Abstract
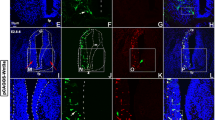
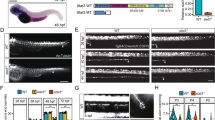
The Wingless/Integrated (Wnt) signaling pathway plays important roles in central nervous system (CNS) development and regeneration, and β-catenin, the central component, has been considered in association with adult neurogenesis. To decipher its roles on spontaneous spinal cord regeneration, we cloned β-catenin from Gekko japonicus and examined its function in regenerating spinal cord. The protein was localized in the neurons and oligodendrocytes and maintained a stable expression levels during the spinal cord regeneration. The temporal pattern of expression has been found to be completely distinct with those of glycogen synthase kinase 3β (GSK3β). Experiments of gain-of-function by overexpression of full length β-catenin or stabilized ΔN90-β-catenin revealed that the accumulated protein attenuates the elongation of neurites and oligodendrocyte process. Knockdown of endogenous β-catenin, however, decreased proliferation of oligodendrocytes by affecting expression of downstream lef1 and c-jun. The upregulated extracellular matrix fibronectin in injured cord was found to be inefficient in regulation of β-catenin expression. Our results suggest that a tightly regulated stable expression of β-catenin is required for the spontaneous spinal cord regeneration.







Similar content being viewed by others
References
Alföldi J, Di Palma F, Grabherr M, Williams C, Kong L, Mauceli E, Russell P, Lowe CB, Glor RE, Jaffe JD, Ray DA, Boissinot S, Shedlock AM, Botka C, Castoe TA, Colbourne JK, Fujita MK, Moreno RG, ten Hallers BF, Haussler D, Heger A, Heiman D, Janes DE, Johnson J, de Jong PJ, Koriabine MY, Lara M, Novick PA, Organ CL, Peach SE, Poe S, Pollock DD, de Queiroz K, Sanger T, Searle S, Smith JD, Smith Z, Swofford R, Turner-Maier J, Wade J, Young S, Zadissa A, Edwards SV, Glenn TC, Schneider CJ, Losos JB, Lander ES, Breen M, Ponting CP, Lindblad-Toh K (2011) The genome of the green anole lizard and a comparative analysis with birds and mammals. Nature 477:587–591
Altschul SF, Madden TL, Schäffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Angers S, Moon RT (2009) Proximal events in Wnt signal transduction. Nat Rev Mol Cell Biol 10:468–477
Bielefeld KA, Amini-Nik S, Whetstone H, Poon R, Youn A, Wang J, Alman BA (2011) Fibronectin and beta-catenin act in a regulatory loop in dermal fibroblasts to modulate cutaneous healing. J Biol Chem 286:27687–27697
Daniels DL, Weis WI (2005) Beta-catenin directly displaces Groucho/TLE repressors from Tcf/Lef in Wnt-mediated transcription activation. Nat Struct Mol Biol 12:364–371
Dong Y, Gu Y, Huan Y, Wang Y, Liu Y, Liu M, Ding F, Gu X, Wang Y (2013) HMGB1 protein does not mediate the inflammatory response in spontaneous spinal cord regeneration: a hint for CNS regeneration. J Biol Chem 288:18204–18218
Fancy SP, Baranzini SE, Zhao C, Yuk DI, Irvine KA, Kaing S, Sanai N, Franklin RJ, Rowitch DH (2009) Dysregulation of the Wnt pathway inhibits timely myelination and remyelination in the mammalian CNS. Gene Dev 23:1571–1585
Funayama N, Fagotto F, McCrea P, Gumbiner BM (1995) Embryonic axis induction by the armadillo repeat domain of beta-catenin: evidence for intracellular signaling. J Cell Biol 128:959–968
Gao X, Arlotta P, Macklis JD, Chen J (2007) Conditional knock-out of beta-catenin in postnatal-born dentate gyrus granule neurons results in dendritic malformation. J Neurosci 27:14317–14325
Ha NC, Tonozuka T, Stamos JL, Choi HJ, Weis WI (2004) Mechanism of phosphorylation-dependent binding of APC to beta-catenin and its role in beta-catenin degradation. Mol Cell 15:511–521
Hanai J, Gloy J, Karumanchi SA, Kale S, Tang J, Hu G, Chan B, Ramchandran R, Jha V, Sukhatme VP, Sokol S (2002) Endostatin is a potential inhibitor of Wnt signaling. J Cell Biol 158:529–539
He TC, Sparks AB, Rago C, Hermeking H, Zawel L, da Costa LT, Morin PJ, Vogelstein B, Kinzler KW (1998) Identification of c-MYC as a target of the APC pathway. Science 281:1509–1512
Hovanes K, Li TW, Munguia JE, Truong T, Milovanovic T, Lawrence Marsh J, Holcombe RF, Waterman ML (2001) Beta-catenin-sensitive isoforms of lymphoid enhancer factor-1 are selectively expressed in colon cancer. Nat Genet 28:53–57
Ikeda S, Kishida M, Matsuura Y, Usui H, Kikuchi A (2000) GSK-3beta-dependent phosphorylation of adenomatous polyposis coli gene product can be modulated by beta-catenin and protein phosphatase 2A complexed with Axin. Oncogene 19:537–545
Kawakami Y, Rodriguez Esteban C, Raya M, Kawakami H, Martí M, Dubova I, Izpisúa Belmonte JC (2006) Wnt/beta-catenin signaling regulates vertebrate limb regeneration. Gene Dev 20:3232–3237
Kiecker C, Niehrs C (2001) A morphogen gradient of Wnt/β-catenin signalling regulates anteroposterior neural patterning in Xenopus. Development 128:4189–4201
Koenig A, Mueller C, Hasel C, Adler G, Menke A (2006) Collagen type I induces disruption of E-cadherin-mediated cell-cell contacts and promotes proliferation of pancreatic carcinoma cells. Cancer Res 66:4662–4671
Komiya Y, Habas R (2008) Wnt signal transduction pathways. Organogenesis 4:68–75
Larsen MC, Brake PB, Pollenz RS, Jefcoate CR (2004) Linked expression of Ah receptor, ARNT, CYP1A1, and CYP1B1 in rat mammary epithelia, in vitro, is each substantially elevated by specific extracellular matrix interactions that precede branching morphogenesis. Toxicol Sci 82:46–61
Lee HY, Kléber M, Hari L, Brault V, Suter U, Taketo MM, Kemler R, Sommer L (2004) Instructive role of Wnt/beta-catenin in sensory fate specification in neural crest stem cells. Science 303:1020–1023
Lee-Liu D, Edwards-Faret G, Tapia VS, Larraín J (2013) Spinal cord regeneration: lessons for mammals from non-mammalian vertebrates. Genesis 51:529–544
Lie DC, Colamarino SA, Song HJ, Désiré L, Mira H, Consiglio A, Lein ES, Jessberger S, Lansford H, Dearie AR, Gage FH (2005) Wnt signalling regulates adult hippocampal neurogenesis. Nature 437:1370–1375
Liu Y, Ding F, Liu M, Jiang M, Yang H, Feng X, Gu X (2006) EST-based identification of genes expressed in brain and spinal cord of Gekko japonicus, a species demonstrating intrinsic capacity of spinal cord regeneration. J Mol Neurosci 29:21–28
Liu M, Gu Y, Liu Y, Li J, He J, Lin S, Gu X (2010) Establishment and characterization of two cell lines derived from primary cultures of Gekko japonicus cerebral cortex. Cell Biol Int 34:153–161
Mann B, Gelos M, Siedow A, Hanski ML, Gratchev A, Ilyas M, Bodmer WF, Moyer MP, Riecken EO, Buhr HJ, Hanski C (1999) Target genes of beta-catenin-T cell-factor/lymphoid-enhancer-factor signaling in human colorectal carcinomas. Proc Natl Acad Sci U S A 96:1603–1608
McHedlishvili L, Mazurov V, Grassme KS, Goehler K, Robl B, Tazaki A, Roensch K, Duemmler A, Tanaka EM (2012) Reconstitution of the central and peripheral nervous system during salamander tail regeneration. Proc Natl Acad Sci U S A 109:E2258–E2266
McLean KE, Vickaryous MK (2011) A novel amniote model of epimorphic regeneration: the leopard gecko, Eublepharis macularius. BMC Dev Biol 16:11–50
Pollack AL, Barth AI, Altschuler Y, Nelson WJ, Mostov KE (1997) Dynamics of beta-catenin interactions with APC protein regulate epithelial tubulogenesis. J Cell Biol 137:1651–1662
Qu Q, Sun G, Li W, Yang S, Ye P, Zhao C, Yu RT, Gage FH, Evans RM, Shi Y (2010) Orphan nuclear receptor TLX activates Wnt/beta-catenin signalling to stimulate neural stem cell proliferation and self-renewal. Nat Cell Biol 12:31–40
Rubinfeld B, Albert I, Porfiri E, Fiol C, Munemitsu S, Polakis P (1996) Binding of GSK3beta to the APC-beta-catenin complex and regulation of complex assembly. Science 272:1023–1026
Sato M, Umetsu D, Murakami S, Yasugi T, Tabata T (2006) DWnt4 regulates the dorsoventral specificity of retinal projections in the Drosophila melanogaster visual system. Nat Neurosci 9:67–75
Takeo M, Chou WC, Sun Q, Lee W, Rabbani P, Loomis C, Taketo MM, Ito M (2013) Wnt activation in nail epithelium couples nail growth to digit regeneration. Nature 499:228–232
Tanaka E, Ferretti P (2009) Considering the evolution of regeneration in the central nervous system. Nat Rev Neurosci 10:713–723
Votin V, Nelson WJ, Barth AI (2005) Neurite outgrowth involves adenomatous polyposis coli protein and beta-catenin. J Cell Sci 118:5699–5708
Wang Y, Wang R, Jiang S, Zhou W, Liu Y, Wang Y, Gu Q, Gu Y, Dong Y, Liu M, Gu X, Ding F, Gu X (2011) Gecko CD59 is implicated in proximodistal identity during tail regeneration. Plos One 6:e17878
Wang Y, Dong Y, Song H, Liu Y, Liu M, Yuan Y, Ding F, Gu X, Wang Y (2012a) Involvement of gecko SNAP25b in spinal cord regeneration by promoting outgrowth and elongation of neurites. Int J Biochem Cell Biol 44:2288–2298
Wang Y, Gu Q, Dong Y, Zhou W, Song H, Liu Y, Liu M, Yuan Y, Ding F, Gu X, Wang Y (2012b) Inhibition of gecko GSK-3β promotes elongation of neurites and oligodendrocyte processes but decreases the proliferation of blastemal cells. J Cell Biochem 113:1842–1851
Yu X, Malenka RC (2003) Beta-catenin is critical for dendritic morphogenesis. Nat Neurosci 6:1169–1177
Zhang L, Yang X, Yang S, Zhang J (2011) The Wnt/β-catenin signaling pathway in the adult neurogenesis. Eur J Neurosci 33:1–8
Zumbrunn J, Kinoshita K, Hyman AA, Nathke IS (2001) Binding of the adenomatous polyposis coli protein to microtubules increases microtubule stability and is regulated by GSK3 beta phosphorylation. Curr Biol 11:44–49
Acknowledgments
This study was supported by the Ministry of Science and Technology of China Grants (973 Program, 2014CB542202), the National Natural Science Foundation of China (No. 31171405; No. 31471011), the Natural Science Foundation of Jiangsu Province (BK20131203), and the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
Conflict of Interest
The authors have declared that no competing interests exist.
Author information
Authors and Affiliations
Corresponding author
Additional information
Honghua Song and Lili Man contributed equally to the work.
Rights and permissions
About this article
Cite this article
Song, H., Man, L., Wang, Y. et al. The Regenerating Spinal Cord of Gecko Maintains Unaltered Expression of β-Catenin Following Tail Amputation. J Mol Neurosci 55, 653–662 (2015). https://doi.org/10.1007/s12031-014-0405-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-014-0405-5