Abstract
Background
Small bowel adenocarcinoma (SBA) is a rare disease. Current recommendations are largely extrapolated from the colorectal literature. For node-negative (N −ve) cases, optimally stratifying cases into high or low risk, may help define optimal management. The objective of this analysis was to determine the importance of lymph node sampling for prognostication and to define what number of lymph nodes sampled is adequate.
Methods
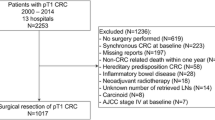
Cases of non-metastatic SBA with complete staging, pathologic, and demographic information were selected from the SEER database and SAS 9.4 software was used. Variables included age, gender, race, grade, TNM staging, and number of lymph nodes were examined. Comparisons were made between N −ve and N +ve cases. Survival analysis using N −ve cases was performed to characterize the impact of nodal sampling on survival and to determine which nodal cut-offs best predict survival.
Results
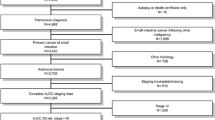
A total of 523 cases from 2004 to 2014 were included in this analysis. Statistically significant differences identified included the median number of nodes sampled between the N −ve and N +ve groups, and the distribution of T stage and grade. Survival analysis in the N −ve cases demonstrated that the strongest predictor of survival was sampling of 16 or more lymph nodes.
Conclusion
In this analysis, lymph node sampling was shown to be the most important pathologic predictor of survival in cases of N −ve SBA. Replicating these findings in a secondary dataset and determining whether a clinical benefit of adjuvant chemotherapy exists for SBA patients with inadequate sampling are both important next steps.




Similar content being viewed by others
References
Bilimoria KY, Bentrem DJ, Wayne JD, Ko CY, Bennett CL, Talamonti MS. Small bowel cancer in the United States: changes in epidemiology, treatment, and survival over the last 20 years. Ann Surg. 2009;249(1):63–71. https://doi.org/10.1097/SLA.0b013e31818e4641.
de Bree E, Rovers KP, Stamatiou D, Souglakos J, Michelakis D, de Hingh IH. The evolving management of small bowel adenocarcinoma. Acta Oncol. 2018;57(6):712–22. https://doi.org/10.1080/0284186X.2018.1433321.
Lepage C, Bouvier AM, Manfredi S, Dancourt V, Faivre J. Incidence and management of primary malignant small bowel cancers: a well-defined French population study. Am J Gastroenterol. 2006;101(12):2826–32. https://doi.org/10.1111/j.1572-0241.2006.00854.x.
Nicholl MB, Ahuja V, Conway WC, Vu VD, Sim MS, Singh G. Small bowel adenocarcinoma: understaged and undertreated? Ann Surg Oncol. 2010;17(10):2728–32. https://doi.org/10.1245/s10434-010-1109-x.
Dabaja BS, Suki D, Pro B, Bonnen M, Ajani J. Adenocarcinoma of the small bowel: presentation, prognostic factors, and outcome of 217 patients. Cancer. 2004;101(3):518–26. https://doi.org/10.1002/cncr.20404.
Bakaeen FG, Murr MM, Sarr MG, Thompson GB, Farnell MB, Nagorney DM, et al. What prognostic factors are important in duodenal adenocarcinoma? Arch Surg. 2000;135(6):635–41 discussion 641-632.
British Columbia Cancer Agency (2018) Small Bowel Malignancies. http://www.bccancer.bc.ca/health-professionals/clinical-resources/cancer-management-guidelines/gastrointestinal/small-bowel-malignancies. Accessed Sept 4 2018.
Locher C, Batumona B, Afchain P, Carrere N, Samalin E, Cellier C, et al. Small bowel adenocarcinoma: French intergroup clinical practice guidelines for diagnosis, treatments and follow-up (SNFGE, FFCD, GERCOR, UNICANCER, SFCD, SFED, SFRO). Dig Liver Dis. 2018;50(1):15–9. https://doi.org/10.1016/j.dld.2017.09.123.
National Comprehensive Cancer Network Colon Cancer (Version 4.2018). https://www.nccn.org/professionals/physician_gls/pdf/colon_blocks.pdf. Accessed January 18, 2018.
Phelps RA, Chidester S, Dehghanizadeh S, Phelps J, Sandoval IT, Rai K, et al. A two-step model for colon adenoma initiation and progression caused by APC loss. Cell. 2009;137(4):623–34. https://doi.org/10.1016/j.cell.2009.02.037.
Najdi R, Holcombe RF, Waterman ML. Wnt signaling and colon carcinogenesis: beyond APC. J Carcinog. 2011;10:5. https://doi.org/10.4103/1477-3163.78111.
Schrock AB, Devoe CE, McWilliams R, Sun J, Aparicio T, Stephens PJ, et al. Genomic profiling of small-bowel adenocarcinoma. JAMA Oncol. 2017;3(11):1546–53. https://doi.org/10.1001/jamaoncol.2017.1051.
Overman MJ, Kopetz S, Lin E, Abbruzzese JL, Wolff RA. Is there a role for adjuvant therapy in resected adenocarcinoma of the small intestine. Acta Oncol. 2010;49(4):474–9. https://doi.org/10.3109/02841860903490051.
Tumbapura A, Kuwada S, DiSario JA. Adenocarcinoma of the small intestine. Curr Treat Options Gastroenterol. 2000;3(1):51–7. https://doi.org/10.1007/s11938-000-0061-4.
Neugut A, Marvin M, Chabot J. Adenocarcinoma of the small bowel. In: Holzheimer R, Mannick J, editors. Surgical treatment: evidence-based and problem-oriented. Munich: Zuckschwerdt; 2001.
Moreno CC, Mittal PK, Sullivan PS, Rutherford R, Staley CA, Cardona K, et al. Colorectal cancer initial diagnosis: screening colonoscopy, diagnostic colonoscopy, or emergent surgery, and tumor stage and size at initial presentation. Clin Colorectal Cancer. 2016;15(1):67–73. https://doi.org/10.1016/j.clcc.2015.07.004.
Negoi I, Paun S, Hostiuc S, Stoica B, Tanase I, Negoi RI, et al. Most small bowel cancers are revealed by a complication. Einstein (Sao Paulo). 2015;13(4):500–5. https://doi.org/10.1590/S1679-45082015AO3380.
Young JI, Mongoue-Tchokote S, Wieghard N, Mori M, Vaccaro GM, Sheppard BC, et al. Treatment and survival of small-bowel adenocarcinoma in the United States: a comparison with colon cancer. Dis Colon Rectum. 2016;59(4):306–15. https://doi.org/10.1097/DCR.0000000000000562.
Ecker BL, McMillan MT, Datta J, Mamtani R, Giantonio BJ, Dempsey DT, et al. Efficacy of adjuvant chemotherapy for small bowel adenocarcinoma: a propensity score-matched analysis. Cancer. 2016;122(5):693–701. https://doi.org/10.1002/cncr.29840.
Kannarkatt J, Joseph J, Kurniali PC, Al-Janadi A, Hrinczenko B. Adjuvant chemotherapy for stage II colon cancer: a clinical dilemma. J Oncol Pract. 2017;13(4):233–41. https://doi.org/10.1200/JOP.2016.017210.
Benson AB 3rd, Schrag D, Somerfield MR, Cohen AM, Figueredo AT, Flynn PJ, et al. American Society of Clinical Oncology recommendations on adjuvant chemotherapy for stage II colon cancer. J Clin Oncol. 2004;22(16):3408–19. https://doi.org/10.1200/JCO.2004.05.063.
Labianca R, Nordlinger B, Beretta GD, Mosconi S, Mandala M, Cervantes A, et al. Early colon cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2013;24(Suppl 6):vi64–72. https://doi.org/10.1093/annonc/mdt354.
Swanson RS, Compton CC, Stewart AK, Bland KI. The prognosis of T3N0 colon cancer is dependent on the number of lymph nodes examined. Ann Surg Oncol. 2003;10(1):65–71.
Le Voyer TE, Sigurdson ER, Hanlon AL, Mayer RJ, Macdonald JS, Catalano PJ, et al. Colon cancer survival is associated with increasing number of lymph nodes analyzed: a secondary survey of intergroup trial INT-0089. J Clin Oncol. 2003;21(15):2912–9. https://doi.org/10.1200/JCO.2003.05.062.
Wu S, Chen JN, Zhang QW, Tang CT, Zhang XT, Tang MY, et al. A new metastatic lymph node classification-based survival predicting model in patients with small bowel adenocarcinoma: a derivation and validation study. EBioMedicine. 2018;32:134–41. https://doi.org/10.1016/j.ebiom.2018.05.022.
Tran TB, Qadan M, Dua MM, Norton JA, Poultsides GA, Visser BC. Prognostic relevance of lymph node ratio and total lymph node count for small bowel adenocarcinoma. Surgery. 2015;158(2):486–93. https://doi.org/10.1016/j.surg.2015.03.048.
Surveillance, Epidemiology, and End Results (SEER) Program (www.seer.cancer.gov) Research Data (1973–2014), National Cancer Institute, DCCPS, Surveillance Research Program, released April 2017, based on the November 2016 submission.
National Cancer Institute Overview of the SEER Program. National Institute of Health. https://seer.cancer.gov/about/overview.html. Accessed Dec 11 2018 2018.
Benchimol EI, Smeeth L, Guttmann A, Harron K, Moher D, Petersen I, et al. The REporting of studies Conducted using Observational Routinely-collected health Data (RECORD) statement. PLoS Med. 2015;12(10):e1001885. https://doi.org/10.1371/journal.pmed.1001885.
Park HS, Lloyd S, Decker RH, Wilson LD, Yu JB. Limitations and biases of the Surveillance, Epidemiology, and End Results database. Curr Probl Cancer. 2012;36(4):216–24. https://doi.org/10.1016/j.currproblcancer.2012.03.011.
Gliklich RE, Dreyer NA, Leavy MB (2014) Registries for evaluating patient outcomes: a user’s guide. Agency for Healthcare Research and Quality (US). https://www.ncbi.nlm.nih.gov/books/NBK208620/. Accessed April 12 2019 2019.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
As this research involved only de-identified patient data, it was exempt from ethical board review and informed consent was not required [31].
Conflict of Interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Thiessen, M., Lee-Ying, R.M., Monzon, J.G. et al. An Examination of Lymph Node Sampling as a Predictor of Survival in Resected Node-Negative Small Bowel Adenocarcinoma: a SEER Database Analysis. J Gastrointest Canc 51, 280–288 (2020). https://doi.org/10.1007/s12029-019-00250-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-019-00250-9