signaling: novel pathways for depressive disorder treatment? Eur J Pharmacol 885:173531. https://doi.org/10.1016/j.ejphar.2020.173531
Li Q, Han X, Lan X, Gao Y, Wan J, Durham F et al (2017) Inhibition of neuronal ferroptosis protects hemorrhagic brain. JCI Insight 2(7):e90777. https://doi.org/10.1172/jci.insight.90777
Li S, Lei Z, Zhao M, Hou Y, Wang D, Xu X et al (2021) Propofol inhibits ischemia/reperfusion-induced cardiotoxicity through the protein kinase C/nuclear factor erythroid 2-related factor pathway. Front Pharmacol 12:655726. https://doi.org/10.3389/fphar.2021.655726
Liu J, Li Y, Xia X, Yang X, Zhao R, Peer J et al (2019) Propofol reduces microglia activation and neurotoxicity through inhibition of extracellular vesicle release. J Neuroimmunol 333:476962. https://doi.org/10.1016/j.jneuroim.2019.05.003
Liu Y, Wang W, Li Y, Xiao Y, Cheng J, Jia J (2015) The 5-lipoxygenase inhibitor zileuton confers neuroprotection against glutamate oxidative damage by inhibiting ferroptosis. Biol Pharm Bull 38(8):1234–1239. https://doi.org/10.1248/bpb.b15-00048
Lu X, Liu Y, Xuan W, Ye J, Yao H, Huang C et al (2019) Circ_1639 induces cells inflammation responses by sponging miR-122 and regulating TNFRSF13C expression in alcoholic liver disease. Toxicol Lett 314:89–97. https://doi.org/10.1016/j.toxlet.2019.07.021
Ma Z, Li K, Chen P, Pan J, Li X, Zhao G (2020) Propofol attenuates inflammatory damage via inhibiting NLRP1-Casp1-Casp6 signaling in ischemic brain injury. Biol Pharm Bull 43(10):1481–1489. https://doi.org/10.1248/bpb.b20-00050
Ngo D, Vo T (2019) An updated review on pharmaceutical properties of gamma-aminobutyric acid. Molecules 24(15):2678. https://doi.org/10.3390/molecules24152678
Okuno T, Koutsogiannaki S, Ohba M, Chamberlain M, Bu W, Lin F et al (2017) Intravenous anesthetic propofol binds to 5-lipoxygenase and attenuates leukotriene B4 production. FASEB J 31(4):1584–1594. https://doi.org/10.1096/fj.201601095R
Pratt DA, Tallman KA, Porter NA (2011) Free radical oxidation of polyunsaturated lipids: new mechanistic insights and the development of peroxyl radical clocks. Acc Chem Res 44(6):458–467. https://doi.org/10.1021/ar200024c (Epub 2011 Apr 12)
Radmark O, Werz O, Steinhilber D, Samuelsson B (2007) 5-Lipoxygenase: regulation of expression and enzyme activity. Trends Biochem Sci 32(7):332–341. https://doi.org/10.1016/j.tibs.2007.06.002
Reichert C, Freitas F, Sampaio J, Rokita L, Barros P, Levy D et al (2020) Ferroptosis mechanisms involved in neurodegenerative diseases. Int J Mol Sci 21(22):8765. https://doi.org/10.3390/ijms21228765
Rossaint J, Rossaint R, Weis J, Fries M, Rex S, Coburn M (2009) Propofol: neuroprotection in an in vitro model of traumatic brain injury. Crit Care 13(2):R61. https://doi.org/10.1186/cc7795
Shintoku R, Takigawa Y, Yamada K, Kubota C, Yoshimoto Y, Takeuchi T et al (2017) Lipoxygenase-mediated generation of lipid peroxides enhances ferroptosis induced by erastin and RSL3. Cancer Sci 108(11):2187–2194. https://doi.org/10.1111/cas.13380
Song Y, Bei Y, Xiao Y, Tong H, Wu X, Chen M (2018) Edaravone, a free radical scavenger, protects neuronal cells’ mitochondria from ischemia by inactivating another new critical factor of the 5-lipoxygenase pathway affecting the arachidonic acid metabolism. Brain Res 1690:96–104. https://doi.org/10.1016/j.brainres.2018.03.006
Sun Q, Zhou H, Mao X (2019) Emerging roles of 5-lipoxygenase phosphorylation in inflammation and cell death. Oxid Med Cell Longev 2019:2749173. https://doi.org/10.1155/2019/2749173
Wei X, Wan X, Zhao B, Hou J, Liu M, Cheng B (2012) Propofol inhibits inflammation and lipid peroxidation following cerebral ischemia/reperfusion in rabbits. Neural Regen Res 7(11):837–841. https://doi.org/10.3969/j.issn.1673-5374.2012.11.007
Werz O, Szellas D, Steinhilber D, Radmark O (2002) Arachidonic acid promotes phosphorylation of 5-lipoxygenase at Ser-271 by MAPK-activated protein kinase 2 (MK2). J Biol Chem 277(17):14793–14800. https://doi.org/10.1074/jbc.M111945200
Wu C, Du M, Yu R, Cheng Y, Wu B, Fu J et al (2022) A novel mechanism linking ferroptosis and endoplasmic reticulum stress via the circPtpn14/miR-351-5p/5-LOX signaling in melatonin-mediated treatment of traumatic brain injury. Free Radic Biol Med 178:271–294. https://doi.org/10.1016/j.freeradbiomed.2021.12.007
Xie CL, Hu LQ, Pan YB, Qian YN (2015) Propofol attenuation of hydrogen peroxide-induced injury in human umbilical vein endothelial cells involves aldose reductase. Pharmazie 70(2):103–109. https://doi.org/10.1691/ph.2015.4734
Xie Y, Hou W, Song X, Yu Y, Huang J, Sun X et al (2016) Ferroptosis: process and function. Cell Death Differ 23(3):369–379. https://doi.org/10.1038/cdd.2015.158
Yagoda N, Rechenberg M, Zaganjor E, Bauer A, Yang W, Fridman D (2007) RAS-RAF-MEK-dependent oxidative cell death involving voltage-dependent anion channels. Nature 447(7146):864–868. https://doi.org/10.1038/nature05859
Yang W, Kim K, Gaschler M, Patel M, Shchepinov M, Stockwell B (2016) Peroxidation of polyunsaturated fatty acids by lipoxygenases drives ferroptosis. Proc Natl Acad Sci USA 113(34):E4966–E4975. https://doi.org/10.1073/pnas.1603244113
Yang W, Stockwell B (2016) Ferroptosis: death by lipid peroxidation. Trends Cell Biol 26(3):165–176. https://doi.org/10.1016/j.tcb.2015.10.014
Zou Y, Li H, Graham E, Deik A, Eaton J, Wang W et al (2020) Cytochrome P450 oxidoreductase contributes to phospholipid peroxidation in ferroptosis. Nat Chem Biol 16(3):302–309. https://doi.org/10.1038/s41589-020-0472-6
Acknowledgements
Over the course of my researching and writing this paper, I would like to express my thanks to all those who have helped me.
Funding
YL received Anhui Province Research and Development Program (No. 17041f0804021).
Author information
Authors and Affiliations
Contributions
WX and ZY designed and performed the experiments. XL analyzed the data. XL participated in manuscript writing/editing. LY and WJ contributed significantly to the experimental design and funds.
Corresponding authors
Ethics declarations
Ethics Approval
This study does not include human or animal samples. Nanjing University Medical School Research Ethics Committee has confirmed that no ethical approval is required.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Wenting Xuan, Xinyi Lu, and Zeyong Yang contributed equally to this study.
Rights and permissions
About this article
Cite this article
Xuan, W., Lu, X., Yang, Z. et al. Propofol Protects Against Erastin-Induced Ferroptosis in HT-22 Cells. J Mol Neurosci 72, 1797–1808 (2022). https://doi.org/10.1007/s12031-022-02017-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-022-02017-7