Abstract
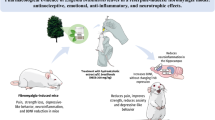
Musculoskeletal pain is a widespread complex regional pain syndrome associated with altered emotional and cognitive functioning along with heightened physical disability that has become a global health concern. Effective management of this disorder and associated disabilities includes accurate diagnosis of its biomarkers and instituting mechanism–based therapeutic interventions. Herein, we explored the role of heraclin, a plant-derived molecule, in musculoskeletal pain and its underlying mechanistic approaches in an experimental mouse model. Reserpine (0.5 mg/kg) for 3 consecutive days evoked hyperalgesia, motor incoordination, lack of exploratory behavior, anxiety, and cognition lapse in mice. Reserpine-challenged mice displayed higher serum cytokine level, altered brain neurotransmitter content, elevated brain and muscle oxidative stress, and upregulated brain nerve growth factor receptor expression. Treatment with heraclin (10 mg/kg for 5 consecutive days) exerted analgesic effect and improved motor coordination and memory deficits in mice. Heraclin arrested serum cytokine rise, normalized brain neurotransmitter content, reduced tissue oxidative stress, and downregulated the nerve growth factor receptor expression. Therefore, it may be suggested that heraclin exerts beneficial effects against reserpine-induced musculoskeletal pain disorder possibly through the attenuation of NGFR-mediated pain and inflammatory signaling.

Graphical Abstract





Similar content being viewed by others
Availability of Data and Material
Data transparency-Yes.
References
Ablin JN, Cohen H, Buskila D (2006) Mechanisms of disease: genetics of fibromyalgia. Nat Clin Pract Rheumatol 2:671–678. https://doi.org/10.1038/ncprheum0349
Aidoo DB, Obiri DD, Osafo N, Antwi AO, Essel LB, Duduyemi BM, Ekor M (2019) Allergic airway-induced hypersensitivity is attenuated by bergapten in murine models of inflammation. AdvPharmacol Sci 2019:6097349. https://doi.org/10.1155/2019/6097349
Ali SA, Zaitone SA, Dessouki AA, Ali AA (2019) Pregabalin affords retinal neuroprotection in diabetic rats: suppression of retinal glutamate, microglia cell expression and apoptotic cell death. Exp Eye Res 184:78–90. https://doi.org/10.1016/j.exer.2019.04.014
Arora V, Kuhad A, Tiwari V, Chopra K (2011) Curcumin ameliorates reserpine-induced pain–depression dyad: Behavioural, biochemical, neurochemical and molecular evidences. Psychoneuroendocrinology 36:1570–1581. https://doi.org/10.1016/j.psyneuen.2011.04.012
Bingham B, Ajit SK, Blake DR, Samad TA (2009) The molecular basis of pain and its clinical implications in rheumatology. Nat Clin Pract Rheumatol 5:28–37. https://doi.org/10.1038/ncprheum0972
Brusco I, Justino AB, Silva CR, Fischer S, Cunha TM, Scussel R, Machado-de-Ávila RA, Ferreira J, Oliveira SM (2019) Kinins and their B1 and B2 receptors are involved in fibromyalgia-like pain symptoms in mice. Biochem Pharmacol 168:119–132. https://doi.org/10.1016/j.bcp.2019.06.023
Cairns BE, Dong X (2008) The role of peripheral glutamate and glutamate receptors in muscle pain. Journal of Musculoskeletal Pain 16:85–91. https://doi.org/10.1080/10582450801960388
Chang CC (1964) A sensitive method for spectrophotofluorometric assay of catecholamines. Int J Neuropharmacol 3:643–649. https://doi.org/10.1016/0028-3908(64)90089-9
Chartier SR, Mitchell SA, Majuta LA, Mantyh PW (2017) Immunohistochemical localization of nerve growth factor, tropomyosin receptor kinase A, and p75 in the bone and articular cartilage of the mouse femur. Mol Pain 13:1744806917745465. https://doi.org/10.1177/1744806917745465
Chen G, Xu Q, Dai M, Liu X (2019) Bergapten suppresses RANKL-induced osteoclastogenesis and ovariectomy-induced osteoporosis via suppression of NF-κB and JNK signaling pathways. Biochem Biophys Res Commun 509:329–334. https://doi.org/10.1016/j.bbrc.2018.12.112
Clauw DJ, Arnold LM, McCarberg BH (2011) The science of fibromyalgia. Mayo Clin Proc 86:907–991. https://doi.org/10.4065/mcp.2011.0206
Conversano C, Marchi L, Ciacchini R, Carmassi C, Contena B, Bazzichi LM, Gemignani A (2018) Personality traits in fibromyalgia (FM): does FM personality exists? A systematic review. Clin Pract Epidemiol Ment Health 14:223–232. https://doi.org/10.2174/1745017901814010223
Cordero MD, de Miguel M, Carmona-López I, Bonal P, Campa F, Moreno-Fernández AM (2010) Oxidative stress and mitochondrial dysfunction in fibromyalgia. Neuro Endocrinol Lett 31:169–173
de Souza AH, da Costa Lopes AM, Castro CJ Jr, Pereira EMR, Klein CP, da Silva CA Jr, da Silva JF, Ferreira J, Gomez MV (2014) The effects of Phα1β, a spider toxin, calcium channel blocker, in a mouse fibromyalgia model. Toxicon 81:37–42. https://doi.org/10.1016/j.toxicon.2014.01.015
Diatchenko L, Fillingim RB, Smith SB, Maixner W (2013) The phenotypic and genetic signatures of common musculoskeletal pain conditions. Nat Rev Rheumatol 9:340–350. https://doi.org/10.1038/nrrheum.2013.43
Du S, Zhang Y, Yang J, Liu X, Wang Y, Xu B, Jia J (2019) Curcumin alleviates β amyloid-induced neurotoxicity in HT22 cells via upregulating SOD2. J Mol Neurosci 67:540–549. https://doi.org/10.1007/s12031-019-01267-2
Ellman GL (1958) A colorimetric method for determining low concentrations of mercaptans. Arch Biochem Biophys 74:443–450. https://doi.org/10.1016/0003-9861(58)90014-6
Favero G, Trapletti V, Bonomini F, Stacchiotti A, Lavazza A, Rodella L, Rezzani R (2017) Oral supplementation of melatonin protects against fibromyalgia-related skeletal muscle alterations in reserpine-induced myalgia rats. Int J Mol Sci 18:1389. https://doi.org/10.3390/ijms18071389
Ghiggia A, Romeo A, Tesio V, Di Tella M, Colonna F, Geminiani GC, Fusaro E, Castelli L (2017) Alexithymia and depression in patients with fibromyalgia: when the whole is greater than the sum of its parts. Psychiatry Res 255:195–197. https://doi.org/10.1016/j.psychres.2017.05.045
Guy S, Mehta S, Leff L, Teasell R, Loh E (2014) Anticonvulsant medication use for the management of pain following spinal cord injury: systematic review and effectiveness analysis. Spinal Cord 52:89–96. https://doi.org/10.1038/sc.2013.146
Häuser W, Bernardy K, Üçeyler N, Sommer C (2009) Treatment of fibromyalgia syndrome with antidepressants: a meta-analysis. JAMA 301:198–209. https://doi.org/10.1001/jama.2008.944
Hefti F (2020) Pharmacology of nerve growth factor and discovery of tanezumab, an anti-nerve growth factor antibody and pain therapeutic. Pharmacol Res 154:104240. https://doi.org/10.1016/j.phrs.2019.04.024
Hsu WH, Lee CH, Chao YM, Kuo CH, Ku WC, Chen CC, Lin YL (2019) ASIC3-dependent metabolomics profiling of serum and urine in a mouse model of fibromyalgia. Sci Rep 9:12123. https://doi.org/10.1038/s41598-019-48315-w
Jain PK (2012) Joshi H. Coumarin: chemical and pharmacological profile 2:236–240. https://doi.org/10.7324/JAPS.2012.2643
Jensen KB, Petzke F, Carville S, Fransson P, Marcus H, Williams SCR, Choy E, Mainguy Y, Gracely R, Ingvar M, Kosek E (2010) Anxiety and depressive symptoms in fibromyalgia are related to poor perception of health but not to pain sensitivity or cerebral processing of pain. Arthritis Rheum 62:3488–3495. https://doi.org/10.1002/art.27649
Kaur A, Singh L, Singh N, Bhatti MS, Bhatti R (2019) Ameliorative effect of imperatorin in chemically induced fibromyalgia: role of NMDA/NFkB mediated downstream signaling. Biochem Pharmacol 166:56–69. https://doi.org/10.1016/j.bcp.2019.05.012
Kulkarni SK, Singh K, Bishnoi M (2008) Comparative behavioural profile of newer antianxiety drugs on different mazes. Indian J Exp Biol 46:633–638
Littlejohn G (2015) Neurogenic neuroinflammation in fibromyalgia and complex regional pain syndrome. Nat Rev Rheumatol 11:639–648. https://doi.org/10.1038/nrrheum.2015.100
Lorenz ND, Comerford EJ, Iff I (2013) Long-term use of gabapentin for musculoskeletal disease and trauma in three cats. J Feline Med Surg 15:507–512. https://doi.org/10.1177/1098612X12470828
Łuszczki JJ, Andres-Mach M, Gleńsk M, Skalicka-Woźniak K (2010) Anticonvulsant effects of four linear furanocoumarins, bergapten, imperatorin, oxypeucedanin, and xanthotoxin, in the mouse maximal electroshock-induced seizure model: a comparative study. Pharmacol Rep 62:1231–1236. https://doi.org/10.1016/s1734-1140(10)70387-x
Matta C, Juhász T, Fodor J, Hajdú T, Katona É, Szűcs-Somogyi C, Takács R, Vágó J, Oláh T, Bartók Á, Varga Z (2019) N-methyl-D-aspartate (NMDA) receptor expression and function is required for early chondrogenesis. Cell Commun Signal 17:166. https://doi.org/10.1186/s12964-019-0487-3
McKelvey L, Shorten GD, O’Keeffe GW (2013) Nerve growth factor-mediated regulation of pain signalling and proposed new intervention strategies in clinical pain management. J Neurochem 124:276–289. https://doi.org/10.1111/jnc.12093
Mills CD, Nguyen T, Tanga FY, Zhong C, Gauvin DM, Mikusa J, Gomez EJ, Salyers AK, Bannon AW (2013) Characterization of nerve growth factor-induced mechanical and thermal hypersensitivity in rats. Eur J Pain 17:469–479. https://doi.org/10.1002/j.1532-2149.2012.00202.x
Nagakura Y, Ohsaka N, Azuma R, Takahashi S, Takebayashi Y, Kawasaki S, Murai S, Miwa M, Saito H (2018) Monoamine system disruption induces functional somatic syndromes associated symptomatology in mice. Physiol Behav 194:505–514. https://doi.org/10.1016/j.physbeh.2018.07.007
Nencini S, Ringuet M, Kim DH, Chen YJ, Greenhill C, Ivanusic JJ (2017) Mechanisms of nerve growth factor signaling in bone nociceptors and in an animal model of inflammatory bone pain. Mol Pain 13:1744806917697011. https://doi.org/10.1177/1744806917697011
Pattanayak SP, Bose P, Sunita P, Siddique MUM, Lapenna A (2018) Bergapten inhibits liver carcinogenesis by modulating LXR/PI3K/Akt and IDOL/LDLR pathways. Biomed Pharmacother 108:297–308. https://doi.org/10.1016/j.biopha.2018.08.145
Sánchez-Domínguez B, Bullón P, Román-Malo L, Marín-Aguilar F, Alcocer-Gómez E, Carrión AM, Sánchez-Alcazar JA, Cordero MD (2015) Oxidative stress, mitochondrial dysfunction and, inflammation common events in skin of patients with Fibromyalgia. Mitochondrion 21:69–75. https://doi.org/10.1016/j.mito.2015.01.010
Sarchielli P, Alberti A, Floridi A, Gallai V (2001) Levels of nerve growth factor in cerebrospinal fluid of chronic daily headache patients. Neurology 57:132–134. https://doi.org/10.1212/wnl.57.1.132
Singh L, Kaur A, Bhatti MS, Bhatti R (2019a) Possible molecular mediators involved and mechanistic insight into fibromyalgia and associated co-morbidities. Neurochem Res 44:1517–1532. https://doi.org/10.1007/s11064-019-02805-5
Singh G, Kaur A, Kaur J, Bhatti MS, Singh P, Bhatti R (2019b) Bergapten inhibits chemically induced nociceptive behavior and inflammation in mice by decreasing the expression of spinal PARP, iNOS, COX-2 and inflammatory cytokines. Inflammopharmacology 27:749–760. https://doi.org/10.1007/s10787-019-00585-6
Singh G, Singh A, Singh P, Bhatti R (2019c) Bergapten ameliorates vincristine-induced peripheral neuropathy by inhibition of inflammatory cytokines and NFκB signaling. ACS Chem Neurosci 10:3008–3017. https://doi.org/10.1021/acschemneuro.9b00206
Skalicka-Woźniak K, Mendel M, Chłopecka M, Dziekan N (2016) Isolation and evaluation of the myorelaxant effect of bergapten on isolated rat jejunum. Pharm Biol 54:48–54. https://doi.org/10.3109/13880209.2015.1014570
Sørensen LB, Boudreau SA, Gazerani P, Graven-Nielsen T (2019) Enlarged areas of pain and pressure hypersensitivityby spatially distributed intramuscular injections of low-dose nerve growth factor. J Pain 20:566–576. https://doi.org/10.1016/j.jpain.2018.11.005
Staud R, Rodriguez ME (2006) Mechanisms of disease: pain in fibromyalgia syndrome. Nat Clin Pract Rheumatol 2:90–98. https://doi.org/10.1038/ncprheum0091
Tully K, Bolshakov VY (2010) Emotional enhancement of memory: how norepinephrine enables synaptic plasticity. Mol Brain 3:15. https://doi.org/10.1186/1756-6606-3-15
Vorhees CV, Williams MT (2006) Morris water maze: procedures for assessing spatial and related forms of learning and memory. Nat Protoc 1:848–858. https://doi.org/10.1038/nprot.2006.116
Wallace DJ, Linker-Israeli M, Hallegua D, Silverman S, Silver D, Weisman MH (2001) Cytokines play an aetiopathogenetic role in fibromyalgia: a hypothesis and pilot study. Rheumatology 40:743–749. https://doi.org/10.1093/rheumatology/40.7.743
Wang Y (2007) Lipopolysaccharide-induced upregulation of tumor necrosis factor-α (TNF-α) and TNF receptors in rat sciatic nerve. J Mol Neurosci 32:207–216. https://doi.org/10.1007/s12031-007-0036-1
Weber T, Meinhardt M, Zastrow S, Wienke A, Fuessel S, Wirth MP (2013) Immunohistochemical analysis of prognostic protein markers for primary localized clear cell renal cell carcinoma. Cancer Investig 31:51–59. https://doi.org/10.3109/07357907.2012.749267
Woolf AD, Pfleger B (2003) Burden of major musculoskeletal conditions. Bull World Health Organ 81:646–656
Wu L, Wang X, Xu W, Farzaneh F, Xu R (2009) The structure and pharmacological functions of coumarins and their derivatives. Curr Med Chem 16:4236–4260. https://doi.org/10.2174/092986709789578187
Yarim E et al (2019) Blockade of spinal α5-GABAA receptors differentially reduces reserpine-induced fibromyalgia-type pain in female rats. Eur J Pharmacol 858:172443. https://doi.org/10.1016/j.ejphar.2019.172443
Zhang Q, Tan Y (2011) Nerve growth factor augments neuronal responsiveness to noradrenaline in cultured dorsal root ganglion neurons of rats. Neuroscience 193:72–79. https://doi.org/10.1016/j.neuroscience.2011.07.027
Funding
Rajbir Bhatti received grant from Department of Science and technology (DST), India, under DST-SERB scheme (EMR/2016/005878), DST-Purse scheme and UGC-RUSA scheme. Anudeep Kaur received fellowship grant from University grants commission (UGC) under Maulana Azad National Fellowship (UGC-MANF) scheme (Award no F1-17.1/201718/MANF-2017-18-PUN-84339).
Author information
Authors and Affiliations
Contributions
Rajbir Bhatti designed the study hypothesis and conceptualized the work. Anudeep Kaur performed the animal studies. Lovedeep Singh and Harmanpreet Kaur performed histological experiments. Nirmal Singh assisted in statistical analyses of results. Saweta Garg helped in animal studies.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethics Approval
Institutional Animal Ethics Committee (IAEC226/CPSCEA2016/05)
Consent to Participate
Yes.
Consent for Publication
Yes, all authors consent to publication in the journal.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
ESM 1
(PDF 1309 kb)
Rights and permissions
About this article
Cite this article
Kaur, A., Singh, L., Garg, S. et al. Involvement of Oxidative Stress and Nerve Growth Factor in Behavioral and Biochemical Deficits of Experimentally Induced Musculoskeletal Pain in Mice: Ameliorative Effects of Heraclin. J Mol Neurosci 71, 347–357 (2021). https://doi.org/10.1007/s12031-020-01656-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-020-01656-y