Abstract
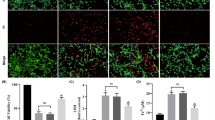
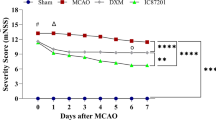
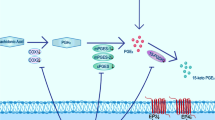
The objective of this study is to explore the neuroprotective effect and mechanism of picroside II on ERK1/2-COX2 signal transduction pathway after cerebral ischemic injury in rats. Focal cerebral ischemic models were established by inserting monofilament threads into the middle cerebral artery in 200 Wistar rats. Twenty four rats were randomly selected into control group, while the other rats were randomly divided into six groups: model group, picroside group, lipopolysaccharide (LPS) with picroside group, U0126 with picroside group, LPS group, and U0126 group with each group containing three subgroups with ischemia at 6, 12, and 24 h. Neurobehavioral function in the rats was evaluated by modified neurological severity score points (mNSS) test; structure of neurons was observed using hematoxylin-eosin (HE) staining; apoptotic cells were counted using terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay; expressions of phosphorylated mitogen/extracellular signal-regulated kinase kinas1/2 (pMEK1/2), phosphorylated extracellular signal-regulated protein kinase1/2 (pERK1/2), and cyclooxygenase (COX2) in the cortex were determined using immunohistochemistry (IHC) and Western blot (WB); and real-time PCR was used to determine the level of COX2 mRNA. The neurological behavioral malfunction appeared in all rats with middle cerebral artery occlusion (MCAO). In the model group, neuron damage was extensive, while the neurobehavioral function score, apoptotic cell index, expression of pMEK1/2, pERK1/2, and COX2 and the level of COX2 mRNA increased significantly when compared to the control group. The peak COX2 mRNA level was in ischemia 12 h, prior to the peak in COX2 protein expression. In the picroside and U0126 groups, the neurological behavioral function was improved, and the number of apoptotic cells and the expression of pMEK1/2, pERK1/2, and COX2 decreased significantly when compared to the model group. In the LPS with picroside group, at ischemia 6 h neuron damage was extensive, and pMEK1/2, pERK1/2, and COX2 expression were much higher than in the model group. But at ischemia 12 and 24 h, the expression of pMEK1/2, pERK1/2, and COX2 decreased slightly, and the neurobehavioral function also improved slightly. In LPS group, neuron damage was extensive, pMEK1/2, pERK1/2, and COX2 expression was still at a high level, and COX2 mRNA peak arrived at ischemic 12 h. Picroside II downregulates COX2 expression after MCAO by inhibiting MEK-ERK1/2 in rats to protect neurons from apoptosis and inflammation.









Similar content being viewed by others
References
Bauer MK, Lieb K, Schulze-Osthoff K, Berger M, Gebicke-Haerter PJ, Bauer J, Fiebich BL (1997) Expression and regulation of cyclooxygenase-2 in rat microglia. Eur J Biochem 243(3):726–731
Bezzi P, Carmignoto G, Pasti L, Vesce S, Rossi D, Rizzini BL, Pozzan T, Volterra A (1998) Prostaglandins stimulate calcium-dependent glutamate release in astrocytes. Nature 391(4):281–285
Bokemeyer D, Sorokin A, Dunn MJ (1996) Multiple intracellular MAP kinase signaling cascades. Kidney Int 49(5):1187–1198
Candelario-Jalil E, González-Falcón A, García-Cabrera M, Alvarez D, Al-Dalain S, Martínez G, León OS, Springer JE (2003) Assessment of the relative contribution of COX-1 and COX-2 isoforms to ischemia-induced oxidative damage and neurodegeneration following transient global cerebral ischemia. J Neurochem 86(3):545–555
Chen J, Sanberg PR, Li Y, Wang L, Lu M, Willing AE, Sanchez-Ramos J, Chopp M (2001) Intravenous administration of human umbilical cord blood reduces behavioral deficits after stroke in rats. Stroke 32(11):2682–688
Collaço-Moraes Y, Aspey B, Harrison M, de Belleroche J (1996) Cyclo-oxygenase-2 messenger RNA induction in focal cerebral ischemia. J Cereb Blood Flow Metab 16(6):1366–1372
Fadeel B, Orrenius S (2005) Apoptosis: a basic biological phenomenon with wide-ranging implications in human disease. J Intern Med 258(6):479–517
Guan Z, Buckman SY, Miller BW, Springer LD, Morrison AR (1998) Morrison, Interleukin-1β-induced cyclooxygenase-2 expression requires activation of both c-Jun NH2-terminal kinase and p38 MAPK signal pathways in rat renal mesangial cells. J Biol Chem 273(44):28670–28676
Guo YL, Xu XL, Li Q, Li Z, Du F (2010) Anti-inflammation effects of picroside 2 in cerebral ischemic injury rats. Behav Brain Funct 6(1):43–49
Henriksson M, Stenman E, Vikman P, Edvinsson L (2007) MEK1/2 inhibition attenuates vascular ETA and ETB receptor alterations after cerebral ischaemia. Exp Brain Res 178(4):470–476
Hwang D, Jang BC, Yu G, Boudreau M (1997) Expression of mitogen-inducible cyclooxygenase induced by lipopolysaccharide. Biochem Pharmacol 54(1):87–96
Jin H, Hwang SK, Yu K, Anderson HK, Lee YS, Lee KH, Prats AC, Morello D, Beck GR Jr, Cho MH (2006) A high inorganic phosphate diet perturbs brain growth, alters AKT-ERK signaling and results in changes in cap-dependent translation. Toxicol Sci 90(1):221–229
Kim R, Emi M, Tanabe K, Murakami S, Uchida Y, Arihiro K (2006) Regulation and interplay of apoptotic and non-apoptotic cell death. J Pathol 208(3):319–326
Kovalska M, Kovalska L, Pavlikova M, Janickova M, Mikuskova K, Adamkov M, Kaplan P, Tatarkova Z, Lehotsky J (2012) Intracellular signaling MAPK pathway after cerebral ischemia reperfusion injury. Neurochem Res 37(7):1568–1577
Levi G, Minghetti L, Aloisi F (1998) Regulation of prostanoid synthesis in microglial cells and effects of prostaglandin E2 on microglial functions. Biochimie 80(11):899–904
Li P, Matsunaga K, Yamakuni T, Ohizumi Y (2000) Potentiation of nerve growth factor-action by picrosides I and II, natural iridoids, in PC12D cells. Eur J Pharmacol 406(2):203–208
Li P, Matsunaga K, Yamakuni T, Ohizumi Y (2002) Picrosides I and II, selective enhancers of the mitogen-activated protein kinase-dependent signaling pathway in the action of neuritogenic substances on PC12D cells. Life Sci 71(15):1821–1835
Li X, Xu XY, Li Z, Guo YL, Li Q, Li XD, Zhou Z (2010a) Picroside II down-regulates matrix metalloproteinase-9 expression following cerebral ischemia/reperfusion injury in rats[J]. Neural Regen Res 5(18):1403–1407
Li Q, Li Z, Xu XY, Guo YL, Du F (2010b) Neuroprotective properties of picroside II in rat model of focal cerebral ischemia. Int J Mol Sci 11(11):4580–4590
Lu Y, Kang J, Bai Y, Zhang Y, Li H, Yang X, Xiang X, Wang X, Huang Y, Su J, Chen Y, Li B, Sun L (2014) Hyperbaric oxygen enlarges the area of brain damage in MCAO rats by blocking autophagy via ERK1/2 activation. Eur J Pharmacol 728(1):93–99
Luo YM, Qin Z, Chen J (2001) Protective effects of COX2 gene knockout on brain from ischemic injury in mice. Chin J Geriatric Heart Brain Vessel Dis 3(5):144–54
Maddahi A, Ansar S, Chen Q, Edvinsson L (2011) Blockade of the MEK/ERK pathway with a Raf inhibitor prevents activation of pro-inflammatory mediators in cerebral arteries and reduction in cerebral blood flow after subarachnoid hemorrhage in a rat model. J Cereb Blood Flow Metab 31(1):144–54
Minghetti L, Polazzi E, Nicolini A, Creminon C, Levi G (1997) Up-regulation of cyclooxygenase-2 expression in cultured microglia by prostaglandin E2, cyclic AMP and non-steroidal anti-inflammatory drugs. Eur J Neurosci 9(5):934–940
Miyamoto O, Tamae K, Kasai H, Hirakawa H, Hayashida Y, Konishi R, Itano T (2003) Suppression of hyperemia and DNA oxidation by indomethacin in cerebral ischemia. Eur J Pharmacol 459(2-3):179–86
Nagasawa H, Kogure K (1989) Correlation between cerebral blood flow and histologic changes in a new rat model of middle cerebral artery occlusion. Stroke 20(8):1037–1043
Namura S, Nagata I, Kikuchi H, Andreucci M, Alessandrini A (2000) Serine-threonine protein kinase Akt does not mediate ischemic tolerance after global ischemia in the gerbil. J Cereb Blood Flow Metab 20(9):1301–1305
Namura S, Iihara K, Takami S et al (2001) Intravenous administration of MEK inhibitor U0126 affords brain protection against forebrain ischemia and focal cerebral ischemia. PNAS 98(20):11569–11574
Pei HT, Su X, Zhao L, Li HY, Guo YL, Zhang MZ, Xin H (2012) Primary study for the therapeutic dose and time window of picroside II in treating cerebral ischemic injury in rats. Int J Mol Sci 13(3):2551–2562
Roskoski R Jr (2012) ERK1/2 MAP kinases: structure, function, and regulation. Pharmacol Res 66(2):105–143
Schweyer S, Soruri A, Meschter O, Heintze A, Zschunke F, Miosge N, Thelen P, Schlott T, Radzun HJ, Fayyazi A (2004) Cisplatin-induced apoptosis in human malignant testicular germ cell lines depends on MEK ⁄ERK activation. Br J Cancer 91(3):589–598
Shioda N, Han F, Fukunaga K (2009) Role of AKT and ERK signaling in the neurogenesis following brain ischemia. Inter Review Neurobiol 85(1):375–387
Smith WL, Garavito RM, DeWitt DL (1996) Prostaglandin endoperoxide H synthases (cyclooxygenases)-1 and -2. J Biol Chem 271(52):33157–33160
Smith WL, DeWitt DL, Garavito RM (2000) Cyclooxygenases: structural, cellular, and molecular biology. Annu Rev Biochem 69:145–182
Stanciu M, Wang Y, Kentor R, Burke N, Watkins S, Kress G, Reynolds I, Klann E, Angiolieri MR, Johnson JW, DeFranco DB (2000) Persistent activation of ERK contributes to glutamate-induced oxidative toxicity in a neuronal cell line and primary cortical neuron cultures. J Biol Chem 275(16):12200–12206
Stanimirovic D, Shapiro A, Wong J, Hutchison J, Durkin J (1997) The induction of ICAM-1 in human cerebromicrovascular endothelial cells (HCEC) by ischemia-like conditions promotes enhanced neutrophil/HCEC adhesion. J Neuroimmunol 76(1-2):193–205
Sun L, Li XD, Wang L, Qin LH, Guo YL, Zhou Z (2011) Anti-oxidant effect of picroside II in a rat model of cerebral ischemia/reperfusion injury. Neural Regen Res 6(15):1141–1146
Wang X, Martindale JL, Holbrook NJ (2000) Requirement for ERK activation in cisplatin-induced apoptosis[J]. J Biol Chem 275(50):39435–39443
Wang X, Wang H, Xu L, Rozanski DJ, Sugawara T, Chan PH, Trzaskos JM, Feuerstein GZ (2003) Significant neuroprotection against ischemic brain injury by inhibition of the MEK1 protein kinase in mice: exploration of potential mechanism associated with apoptosis. J Pharmacol Exp Ther 304(1):172–178
Wang ZQ, Wu DC, Huang FP, Yang GY (2004a) Inhibition of MEK/ERK 1/2 pathway reduces pro-inflammatory cytokine interleukin-1 expression in focal cerebral ischemia. Brain Res 996(1):55–56
Wang T, Qin L, Liu B, Liu Y, Wilson B, Eling TE, Langenbach R, Taniura S, Hong JS (2004b) Role of reactive oxygen species in LPS-induced production of prostaglandin E2 in microglia. J Neurochem 88(4):939–947
Ye Z, Wang N, Xia P, Wang E, Yuan Y, Guo Q (2012) Delayed administration of parecoxib, a specific COX-2 inhibitor, attenuated postischemic neuronal apoptosis by phosphorylation Akt and GSK-3β. Neurochem Res 37(2):321–329
Zhang YQ, Chen RW, Chen CQ, Steven GH (2004) Role of Role of cyclooxygenase-2 in injury induced by hypoxia/reoxygenation in cultured rat cortical neurons. Chin J Pathophysiol 20(12):2280–2283
Zhao Y, Patzer A, Herdegen T, Gohlke P, Culman J (2006) Activation of cerebral peroxisome proliferator-activated receptors gamma promotes neuroprotection by attenuation of neuronal cyclooxygenase-2 overexpression after focal cerebral ischemia in rats. FASEB J 20(8):1162–1175
Zhao L, Guo YL, Ji XJ, Zhang MZ (2014) The neuroprotective effect of picroside II via regulating the expression of myelin basic protein after cerebral ischemia injury in rats. BMC Neurosci 15(2):25–33
Acknowledgments
We thank LetPub (www.letpub.com) for its linguistic assistance during the preparation of this manuscript.
Conflict of interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, T., Zhai, L., Zhang, H. et al. Picroside II Inhibits the MEK-ERK1/2-COX2 Signal Pathway to Prevent Cerebral Ischemic Injury in Rats. J Mol Neurosci 57, 335–351 (2015). https://doi.org/10.1007/s12031-015-0623-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-015-0623-5