Abstract
Background
We investigated in a porcine model of anhepatic acute liver failure (ALF), the value of two parameters describing cerebrovascular autoregulatory capacity, pressure reactivity index (PRx) and brain tissue oxygen pressure reactivity (ORx), regarding their power to predict the development of intracranial hypertension.
Methods
In six pigs, hepatectomy was performed. Only one animal was sham operated. All animals received neuromonitoring including arterial blood pressure, intracranial pressure (ICP), and brain tissue partial oxygen pressure (PbrO2). The average time of neuromonitoring was 31.0 h. Cerebral perfusion pressures (CPP), cerebrovascular pressure reactivity index (PRx) and brain tissue oxygen reactivity index (ORx) were calculated.
Results
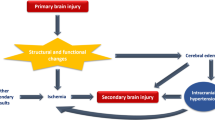
Perioperative disturbance of AR improved within 4 h after surgery. From 6 to 16 h post hepatectomy, ICP did slowly increase by 4 mmHg from baseline; CPP remained stable around 40 mmHg. PRx and ORx, however, indicated in this period a progressive loss of AR, reflected in a decrease of PbrO2 despite unchanged CPP. Beyond 16 h, ICP rose quickly. At CPP levels below 35 mmHg, PbrO2 fell to ischemic levels.
Conclusions
The loss of cerebrovascular autoregulatory capacity, indicated by a rise of PRx and ORx precedes the final crisis of uncontrollable intracranial hypertension in this animal model by hours. During this phase cerebral blood flow, as reflected in tissue oxygenation, deteriorates despite unchanged CPP. Monitoring of AR during ALF therefore seems to carry the power to identify a risk for development of critical CBF and intracranial hypertension.






Similar content being viewed by others
Abbreviations
- AR:
-
Autoregulation
- PRx:
-
Cerebrovascular pressure reactivity index
- ORx:
-
Brain tissue oxygen pressure reactivity
- CBF:
-
Cerebral blood flow
- CPP:
-
Cerebral perfusion pressure
- ALF:
-
Acute liver failure
- TCD:
-
Transcraniel Doppler
- MAP:
-
Mean arterial pressure
- PbrO2 :
-
Brain tissue oxygen
- LLA:
-
Lower limit of the autoregulation
- NO:
-
Nitric oxide
References
Lassen NA. Autoregulation of Cerebral Blood Flow. Circ Res. 1964;15:201–4.
Paulson OB, Strandgaard S, Edvinsson L. Cerebral autoregulation. Cerebrovasc Brain Metab Rev. 1990;2:161–92.
MacKenzie ET, Farrar JK, Fitch W, Graham DI, Gregory PC, Harper AM. Effects of hemorrhagic hypotension on the cerebral circulation. I. Cerebral blood flow and pial arteriolar caliber. Stroke. 1979;10:711–8.
Larsen FS, Ejlersen E, Hansen BA, Knudsen GM, Tygstrup N, Secher NH. Functional loss of cerebral blood flow autoregulation in patients with fulminant hepatic failure. J Hepatol. 1995;23:212–7.
Dethloff TJ, Knudsen GM, Larsen FS. Cerebral blood flow autoregulation in experimental liver failure. J Cereb Blood Flow Metab. 2008;28:916–26.
Keller E, Wietasch G, Ringleb P, Scholz M, Schwarz S, Stingele R, Schwab S, Hanley D, Hacke W. Bedside monitoring of cerebral blood flow in patients with acute hemispheric stroke. Crit Care Med. 2000;28:511–6.
Czosnyka M, Smielewski P, Kirkpatrick P, Menon DK, Pickard JD. Monitoring of cerebral autoregulation in head-injured patients. Stroke. 1996;27:1829–34.
Steinmeier R, Hofmann RP, Bauhuf C, Hubner U, Fahlbusch R. Continuous cerebral autoregulation monitoring by cross-correlation analysis. J Neurotrauma. 2002;19:1127–38.
Lam JM, Hsiang JN, Poon WS. Monitoring of autoregulation using laser Doppler flowmetry in patients with head injury. J Neurosurg. 1997;86:438–45.
Czosnyka M, Brady K, Reinhard M, Smielewski P, Steiner LA. Monitoring of cerebrovascular autoregulation: facts, myths, and missing links. Neurocrit Care. 2009;10:373–86.
Valadka AB, Hlatky R, Furuya Y, Robertson CS. Brain tissue PO2: correlation with cerebral blood flow. Acta Neurochir Suppl. 2002;81:299–301.
Jaeger M, Soehle M, Schuhmann MU, Winkler D, Meixensberger J. Correlation of continuously monitored regional cerebral blood flow and brain tissue oxygen. Acta Neurochir (Wien). 2005;147:51–6; discussion 56.
Czosnyka M, Smielewski P, Kirkpatrick P, Laing RJ, Menon D, Pickard JD. Continuous assessment of the cerebral vasomotor reactivity in head injury. Neurosurgery. 1997;41:11–7; discussion 17–9.
Steiner LA, Coles JP, Johnston AJ, Chatfield DA, Smielewski P, Fryer TD, Aigbirhio FI, Clark JC, Pickard JD, Menon DK, Czosnyka M. Assessment of cerebrovascular autoregulation in head-injured patients: a validation study. Stroke. 2003;34:2404–9.
Brady KM, Lee JK, Kibler KK, Easley RB, Koehler RC, Shaffner DH. Continuous measurement of autoregulation by spontaneous fluctuations in cerebral perfusion pressure: comparison of 3 methods. Stroke. 2008;39:2531–7.
Jaeger M, Schuhmann MU, Soehle M, Meixensberger J. Continuous assessment of cerebrovascular autoregulation after traumatic brain injury using brain tissue oxygen pressure reactivity. Crit Care Med. 2006;34:1783–8.
Radolovich DK, Czosnyka M, Timofeev I, Lavinio A, Hutchinson P, Gupta A, Pickard JD, Smielewski P. Reactivity of brain tissue oxygen to change in cerebral perfusion pressure in head injured patients. Neurocrit Care. 2009;10:274–9.
Jaeger M, Schuhmann MU, Soehle M, Nagel C, Meixensberger J. Continuous monitoring of cerebrovascular autoregulation after subarachnoid hemorrhage by brain tissue oxygen pressure reactivity and its relation to delayed cerebral infarction. Stroke. 2007;38:981–6.
Durham S, Yonas H, Aggarwal S, Darby J, Kramer D. Regional cerebral blood flow and CO2 reactivity in fulminant hepatic failure. J Cereb Blood Flow Metab. 1995;15:329–35.
Dethloff T, Knudsen GM, Hansen BA, Larsen FS. Effects of porta-systemic shunting and ammonia infusion on cerebral blood flow autoregulation in the rat. Neurocrit Care. 2005;3:86–90.
Knubben K, Thiel C, Schenk M, Etspuler A, Schenk T, Morgalla MH, Konigsrainer A. A new surgical model for hepatectomy in pigs. Eur Surg Res. 2008;40:41–6.
Smielewski P, Lavinio A, Timofeev I, Radolovich D, Perkes I, Pickard JD, Czosnyka M. ICM+, a flexible platform for investigations of cerebrospinal dynamics in clinical practice. Acta Neurochir Suppl. 2008;102:145–51.
Jaeger M, Dengl M, Meixensberger J, Schuhmann MU. Effects of cerebrovascular pressure reactivity-guided optimization of cerebral perfusion pressure on brain tissue oxygenation after traumatic brain injury. Crit Care Med. 2010;38:1343–7.
Jaeger M. Different indices to assess cerebrovascular autoregulation have different dynamic properties. Neurocrit Care. 2010;13:163–5.
Johnston AJ, Steiner LA, Coles JP, Chatfield DA, Fryer TD, Smielewski P, Hutchinson PJ, O’Connell MT, Al-Rawi PG, Aigbirihio FI, et al. Effect of cerebral perfusion pressure augmentation on regional oxygenation and metabolism after head injury. Crit Care Med. 2005;33:189–95; discussion 255–7.
Strauss G, Hansen BA, Kirkegaard P, Rasmussen A, Hjortrup A, Larsen FS. Liver function, cerebral blood flow autoregulation, and hepatic encephalopathy in fulminant hepatic failure. Hepatology. 1997;25:837–9.
Master S, Gottstein J, Blei AT. Cerebral blood flow and the development of ammonia-induced brain edema in rats after portacaval anastomosis. Hepatology. 1999;30:876–80.
Larsen FS, Gottstein J, Blei AT. Cerebral hyperemia and nitric oxide synthase in rats with ammonia-induced brain edema. J Hepatol. 2001;34:548–54.
Jalan R, Olde Damink SW, Hayes PC, Deutz NE, Lee A. Pathogenesis of intracranial hypertension in acute liver failure: inflammation, ammonia and cerebral blood flow. J Hepatol. 2004;41:613–20.
Brian JE Jr, Moore SA, Faraci FM. Expression and vascular effects of cyclooxygenase-2 in brain. Stroke. 1998;29:2600–6.
Vaquero J, Butterworth RF. Mechanisms of brain edema in acute liver failure and impact of novel therapeutic interventions. Neurol Res. 2007;29:683–90.
Vaquero J, Chung C, Blei AT. Cerebral blood flow in acute liver failure: a finding in search of a mechanism. Metab Brain Dis. 2004;19:177–94.
Rama Rao KV, Jayakumar AR, Norenberg MD. Role of oxidative stress in the ammonia-induced mitochondrial permeability transition in cultured astrocytes. Neurochem Int. 2005;47:31–8.
Bjerring PN, Hauerberg J, Frederiksen HJ, Jorgensen L, Hansen BA, Tofteng F, Larsen FS. Cerebral glutamine concentration and lactate–pyruvate ratio in patients with acute liver failure. Neurocrit Care. 2008;9:3–7.
Bell MJ, Kochanek PM, Carcillo JA, Mi Z, Schiding JK, Wisniewski SR, Clark RS, Dixon CE, Marion DW, Jackson E. Interstitial adenosine, inosine, and hypoxanthine are increased after experimental traumatic brain injury in the rat. J Neurotrauma. 1998;15:163–70.
Clark RS, Carcillo JA, Kochanek PM, Obrist WD, Jackson EK, Mi Z, Wisneiwski SR, Bell MJ, Marion DW. Cerebrospinal fluid adenosine concentration and uncoupling of cerebral blood flow and oxidative metabolism after severe head injury in humans. Neurosurgery. 1997;41:1284–92; discussion 1292–3.
Kristiansen RG, Lindal S, Myreng K, Revhaug A, Ytrebo LM, Rose CF. Neuropathological changes in the brain of pigs with acute liver failure. Scand J Gastroenterol. 2010;45:935–43.
Menon DK, Coles JP, Gupta AK, Fryer TD, Smielewski P, Chatfield DA, Aigbirhio F, Skepper JN, Minhas PS, Hutchinson PJ, et al. Diffusion limited oxygen delivery following head injury. Crit Care Med. 2004;32:1384–90.
Rama Rao KV, Jayakumar AR, Tong X, Curtis KM, Norenberg MD. Brain aquaporin-4 in experimental acute liver failure. J Neuropathol Exp Neurol. 2010;69:869–79.
Acknowledgments
The authors thank T. O. Greiner, A. Stolz, and M. Seitzer for their excellent veterinary and technical assistance. The authors owe great thanks to P. Lewis, M. Jaeger, and M. Czosnyka for critically reading and commenting on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Grözinger, G., Schenk, M., Morgalla, M.H. et al. The Values of Cerebrovascular Pressure Reactivity and Brain Tissue Oxygen Pressure Reactivity in Experimental Anhepatic Liver Failure. Neurocrit Care 17, 271–280 (2012). https://doi.org/10.1007/s12028-012-9714-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-012-9714-0