Abstract
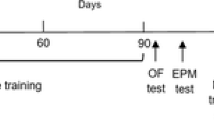
A decline of estrogen level leads to spatial learning and memory impairments, which mediated by hippocampus and cortex. Accumulating evidences demonstrated that aerobic exercise improved memory of postmenopausal women and ovariectomized (OVX) mice. However, the molecular mechanisms for this protection of exercise are not completely clear. Accordingly, the present study was designed to examine the effect of aerobic exercise on the dendritic morphology in the hippocampus and cerebral cortex, as well as the BNDF-mTOR signaling pathway of OVX mice. Adult female C57BL/6 mice were divided into four groups (n = 10/group): sham-operated (SHAM/CON), sham-operated with 8-week treadmill exercise (SHAM/EX), ovariectomized operated (OVX/CON), and ovariectomized operated with exercise (OVX/EX). Aerobic exercise improved the impairment of dendritic morphology significantly induced by OVX that was tested by Golgi staining, and it also upregulated the synaptic plasticity-related protein expression of PSD95 and GluR1 as well as activated BDNF-mTOR signaling pathway in the hippocampus and cerebral cortex. In conclusion, aerobic exercise reversed the change of dendritic morphology and increased the synaptic plasticity-related protein expression in the hippocampus and cerebral cortex of OVX mice. The positive effects induced by exercise might be mediated through the BDNF-mTOR signaling pathway.












Similar content being viewed by others
Data Availability and Material
All relevant data are within the paper.
References
Aimone JB, Deng W, Gage FH (2010) Adult neurogenesis: integrating theories and separating functions. Trends Cogn Sci 14:325–337
Allison DW, Deborah AF, Mindy SK et al (2018) Quality of life among postmenopausal women enrolled in the Minnesota Green Tea Trial. Maturitas 108:1–6
Baker FC, Sattari N, Zambotti Md et al (2019) Impact of sex steroids and reproductive stage on sleep-dependent memory consolidation in women. Neurobiol Learn Mem 160:118–131
Barker GR, Bird F, Alexander V et al (2007) Recognition memory for objects, place, and temporal order: a disconnection analysis of the role of the medial prefrontal cortex and perirhinal cortex. J Neurosci 27:2948–2957
Basso JC, Shang A, Elman M et al (2015) Acute exercise improves prefrontal cortex but not hippocampal function in healthy adults. J Int Neuropsychol Soc 21:791–801
Beique JC, Andrade R (2003) PSD-95 regulates synaptic transmission and plasticity in rat cerebral cortex. J Physiol 546:859–867
Ben J, Soares FM, Eb S et al (2010) Running exercise effects on spatial and avoidance tasks in ovariectomized rats. Neurobiol Learn Mem 94:312–317
Bockaert J, Marin P (2015) mTOR in brain physiology and pathologies. Physiol Rev 95:1157–1187
Brockett AT, LaMarca EA, Gould E (2015) Physical exercise enhances cognitive flexibility as well as astrocytic and synaptic markers in the medial prefrontal cortex. PLoS ONE 10:e0124859
Chen A, Xiong LJ, Tong Y et al (2013) Neuroprotective effect of brain-derived neurotrophic factor mediated by autophagy through the PI3K/Akt/mTOR pathway. Mol Med Rep 8:1011–1016
Clark RE, Zola SM, Squire LR (2000) Impaired recognition memory in rats after damage to the hippocampus. J Neurosci 20:8853–8860
Cui J, Jothishankar B, He P et al (2014) Amyloid precursor protein mutation disrupts reproductive experience-enhanced normal cognitive development in a mouse model of Alzheimer’s disease. Mol Neurobiol 49:103–112
Drew M, Hen R (2007) Adult hippocampal neurogenesis as target for the treatment of depression. Cns Neurol Disord Drug Targets 6:205–218
Ehrlich I, Klein M, Rumpel S et al (2007) PSD-95 is required for activity-driven synapse stabilization. Proc Natl Acad Sci USA 104:4176–4181
Eriksson J, Vogel EK, Lansner A et al (2015) Neurocognitive architecture of working memory. Neuron 88:33–46
Fernandes M, Santos G, Santos TM et al (2020) Effects of physical exercise on neuroplasticity and brain function: a systematic review in human and animal studies. Neural Plast 2020:8856621
Guo W, Nagappan G, Lu B (2018) Differential effects of transient and sustained activation of BDNF-TrkB signaling. Dev Neurobiol 78:647–659
Horwood JM, Dufour F, Laroche S et al (2006) Signalling mechanisms mediated by the phosphoinositide 3-kinase/Akt cascade in synaptic plasticity and memory in the rat. Eur J Neurosci 23:3375–3384
Howland JG, Wang YT (2008) Synaptic plasticity in learning and memory: stress effects in the hippocampus. Prog Brain Res 169:145–158
Huang W, Zhu PJ, Zhang S et al (2013) mTORC2 controls actin polymerization required for consolidation of long-term memory. Nat Neurosci 16:441–448
Isken F, Pfeiffer AF, Nogueiras R et al (2008) Deficiency of glucose-dependent insulinotropic polypeptide receptor prevents ovariectomy-induced obesity in mice. Am J Physiol Endocrinol Metab 295:E350-355
Jonasson LS, Nyberg L, Kramer A et al (2016) Aerobic exercise intervention, cognitive performance, and brain structure: results from the physical influences on brain in aging (PHIBRA) study. Front Aging Neurosci 8:336
Kang EB, Cho JY (2015) Effect of treadmill exercise on PI3K/AKT/mTOR, autophagy, and Tau hyperphosphorylation in the cerebral cortex of NSE/htau23 transgenic mice. J Exerc Nutrition Biochem 19:199–209
Kretz O, Fester L, Wehrenberg U et al (2004) Hippocampal synapses depend on hippocampal estrogen synthesis. J Neurosci 24:5913–5921
Leal G, Comprido D, Duarte CB (2014) BDNF-induced local protein synthesis and synaptic plasticity. Neuropharmacology 76:639–656
Lee CC, Huang CC, My Wu et al (2005) Insulin stimulates postsynaptic density-95 protein translation via the phosphoinositide 3-kinase-Akt-mammalian target of rapamycin signaling pathway. J Biol Chem 280:18543–18550
Lin TW, Tsai SF, Kuo YM (2018) Physical exercise enhances neuroplasticity and delays Alzheimer’s disease. Brain Plast 4:95–110
Lu Y, Sareddy GR, Wang J et al (2019) Neuron-derived estrogen regulates synaptic plasticity and memory. J Neurosci 39:2792–2809
Mackay CP, Kuys SS, Brauer SG (2017) The effect of aerobic exercise on brain-derived neurotrophic factor in people with neurological disorders: a systematic review and meta-analysis. Neural Plast 2017:4716197
Moriya M, Aoki C, Sakatani K (2016) Effects of physical exercise on working memory and prefrontal cortex function in post-stroke patients. Adv Exp Med Biol 923:203–208
Neill EC, Sammel MD, Freeman EW (2013) Menopause effects on verbal memory: findings from a longitudinal community cohort. J Clin Endocrinol Metab 98:3829–3838
Ocampo AC, Squire LR, Clark RE (2017) Hippocampal area CA1 and remote memory in rats. Learn Mem 24:563–568
Perrachione TK, Ghosh SS, Ostrovskaya I et al (2017) Phonological working memory for words and nonwords in cerebral cortex. J Speech Lang Hear Res 60:1959–1979
Pike CJ, Carroll JC, Rosario ER et al (2009) Protective actions of sex steroid hormones in Alzheimer’s disease. Front Neuroendocrinol 30:239–258
Redila VA, Christie BR (2006) Exercise-induced changes in dendritic structure and complexity in the adult hippocampal dentate gyrus. Neuroscience 137:1299–1307
Saadati H, Esmaeili-Mahani S, Esmaeilpour K et al (2015) Exercise improves learning and memory impairments in sleep deprived female rats. Physiol Behav 138:285–291
Shih PC, Yang YR, Wang RY (2013) Effects of exercise intensity on spatial memory performance and hippocampal synaptic plasticity in transient brain ischemic rats. PLoS ONE 8:e78163
Slipczuk L, Bekinschtein P, Katche C et al (2009) BDNF activates mTOR to regulate GluR1 expression required for memory formation. PLoS ONE 4:e6007
Stern Y, MacKay-Brandt A, Lee S et al (2019) Effect of aerobic exercise on cognition in younger adults: a randomized clinical trial. Neurology 92:e905–e916
Stranahan AM, Lee K, Martin B et al (2009) Voluntary exercise and caloric restriction enhance hippocampal dendritic spine density and BDNF levels in diabetic mice. Hippocampus 19:951–961
Suzuki E, Kamiya H (2016) PSD-95 regulates synaptic kainate receptors at mouse hippocampal mossy fiber-CA3 synapses. Neurosci Res 107:14–19
Szuhany KL, Bugatti M, Otto MW (2015) A meta-analytic review of the effects of exercise on brain-derived neurotrophic factor. J Psychiatr Res 60:56–64
Tao X, Yan M, Wang L et al (2020) Effects of estrogen deprivation on memory and expression of related proteins in ovariectomized mice. Ann Transl Med 8:356
Trinh MA, Klann E (2013) Translational control by eIF2α kinases in long-lasting synaptic plasticity and long-term memory. Neurobiol Learn Mem 105:93–99
Tuscher JJ, Luine V, Frankfurt M et al (2016) Estradiol-mediated spine changes in the dorsal hippocampus and medial prefrontal cortex of ovariectomized female mice depend on ERK and mTOR activation in the dorsal hippocampus. J Neurosci 36:1483–1489
Venezia AC, Quinlan E, Roth SM (2017) A single bout of exercise increases hippocampal Bdnf: influence of chronic exercise and noradrenaline. Genes Brain Behav 16:800–811
Wallace M, Luine V, Arellanos A et al (2006) Ovariectomized rats show decreased recognition memory and spine density in the hippocampus and prefrontal cortex. Brain Res 1126:176–182
Wang Y, Barbaro MF, Baraban SC (2006) A role for the mTOR pathway in surface expression of AMPA receptors. Neurosci Lett 401:35–39
Warden SJ, Galley MR, Al H et al (2014) Cortical and trabecular bone benefits of mechanical loading are maintained long term in mice independent of ovariectomy. J Bone Miner Res 29:1131–1140
Witte MM, Resuehr D, Chandler A et al (2010) Female mice and rats exhibit species-specific metabolic and behavioral responses to ovariectomy. Gen Comp Endocrinol 166:520–528
Wong C, Pearson KG, Lomber SG (2018) Contributions of parietal cortex to the working memory of an obstacle acquired visually or tactilely in the locomoting cat. Cereb Cortex 28:3143–3158
Wu YW, Du X, Van DBM et al (2015) Analyzing the influence of BDNF heterozygosity on spatial memory response to 17beta-estradiol. Transl Psychiatry 5:e498
Yamazaki Y, Sato D, Yamashiro K et al (2018) Inter-individual differences in working memory improvement after acute mild and moderate aerobic exercise. PLoS ONE 13:e0210053
Ye Z, Cudmore RH, Linden DJ (2019) Estrogen-dependent functional spine dynamics in neocortical pyramidal neurons of the mouse. J Neurosci 39:4874–4888
Yoshii A, Constantine-Paton M (2007) BDNF induces transport of PSD-95 to dendrites through PI3K-AKT signaling after NMDA receptor activation. Nature Neuroence 10:702
Zhang YY, Liu MY, Liu Z et al (2019) GPR30-mediated estrogenic regulation of actin polymerization and spatial memory involves SRC-1 and PI3K-mTORC2 in the hippocampus of female mice. CNS Neurosci Ther 25:714–733
Acknowledgements
The authors would like to give our sincere appreciation to the reviewers for their helpful comments on this article.
Funding
This work was supported by the National Natural Science Foundation of China (No. 31971098) and the Shanghai Key Lab of Human Performance (Shanghai University of Sport) (No. 11DZ2261100).
Author information
Authors and Affiliations
Contributions
Yu Feng and Shujie Lou designed the experiments and wrote the manuscript. Yu Feng and Miao Zhang performed the experiments, analyzed the data, and interpreted the results. Yu Feng, Tian Xu, and Shujie Lou revised the text.
Corresponding author
Ethics declarations
Ethics Approval
The animal use protocol was approved by the Ethical Committee for Science Research at Shanghai University of Sport (Approval No. 2018013). Animals were treated in accordance with the Guide for the Care and Use of Laboratory Animals.
Consent for Publication
Written informed consent for publication was obtained from all participants.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Feng, Y., Tian, X., Zhang, M. et al. Treadmill Exercise Reverses the Change of Dendritic Morphology and Activates BNDF-mTOR Signaling Pathway in the Hippocampus and Cerebral Cortex of Ovariectomized Mice. J Mol Neurosci 71, 1849–1862 (2021). https://doi.org/10.1007/s12031-021-01848-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-021-01848-0