Abstract
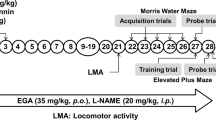
Alzheimer’s disease (AD) is a neurodegenerative disease characterized by impaired memory function and oxidative damage. NO is a major signaling molecule produced in the central nervous system to modulate neurological activity through modulating nitric oxide synthase. Recently, PPAR-γ agonists have shown neuroprotective effects in neurodegenerative disorders. However, there have been only a few studies identifying mechanisms through which cognitive benefits may be exerted. The present study was designed to investigate the possible nitric oxide mechanism in the protective effect of pioglitazone against streptozotocin (STZ)-induced memory dysfunction. Wistar rats were intracerebroventricularly (ICV) injected with STZ. Then rats were treated with pioglitazone, NO modulators [l-arginine and nitro-l-arginine methyl ester (L-NAME)] for 21 days. Behavioral alterations were assessed in between the study period. Animals were sacrificed immediately after behavioral session, and mito-oxidative parameters, TNF-α, IL-6, and caspase-3 activity were measured. STZ-treated rats showed a memory deficit and significantly increased in mito-oxidative damage and inflammatory mediators and apoptosis in the hippocampus. Chronic treatment of pioglitazone significantly improved memory retention and attenuated mito-oxidative damage parameters, inflammatory markers, and apoptosis in STZ-treated rats. However, l-arginine pretreatment with lower dose of pioglitazone has not produced any protective effect as compared to per se. Furthermore, pretreatment of L-NAME significantly potentiated its protective effect, which indicates the involvement of nitric oxide for activation of PPAR-γ action. These results demonstrate that pioglitazone offers protection against STZ-induced memory dysfunction possibly due to its antioxidant, anti-inflammatory, and anti-apoptotic action mediating nitric oxide pathways and, therefore, could have a therapeutic potential in AD.







Similar content being viewed by others
References
Allami N et al (2011) Suppression of nitric oxide synthesis by L-NAME reverses the beneficial effects of pioglitazone on scopolamine-induced memory impairment in mice. Eur J Pharmacol 650:240–248
Amor S, Puentes F, Baker D, van der Valk P (2010) Inflammation in neurodegenerative diseases. Immunology 129:154–169
Beckman JS, Chen J, Crow JP, Ye YZ (1994) Reactions of nitric-oxide, superoxide and peroxynitrite with superoxide-dismutase in neurodegeneration. Neural Regen 103:371–380
Berger J, Moller DE (2002) The mechanisms of action of PPARs. Annu Rev Med 53:409–435
Berman SB, Hastings TG (1999) Dopamine oxidation alters mitochondrial respiration and induces permeability transition in brain mitochondria: implications for Parkinson’s disease. J Neurochem 73:1127–1137
Butow RA, Avadhani NG (2004) Mitochondrial signaling: the retrograde response. Mol Cell 14:1–15
Choi BM, Pae HO, Jang SI, Kim YM, Chung HT (2002) Nitric oxide as a pro-apoptotic as well as anti-apoptotic modulator. J Biochem Mol Biol 35:116–126
Collino M et al (2006) Modulation of the oxidative stress and inflammatory response by PPAR-gamma agonists in the hippocampus of rats exposed to cerebral ischemia/reperfusion. Eur J Pharmacol 530:70–80
Culman J, Zhao Y, Gohlke P, Herdegen T (2007) PPAR-gamma: therapeutic target for ischemic stroke. Trends Pharmacol Sci 28:244–249
Dehmer T, Heneka MT, Sastre M, Dichgans J, Schulz JB (2004) Protection by pioglitazone in the MPTP model of Parkinson’s disease correlates with I kappa B alpha induction and block of NF kappa B and iNOS activation. J Neurochem 88:494–501
Duff K et al (2010) Mild cognitive impairment in prediagnosed Huntington disease. Neurology 75:500–507
Eliasson MJL et al (1999) Neuronal nitric oxide synthase activation and peroxynitrite formation in ischemic stroke linked to neural damage. J Neurosci 19:5910–5918
Ellman GL (1959) Tissue Sulfhydryl Groups. Arch Biochem Biophys 82:70–77
Ellman Gl, Courtney KD, Andres V Jr, Feather-Stone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95
Feil R, Kleppisch T (2008) NO/cGMP-dependent modulation of synaptic transmission. Handb Exp Pharmacol. 184:529–560
Feinstein DL (2003) Therapeutic potential of peroxisome proliferator-activated receptor agonists for neurological disease. Diabetes Technol Ther 5:67–73
Feinstein DL et al (2005) Receptor-independent actions of PPAR thiazolidinedione agonists: is mitochondrial function the key? Biochem Pharmacol 70:177–188
Fuenzalida K et al (2007) Peroxisome proliferator-activated receptor gamma up-regulates the Bcl-2 anti-apoptotic protein in neurons and induces mitochondrial stabilization and protection against oxidative stress and apoptosis. J Biol Chem 282:37006–37015
Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR (1982) Analysis of nitrate, nitrite, and [15N]nitrate in biological fluids. Anal Biochem 126:131–138
Grunblatt E, Salkovic-Petrisic M, Osmanovic J, Riederer P, Hoyer S (2007) Brain insulin system dysfunction in streptozotocin intracerebroventricularly treated rats generates hyperphosphorylated tau protein. J Neurochem 101:757–770
Guo J et al (2010) Impaired neural stem/progenitor cell proliferation in streptozotocin-induced and spontaneous diabetic mice. Neurosci Res 68:329–336
Ha HC, Snyder SH (1999) Poly(ADP-ribose) polymerase is a mediator of necrotic cell death by ATP depletion. Proc Natl Acad Sci U S A 96:13978–13982
Hoyer S, Muller D, Plaschke K (1994) Desensitization of brain insulin receptor. Effect on glucose/energy and related metabolism. J Neural Transm Suppl 44:259–268
Kapadia R, Yi JH, Vemuganti R (2008) Mechanisms of anti-inflammatory and neuroprotective actions of PPAR-gamma agonists. Front Biosci 13:1813–1826
Kaundal RK, Sharma SS (2010) Peroxisome proliferator-activated receptor gamma agonists as neuroprotective agents. Drug News Perspect 23:241–256
Kiaei M, Kipiani K, Chen J, Calingasan NY, Beal MF (2005) Peroxisome proliferator-activated receptor-gamma agonist extends survival in transgenic mouse model of amyotrophic lateral sclerosis. Exp Neurol 191:331–336
King TE (1967) Preparation of succinate dehydrogenase and reconstitution of succinate oxidase. Methods Enzymol 10:322-331
King TE, Howard RL (1967) Preparations and properties of soluble NADH dehydrogenases from cardiac muscle. Methods Enzymol 10:275-294
Kono Y (1978) Generation of superoxide radical during autoxidation of hydroxylamine and an assay for superoxide dismutase. Arch Biochem Biophys 186:189–195
Kubera M, Obuchowicz E, Goehler L, Brzeszcz J, Maes M (2011) In animal models, psychosocial stress-induced (neuro)inflammation, apoptosis and reduced neurogenesis are associated to the onset of depression. Prog Neuropsychopharmacol Biol Psychiatry 35:744–759
Kumar A, Vashist A, Kumar P (2010) Potential role of pioglitazone, caffeic acid and their combination against fatigue syndrome-induced behavioural, biochemical and mitochondrial alterations in mice. Inflammopharmacology 18:241–251
Landreth GE, Heneka MT (2001) Anti-inflammatory actions of peroxisome proliferator-activated receptor gamma agonists in Alzheimer’s disease. Neurobiol Aging 22:937–944
Law A, Gauthier S, Quirion R (2001) Say NO to Alzheimer’s disease: the putative links between nitric oxide and dementia of the Alzheimer’s type. Brain Res Rev 35:73–96
Li Z et al (2010) Caspase-3 activation via mitochondria is required for long-term depression and AMPA receptor internalization. Cell 141:859–871
Liu Y, Peterson DA, Kimura H, Schubert D (1997) Mechanism of cellular 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) reduction. J Neurochem 69:581–593
Liu RL, Liu W, Doctrow SR, Baudry M (2003) Iron toxicity in organotypic cultures of hippocampal slices: role of reactive oxygen species. J Neurochem 85:492–502
Liu P, Fleete MS, Jing Y, Collie ND, Curtis MA, Waldvogel HJ, Faull RL, Abraham WC, Zhang H (2014) Altered arginine metabolism in Alzheimer’s disease brains. Neurobiol Aging 35:1992–2003
Luo Y, Yue W, Quan X, Wang Y, Zhao B, Lu Z (2015) Asymmetric dimethylarginine exacerbates Aβ-induced toxicity and oxidative stress in human cell and Caenorhabditis elegans models of Alzheimer disease. Free Radic Biol Med 79:117–126
Matsumoto T, Noguchi E, Kobayashi T, Kamata K (2007) Mechanisms underlying the chronic pioglitazone treatment-induced improvement in the impaired endothelium-dependent relaxation seen in aortas from diabetic rats. Free Radic Biol Med 42:993–1007
Merrill JE, Benveniste EN (1996) Cytokines in inflammatory brain lesions: helpful and harmful. Trends Neurosci 19:331–338
Meyer RC, Spangler EL, Patel N, London ED, Ingram DK (1998) Impaired learning in rats in a 14-unit T-maze by 7-nitroindazole, a neuronal nitric oxide synthase inhibitor, is attenuated by the nitric oxide donor, molsidomine. Eur J Pharmacol 341:17–22
Miglio G, Rosa AC, Rattazzi L, Collino M, Lombardi G, Fantozzi R (2009) PPAR gamma stimulation promotes mitochondrial biogenesis and prevents glucose deprivation-induced neuronal cell loss. Neurochem Int 55:496–504
Moreno S, Farioli-Vecchioli S, Ceru MP (2004) Immunolocalization of peroxisome proliferator-activated receptors and retinoid X receptors in the adult rat CNS. Neuroscience 123:131–145
Nicolakakis N, Hamel E (2010) The nuclear receptor PPARgamma as a therapeutic target for cerebrovascular and brain dysfunction in Alzheimer’s disease. Front Aging Neurosci. doi:10.3389/fnagi.2010.00021
Penna C, Perrelli MG, Pagliaro P (2012) Mitochondrial pathways, permeability transition pore and redox signaling in cardioprotection: therapeutic implications. Antioxid Redox Signal 18(5):556–599
Pike KE et al (2011) Cognition and beta-amyloid in preclinical Alzheimer’s disease: data from the AIBL study. Neuropsychologia 49:2384–2390
Piotrowski P, Wierzbicka K, Smialek M (2001) Neuronal death in the rat hippocampus in experimental diabetes and cerebral ischaemia treated with antioxidants. Folia Neuropathol 39:147–154
Prakash AK, Kumar A (2009) Effect of chronic treatment of carvedilol on oxidative stress in an intracerebroventricular streptozotocin induced model of dementia in rats. J Pharm Pharmacol 61:1665–1672
Prakash A, Kumar A (2013) Pioglitazone alleviates the mitochondrial apoptotic pathway and mito-oxidative damage in the d-galactose-induced mouse model. Clin Exp Pharmacol Physiol 40:644–651
Prickaerts J, Fahrig T, Blokland A (1999) Cognitive performance and biochemical markers in septum, hippocampus and striatum of rats after an i.c.v. injection of streptozotocin: a correlation analysis. Behav Brain Res 102:73–88
Raza H, John A (2012) Streptozotocin-induced cytotoxicity, oxidative stress and mitochondrial dysfunction in human hepatoma HepG2 cells. Int J Mol Sci 13:5751–5767
Riobo NA et al (2001) Nitric oxide inhibits mitochondrial NADH:ubiquinone reductase activity through peroxynitrite formation. Biochem J 359:139–145
Salkovic-Petrisic M, Hoyer S (2007) Central insulin resistance as a trigger for sporadic Alzheimer-like pathology: an experimental approach. J Neural Transm Suppl 72:217–233
Sauerbeck A et al (2011) Pioglitazone attenuates mitochondrial dysfunction, cognitive impairment, cortical tissue loss, and inflammation following traumatic brain injury. Exp Neurol 227:128–135
Schnegg CI, Robbins ME (2011) Neuroprotective mechanisms of PPARdelta: modulation of oxidative stress and inflammatory processes. PPAR Res 2011:373560
Siemers E (2011) Designing clinical trials for early (pre-dementia) Alzheimer’s disease: determining the appropriate population for treatment. J Nutr Health Aging 15:22–24
Sottocasa GL, Kuylenstierna B, Ernster L, Bergstrand A (1967) An electron-transport system associated with the outer membrane of liver mitochondria. A biochemical and morphological study. J Cell Biol 32:415–438
Steinert JR, Chernova T, Forsythe ID (2010) Nitric oxide signaling in brain function, dysfunction, and dementia. Neuroscientist 16:435–452
Storer PD, Xu J, Chavis J, Drew PD (2005) Peroxisome proliferator-activated receptor-gamma agonists inhibit the activation of microglia and astrocytes: implications for multiple sclerosis. J Neuroimmunol 161:113–122
Susswein AJ, Katzoff A, Miller N, Hurwitz I (2004) Nitric oxide and memory. Neuroscientist 10:153–162
Tan SE (2007) Roles of hippocampal nitric oxide and calcium/calmodulin-dependent protein kinase II in inhibitory avoidance learning in rats. Behav Pharmacol 18:29–38
Tota S, Kamat PK, Shukla R, Nath C (2011) Improvement of brain energy metabolism and cholinergic functions contributes to the beneficial effects of silibinin against streptozotocin induced memory impairment. Behav Brain Res 221:207–215
Turrens JF (1997) Superoxide production by the mitochondrial respiratory chain. Biosci Rep 17:3–8
Valina LD, Ted MD (1996) Nitric oxide neurotoxicity. J Chem Neuroanat 10:179–190
Van Dyke K, Jabbour N, Hoeldtke R, Van Dyke C, Van Dyke M (2010) Oxidative/nitrosative stresses trigger type I diabetes: preventable in streptozotocin rats and detectable in human disease. Ann N Y Acad Sci 1203:138–145
Wills ED (1966) Mechanisms of lipid peroxide formation in animal tissues. Biochem J 99:667–676
Yamada K et al (1995) Role of nitric oxide in learning and memory and in monoamine metabolism in the rat brain. Br J Pharmacol 115:852–858
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Prakash, A., Kumar, A., Ming, L.C. et al. Modulation of the Nitrergic Pathway via Activation of PPAR-γ Contributes to the Neuroprotective Effect of Pioglitazone Against Streptozotocin-Induced Memory Dysfunction. J Mol Neurosci 56, 739–750 (2015). https://doi.org/10.1007/s12031-015-0508-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-015-0508-7