Abstract
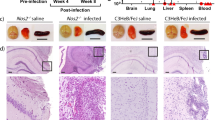
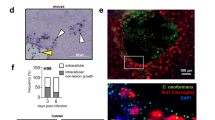
Mycobacterium tuberculosis (M. tuberculosis) invading and activating microglia causes the most serious subtypes of tuberculosis called tubercular meningitis. However, the developmental process of tubercular meningitis, especially the early phase, is poorly understood due to lacking well-established and well-accepted visible models in vitro and in vivo. Here, consistent with one recent report, we found Mycobacterium marinum (M. marinum) invade the zebrafish brain and subsequently cause granuloma-like structures. We further showed that M. marinum, which shares similar characteristics with M. tuberculosis, can invade microglia and replicate in microglia, which subsequently promote the secretion of pro-inflammatory cytokines such as IL-1β, IL-6, and TNF-α. M. marinum infection in microglia can also promote autophagy, which conversely limits the replication of M. marinum. Thus, pharmacological activation of autophagy by rapamycin could prevent M. marinum replication. Our study provides in vivo and in vitro models to study underlying pathogenic mechanisms of tubercular meningitis by using M. marinum. Our results also showed that activation of autophagy could be a meaningful way to prevent tubercular meningitis.




Similar content being viewed by others
References
Be NA, Kim KS, Bishai WR, Jain SK (2009) Pathogenesis of central nervous system tuberculosis. Curr Mol Med 9(2):94–99. https://doi.org/10.2174/156652409787581655
Benard EL, van der Sar AM, Ellett F, Lieschke GJ, Spaink HP, Meijer AH (2012) Infection of zebrafish embryos with intracellular bacterial pathogens. J Vis Exp (61). JoVE. https://doi.org/10.3791/3781
Berg RD, Levitte S, O'Sullivan MP, O'Leary SM, Cambier CJ, Cameron J, Takaki KK, Moens CB, Tobin DM, Keane J, Ramakrishnan L (2016) Lysosomal disorders drive susceptibility to tuberculosis by compromising macrophage migration. Cell 165(1):139–152. https://doi.org/10.1016/j.cell.2016.02.034
Beyer M, Gimsa U, Eyupoglu IY, Hailer NP, Nitsch R (2000) Phagocytosis of neuronal or glial debris by microglial cells: upregulation of MHC class II expression and multinuclear giant cell formation in vitro. Glia 31(3):262–266. https://doi.org/10.1002/1098-1136(200009)31:3<262::AID-GLIA70>3.0.CO;2-2
Carson MJ, Thrash JC, Lo D (2004) Analysis of microglial gene expression: identifying targets for CNS neurodegenerative and autoimmune disease. Am J Pharmacogenomics 4(5):321–330. https://doi.org/10.2165/00129785-200404050-00005
Dinarello CA (1994) The interleukin-1 family: 10 years of discovery. FASEB J 8(15):1314–1325
Garcia-Perez BE, Mondragon-Flores R, Luna-Herrera J (2003) Internalization of Mycobacterium tuberculosis by macropinocytosis in non-phagocytic cells. Microb Pathog 35(2):49–55. https://doi.org/10.1016/S0882-4010(03)00089-5
Guirado E, Schlesinger LS (2013) Modeling the Mycobacterium tuberculosis granuloma—the critical battlefield in host immunity and disease. Front Immunol 4:98
Gutierrez MG, Master SS, Singh SB, Taylor GA, Colombo MI, Deretic V (2004) Autophagy is a defense mechanism inhibiting BCG and Mycobacterium tuberculosis survival in infected macrophages. Cell 119(6):753–766. https://doi.org/10.1016/j.cell.2004.11.038
Henn A, Lund S, Hedtjarn M, Schrattenholz A, Porzgen P, Leist M (2009) The suitability of BV2 cells as alternative model system for primary microglia cultures or for animal experiments examining brain inflammation. ALTEX 26(2):83–94
Hirt UA, Leist M (2003) Rapid, noninflammatory and PS-dependent phagocytic clearance of necrotic cells. Cell Death Differ 10(10):1156–1164. https://doi.org/10.1038/sj.cdd.4401286
Kaushik DK, Gupta M, Das S, Basu A (2010) Kruppel-like factor 4, a novel transcription factor regulates microglial activation and subsequent neuroinflammation. J Neuroinflammation 7(1):68. https://doi.org/10.1186/1742-2094-7-68
Kingkaew N, Sangtong B, Amnuaiphon W, Jongpaibulpatana J, Mankatittham W, Akksilp S, Sirinak C, Nateniyom S, Burapat C, Kittikraisak W, Monkongdee P, Varma JK (2009) HIV-associated extrapulmonary tuberculosis in Thailand: epidemiology and risk factors for death. Int J Infect Dis 13(6):722–729. https://doi.org/10.1016/j.ijid.2008.11.013
Klionsky DJ, Cuervo AM, Seglen PO (2007) Methods for monitoring autophagy from yeast to human. Autophagy 3(3):181–206. https://doi.org/10.4161/auto.3678
Kreutzberg GW (1996) Microglia: a sensor for pathological events in the CNS. Trends Neurosci 19(8):312–318. https://doi.org/10.1016/0166-2236(96)10049-7
Levine B (2005) Eating oneself and uninvited guests: autophagy-related pathways in cellular defense. Cell 120(2):159–162. https://doi.org/10.1016/j.cell.2005.01.005
Levitte S, Adams KN, Berg RD, Cosma CL, Urdahl KB, Ramakrishnan L (2016) Mycobacterial acid tolerance enables phagolysosomal survival and establishment of tuberculous infection in vivo. Cell Host Microbe 20(2):250–258. https://doi.org/10.1016/j.chom.2016.07.007
Lienard J, Movert E, Valfridsson C, Sturegard E, Carlsson F (2016) ESX-1 exploits type I IFN-signalling to promote a regulatory macrophage phenotype refractory to IFNgamma-mediated autophagy and growth restriction of intracellular mycobacteria. Cell Microbiol 18(10):1471–1485. https://doi.org/10.1111/cmi.12594
Lund S, Christensen KV, Hedtjarn M, Mortensen AL, Hagberg H, Falsig J, Hasseldam H, Schrattenholz A, Porzgen P, Leist M (2006) The dynamics of the LPS triggered inflammatory response of murine microglia under different culture and in vivo conditions. J Neuroimmunol 180(1-2):71–87. https://doi.org/10.1016/j.jneuroim.2006.07.007
Marino G, Lopez-Otin C (2004) Autophagy: molecular mechanisms, physiological functions and relevance in human pathology. Cell Mol Life Sci 61(12):1439–1454. https://doi.org/10.1007/s00018-004-4012-4
Miura TA, Holmes KV (2009) Host-pathogen interactions during coronavirus infection of primary alveolar epithelial cells. J Leukoc Biol 86(5):1145–1151. https://doi.org/10.1189/jlb.0209078
Munoz S, Rivas-Santiago B, Enciso JA (2009) Mycobacterium tuberculosis entry into mast cells through cholesterol-rich membrane microdomains. Scand J Immunol 70(3):256–263. https://doi.org/10.1111/j.1365-3083.2009.02295.x
Noda T, Ohsumi Y (1998) Tor, a phosphatidylinositol kinase homologue, controls autophagy in yeast. J Biol Chem 273(7):3963–3966. https://doi.org/10.1074/jbc.273.7.3963
Oppenheim JJ, Zachariae CO, Mukaida N, Matsushima K (1991) Properties of the novel proinflammatory supergene “intercrine” cytokine family. Annu Rev Immunol 9(1):617–648. https://doi.org/10.1146/annurev.iy.09.040191.003153
Qin Y, Sun X, Shao X, Cheng C, Feng J, Sun W, Gu D, Liu W, Xu F, Duan Y (2015) Macrophage-microglia networks drive M1 microglia polarization after Mycobacterium infection. Inflammation 38(4):1609–1616. https://doi.org/10.1007/s10753-015-0136-y
Ramakrishnan L (2013) The zebrafish guide to tuberculosis immunity and treatment. Cold Spring Harb Symp Quant Biol 78(0):179–192. https://doi.org/10.1101/sqb.2013.78.023283
Ramilo O, Saez-Llorens X, Mertsola J, Jafari H, Olsen KD, Hansen EJ, Yoshinaga M, Ohkawara S, Nariuchi H, McCracken GJ (1990) Tumor necrosis factor alpha/cachectin and interleukin 1 beta initiate meningeal inflammation. J Exp Med 172(2):497–507. https://doi.org/10.1084/jem.172.2.497
Rawji KS, Mishra MK, Michaels NJ, Rivest S, Stys PK, Yong VW (2016) Immunosenescence of microglia and macrophages: impact on the ageing central nervous system. Brain 139(3):653–661. https://doi.org/10.1093/brain/awv395
SAITO H (1956) Pathogenesis of tuberculous meningitis. Acta Tuberc Jpn 6(1):20–36
Saura J, Tusell JM, Serratosa J (2003) High-yield isolation of murine microglia by mild trypsinization. Glia 44(3):183–189. https://doi.org/10.1002/glia.10274
Sharma SK, Mohan A, Kadhiravan T (2005) HIV-TB co-infection: epidemiology, diagnosis & management. Indian J Med Res 121(4):550–567
Teitelbaum R, Schubert W, Gunther L, Kress Y, Macaluso F, Pollard JW, McMurray DN, Bloom BR (1999) The M cell as a portal of entry to the lung for the bacterial pathogen Mycobacterium tuberculosis. Immunity 10(6):641–650. https://doi.org/10.1016/S1074-7613(00)80063-1
Tobin DM, Ramakrishnan L (2008) Comparative pathogenesis of Mycobacterium marinum and Mycobacterium tuberculosis. Cell Microbiol 10(5):1027–1039. https://doi.org/10.1111/j.1462-5822.2008.01133.x
Tsenova L, Bergtold A, Freedman VH, Young RA, Kaplan G (1999) Tumor necrosis factor {alpha} is a determinant of pathogenesis and disease progression in mycobacterial infection in the central nervous system. PNAS 96(10):5657–5662. https://doi.org/10.1073/pnas.96.10.5657
van der Vaart M, Korbee CJ, Lamers GE, Tengeler AC, Hosseini R, Haks MC, Ottenhoff TH, Spaink HP, Meijer AH (2014) The DNA damage-regulated autophagy modulator DRAM1 links mycobacterial recognition via TLR-MYD88 to autophagic defense [corrected]. Cell Host Microbe 15(6):753–767. https://doi.org/10.1016/j.chom.2014.05.005
van Leeuwen LM, van der Kuip M, Youssef SA, de Bruin A, Bitter W, van Furth AM, van der Sar AM (2014) Modeling tuberculous meningitis in zebrafish using Mycobacterium marinum. Dis Model Mech 7(9):1111–1122. https://doi.org/10.1242/dmm.015453
Wang F, Wu LA, Li W, Yang Y, Guo F, Gao Q, Chuang HH, Shoff L, Wang W, Chen S (2013) Immortalized mouse dental papilla mesenchymal cells preserve odontoblastic phenotype and respond to bone morphogenetic protein 2. In Vitro Cell Dev Biol Anim 49(8):626–637. https://doi.org/10.1007/s11626-013-9641-1
Wolf AJ, Linas B, Trevejo-Nunez GJ, Kincaid E, Tamura T, Takatsu K, Ernst JD (2007) Mycobacterium tuberculosis infects dendritic cells with high frequency and impairs their function in vivo. J Immunol 179(4):2509–2519. https://doi.org/10.4049/jimmunol.179.4.2509
Wolzak NK, Cooke ML, Orth H, van Toorn R (2012) The changing profile of pediatric meningitis at a referral centre in Cape Town, South Africa. J Trop Pediatr 58(6):491–495. https://doi.org/10.1093/tropej/fms031
Yang CS, Lee HM, Lee JY, Kim JA, Lee SJ, Shin DM, Lee YH, Lee DS, El-Benna J, Jo EK (2007) Reactive oxygen species and p47phox activation are essential for the Mycobacterium tuberculosis-induced pro-inflammatory response in murine microglia. J Neuroinflammation 4(1):27. https://doi.org/10.1186/1742-2094-4-27
Acknowledgements
We thank Dr. Dong Liu (Jiangsu Key Laboratory of Neuroregeneration, Co-innovation Center of Neuroregeneration, Nantong University) for the injection of zebrafish.
Funding
This work was supported by the National Natural Science Foundation of China (grant numbers: 81201252, 81771767), a project funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD), a project funded by the Natural the Scientific Research Programme of Jiangsu Province (13KJB310013), and a project funded by the Scientific Research Programme of Nantong (MS12016007, MS12016009).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Electronic supplementary material
Fig. S1
The bacterial cord and granuloma-like structures were not observed in the non-infected zebrafish nerve system. (GIF 47 kb)
Rights and permissions
About this article
Cite this article
Chen, Z., Shao, Xy., Wang, C. et al. Mycobacterium marinum Infection in Zebrafish and Microglia Imitates the Early Stage of Tuberculous Meningitis. J Mol Neurosci 64, 321–330 (2018). https://doi.org/10.1007/s12031-018-1026-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-018-1026-1