Abstract
Introduction
Neurologic disorders with autoimmune dysregulation are commonly encountered in the critical care setting. Frequently encountered diseases include Guillain–Barré syndrome (GBS), myasthenia gravis, multiple sclerosis, acute demyelinating encephalomyelitis, and encephalitides. Immunomodulatory therapies, including high-dose corticosteroids, plasmapheresis, and intravenous immunoglobulins, are the cornerstone of the treatment of these diseases. Here we review the efficacy and side effects of immunomodulatory therapies commonly utilized in critically ill neurologic patients in the intensive care setting.
Methods
Search of Medline, Cochrane databases, and manual review of article bibliographies.
Results
The efficacy of high-dose corticosteroids, plasmapheresis, and intravenous immunoglobulins have been studied extensively in GBS, myasthenia gravis, and demyelinating disorders such as multiple sclerosis and acute demyelinating encephalomyelitis. For these diseases, however, the duration of treatment, dosing regimens, and choices among different therapeutic modalities remain controversial. For many of the other diseases (e.g., encephalitis and status epilepticus of autoimmune etiology) discussed in this review, evidence is limited to small case series.
Conclusions
There is good evidence for the efficacy and tolerability of immunomodulatory therapies in GBS, myasthenia gravis, and acute central nervous system demyelination, though data to establish superiority of one therapeutic regimen over another remains lacking. For most other conditions, the data for immunomodulatory therapies are limited, and further research is required.
Similar content being viewed by others
Introduction
Neurologic disorders with autoimmune dysregulation as a central feature are an important indication for admission to critical care units. At one large Neurosciences Critical Care Unit, for example, admissions for non-vascular and non-traumatic etiologies comprised 30% of 1,155 consecutive admissions; of these admissions, about one-third had a primary autoimmune etiology (Guillain–Barré syndrome, myasthenia gravis, multiple sclerosis, acute demyelinating encephalomyelitis) and one half had a disease process in which autoimmunity may play a substantial role (encephalitis/meningitis, status epilepticus) [1]. Many of these diseases are treated with high-dose corticosteroids, plasmapheresis, or intravenous (IV) immunoglobulins. We will briefly review the mechanism of action and side effects of these medications and then critically appraise the use of immunomodulatory therapies in the treatment of neurological diseases commonly encountered in the critical care setting.
Corticosteroids
Corticosteroids are thought to act through genomic and non-genomic mechanisms; the genomic effects are mediated by the cytosolic glucocorticoid receptor while the non-genomic effects are likely the result of interactions with receptors on the cell surface. While the non-genomic effects are more rapid, the net effect of both pathways on the immune system is to decrease inflammation by immunosuppression and decreased proliferation of inflammatory cells [2]. Side effects of corticosteroids are well known, but a few features specific to their use in neurologic disease deserve mention. The dosages used for many diseases—typically methylprednisolone 1 g/day for 3–5 days (equivalent to 1,250 mg/d of prednisone)—are much higher than for non-neurologic indications. Well known side effects include cognitive disturbances (agitation, psychosis), gastrointestinal ulcers, myalgia, and arthralgia. In critically ill patients, particularly those exposed to neuromuscular blocking agents, there is an increased risk of critical illness neuropathy and/or myopathy [3]. There have also been theoretical concerns regarding an increased risk of infection; however, there does not appear to be a clinically relevant increase in infections in a variety of different disease states [3–5].
Plasmapheresis
Plasmapheresis eliminates circulating immunoglobulins and immune complexes directed at components of the central and peripheral nervous system. The technique involves separation of plasma from the cellular elements of blood, either by centrifugation or through permeable filters. Filter pore diameters measure up to 0.2 μM, resulting in filtration of substances up to a molecular weight of approximately 3 × 106 Da. Because filtration results in hemoconcentration, replacement solution is necessary for the plasma discarded during plasmapheresis. The most common choices are fresh-frozen plasma (FFP) or combinations of colloid and crystalloid (albumin, albumin and saline, or albumin and plasma expanders) [6]. While FFP is preferred for certain diseases (hemolytic-uremic syndrome, thrombotic thrombocytopenic purpura), for neurological indications colloid solutions are generally chosen due to cost and relative paucity of side effects. FFP is associated with a variety of adverse reactions, including severe side effects such as transfusion-associated lung injury and anaphylaxis. The incidence of adverse reactions from replacement with FFP is more than 10 times that of albumin replacement (20 vs. 1.4%) [7]. Because plasmapheresis can decrease levels of clotting factors, patients at high risk for bleeding are an exception; in such patients, use of FFP is recommended. Plasmapheresis is generally administered as 5–6 exchanges (daily or every other day) of 1.0–1.5 plasma volumes per treatment with albumin as the typical replacement fluid. Plasmapheresis has been shown to reduce IgG, IgM, and total compliment levels by 63.4, 68.9, and 57.1%, respectively, after one exchange and 80.1, 79.5, and 59.7% after five [8].
Other complications of plasmapheresis relate to the need for central venous access as well as to the technique itself. Generally, plasmapheresis requires a temporary hemodialysis catheter placed in the subclavian or internal jugular vein. Risks include infection, hematoma, and pneumothorax. Hypotension is relatively common, although a decrease from baseline of more than 15% is generally avoided by use of continuous filtration systems. Patients with autonomic nervous system involvement or pre-existing hypotension, however, are at risk for symptomatic hypotension [9]. Because of hemoconcentration, anticoagulation with either heparin or citrate is often necessary. Both may cause bleeding, and heparin both systemically and within the filtration system can result in heparin-induced thrombocytopenia. Citrate can cause hypocalcemia and metabolic alkalosis. Plasmapheresis may also result in reduction of drug levels, particularly of those drugs which are not highly protein bound as well as reduction in IgG levels (>80% reduction from pre-treatment).
Intravenous Immunoglobulins (IVIG)
IVIG consists of purified immunoglobulins from the pooled plasma of up to 100,000 healthy human donors. The beneficial effects of IVIG are multifaceted, but likely include modulation of the Fc receptor by enhancing expression of the inhibitory IgG Fc receptor IIB, interference with complement activation, modulation of pro-inflammatory cytokines, alteration of both T and B cell activation and proliferation, and decrease in inflammatory cell adhesion and diapedesis [10–12].
Overall, adverse reactions occur in less than 5% of patients. Common reactions include headache, chills, and myalgia which may be pretreated with ibuprofen. Chest discomfort within the first hour after infusion is non-cardiac in origin and generally resolves when the infusion is stopped for 30 min. Afterward, it is often possible to avoid this side effect by decreasing the infusion rate [10]. Self-limited aseptic meningitis occasionally occurs 48–72 h after the first dose. Serious reactions are generally uncommon. Venous or arterial thrombo-embolic events due to transient hyper-viscosity have been reported at a rate of between 0.15 and 1.2% per treatment course [13]. This risk may be limited by slower infusion rates [14, 15]. Renal failure due to tubular damage induced by sucrose may result in a reversible increase in creatinine with incidence peaking at about 5 days after initiation of therapy [15]. Although IgG predominates, trace amounts of IgA may be present which rarely results in anaphylaxis in IgA deficient individuals and some centers require determination of IgA levels to rule out IgA deficiency prior to the initiation of IVIG therapy.
Immune-Mediated Neuropathies
Guillain–Barré syndrome (GBS) is characterized by autoimmune demyelination and/or axonal destruction of the peripheral nerves. Current primary criteria for diagnosis include progressive weakness (classically ascending) and areflexia [16]. Supportive of the diagnosis are progression to nadir in <4 weeks, relative symmetry, elevated cerebrospinal fluid (CSF) protein with <10 WBC, and features of slowed nerve conduction or conduction block on nerve conduction studies [16]. The world wide incidence is estimated at 1.3/100,000 with a male predominance [17]. As many as 30% of patients will require ventilatory support [18] and up to 65% will demonstrate autonomic dysfunction [19]. Mortality is approximately 5–10% [18] and about 65% of patients recover fully or with minimal deficits [20].
The immunopathogenesis of GBS likely involves several mechanisms, including molecular mimicry of myelin components by infectious agents (including Clostridium jejuni), antiganglioside antibodies, compliment activation, and host factors [21]. The axonal forms of GBS (acute motor axonal neuropathy (AMAN) and acute motor and sensory axonal neuropathy (AMSAN)) represent 5–10% of cases in North America and are associated with antibodies to GM1, GD1a, GM1b, and GalNAc-GD1a [22]. These variants have a more severe course, increased rate of ventilator dependence, and poorer long-term outcome [21, 23]. GBS with cranial nerve involvement (Miller–Fisher syndrome) is associated with GQ1b, GT1a, GD1b, and GD3 antibodies [22]. Once they bind to their respective epitopes, anti-ganglioside antibodies cause complement activation with variable destruction of the Schwann cell, myelin, and the underlying axon [24, 25].
The efficacy of plasmapheresis was established by the North American Plasma Exchange study in 1985, and confirmed by a later French trial [26, 27]. A Cochrane meta-analysis of six randomized controlled trials (RCTs) evaluating plasmapheresis with a total of 649 participants demonstrated a reduction in time to onset of motor recovery from 10 to 6 days and time to walking without aid from 85–111 to 53–70 days. Plasmapheresis decreased the likelihood of requiring mechanical ventilation (RR 0.53, 95% CI: 0.39–0.74) and decreased total ventilator time by 5.09 days. The odds of full motor recovery at one year were significantly greater (RR 1.24, 95% CI: 1.07–1.45). Treatment was effective when started up to 30 days from onset but most effective if started within 7 days [28, 29].
Six trials of IVIG in GBS with 536 participants have been performed. A Cochrane review reported that IVIG and plasmapheresis are equally efficacious in severe disease [30]. One trial of plasmapheresis followed by IVIG failed to demonstrate incremental benefit [31].
Corticosteroids have also been evaluated in six trials (587 participants) which have consistently shown a trend toward poor outcomes with oral corticosteroids and no significant effect of IV corticosteroids [32].
American Academy of Neurology guidelines conclude that plasmapheresis and IVIG are equally efficacious. Based on level I evidence (Table 1), plasmapheresis is recommended for non-ambulatory patients presenting within 4 weeks and ambulatory patients presenting within 2 weeks. A typical regimen would be 5–6 1.0–1.5 volume exchanges over 7–10 days. IVIG (typically 0.4 g/kg q 24 h for five doses) is recommended for non-ambulatory patients presenting within 2 (and possibly, up to 4) weeks. Oral and IV corticosteroids are discouraged [33].
Chronic inflammatory demyelinating polyradiculoneuropathy (CIDP) is a progressive, relapsing neuropathy which on occasion results in admission to an ICU. Several case series [34, 35] have supported the use of corticosteroids. The only randomized trial (prednisone starting at 120 mg PO QOD and tapering off over 12 weeks) found a modest benefit [36]. Two trials have found a more significant benefit to plasmapheresis, though many patients experienced rebound symptoms following treatment [37, 38]. IVIG has a larger body of evidence. A recent systematic meta-analysis identified five placebo controlled trials (235 patients total) and found a RR for improvement at one month of 2.40 (95% CI: 1.72–3.36) in favor of IVIG [39]. One trial comparing plasmapheresis and IVIG failed to find a difference in efficacy or cost effectiveness [40]. A recent Cochrane review failed to find sufficient evidence for cytotoxic drugs or interferons [41].
Paraprotein-mediated neuropathies account for up to 10% of neuropathies not attributable to another illness [42]. A Cochrane review of immunotherapy for IgM anti-myelin-associated glycoprotein (MAG) neuropathies reported that IVIG may produce some short-term benefit but that there was insufficient evidence to recommend any particular immunotherapy [43]. More recently, rituximab has shown efficacy as well [44]. There is currently limited data for the use of immunomodulatory therapies for IgG and IgA paraproteinemic neuropathies [45].
Multifocal motor neuropathy is a rare disorder characterized by demyelination of peripheral motor nerves and is frequently associated with serum IgM GM1 antibodies. A Cochrane review incorporated four trials with 34 patients. Improvement in strength occurred in 78% of IVIG treated patients vs. only 4% treated with placebo, and consequently IVIG has become the mainstay of treatment [46]. There is currently inadequate evidence for other immunomodulatory treatments [47].
Vasculitic neuropathy is a rare but potentially devastating form of neuropathy. While numerous studies of immunomodulatory therapies have been published there is insufficient evidence from RCTs to recommend any specific therapy [48].
Myasthenia Gravis (MG)
MG is characterized by waxing and waning exertion-evoked weakness with a predilection for the extra-ocular and bulbar musculature. The lifetime risk for mechanical ventilation is 10–20% [49]. Mortality due to complications of MG is estimated at 11–14% [50].
MG is caused by acetylcholine receptor (AChR) antibodies in 85% of patients [51, 52]. These antibodies bind to the AChR, promoting complement fixation and lysis of the postsynaptic membrane, which in turn impairs the transmission of signals through the neuromuscular junction [51, 53]. Of the 15% of patients without AChR antibodies, 41% have antibodies to muscle specific kinase (MUSK), a tyrosine kinase involved with formation of the neuromuscular junction itself [25, 54].
Lack of data from RCTs limits standardization of treatment for MG; however, the fundamental treatment approach at most institutions is similar. Acute exacerbations are controlled via therapies with short- or medium-term effects. These include acetylcholinesterase inhibitors, corticosteroids, plasmapheresis, and IVIG. For patients with multiple or severe relapses, long-term immunosuppression is pursued. Typical agents include azathioprine, methotrexate, and cyclophosphamide. In the outpatient setting, as patients stabilize, the shorter acting medications are titrated off first and the long-term immunosuppressants continued. The use of cholinesterase inhibitors in patients with respiratory failure is declining due to the side effect of increased salivary and tracheobronchial secretions as well as variable pharmacokinetics in critically ill patients [55].
Corticosteroids likely hasten motor recovery in acute MG [56] but, in about 50% of patients, their use is associated with a transient worsening of weakness (beginning at an average of 5 days after initiation, range 1–21 days) [57]. This is thought to be related in part to lysis of lymphocytes with release of immunoglobulin and in part due to direct effects on the neuromuscular junction [57]. Multiple treatment regimens exist. A common regimen is oral prednisone at 10–100 mg/d (often 60 or 80 mg/d) or 100 mg QOD followed by a slow taper over weeks to months. An alternative is high dose pulse therapy (2 g IV methylprednisolone repeated every 5 days for a maximum of three doses) [57]. To mitigate transient worsening, many centers concurrently treat with either IVIG or plasmapheresis [58].
Though there are no randomized placebo-controlled trials, numerous observational studies support the use of plasmapheresis in the management of myasthenic crisis. The largest of these included 70 patients and a total of 156 cycles of plasmapheresis. Improvement was seen in 70% [59]. A Cochrane review identified seven studies with a total of 316 patients with a similar aggregate improvement of 76.4%. A RCT comparing daily exchanges to QOD exchanges reported that daily exchanges were superior [60]. A typical protocol includes 5–6 1.0–1.5 volume exchanges daily (usually 2–4 l each) [61]. The duration of effect of a single course of plasmapheresis is 1–2 months and the efficacy may be improved by concomitant administration of corticosteroids or cyclophosphamide [62].
IVIG is an alternative to plasmapheresis for acute exacerbations of MG. A single randomized trial found a substantial benefit for IVIG over placebo, particularly in patients with severe disease [63]. Two RCTs have been performed comparing IVIG with plasmapheresis. The first failed to find a difference between the two groups [64]. The second compared three 1.5 plasma volume exchanges with IVIG for either 3 or 5 days. No difference was noted between the three groups though a statistically insignificant trend to more rapid improvement was observed in the plasmapheresis-treated group (9 vs. 15 days, P = 0.14) [64]. A recent Cochrane review concluded that there is evidence for the efficacy of IVIG in MG exacerbation but that there is currently not enough evidence to establish the relative efficacy of IVIG versus plasmapheresis [65]. A typical regimen for IVIG is 0.4 mg/kg/day for 3–5 days.
Recently, semi-selective adsorption of AChR antibodies has been performed, although the technique is not yet in routine clinical use. One trial enrolling all patients that were AChR antibody positive (20 patients), had a response rate of 55% [66].
Acute Demyelinating Encephalomyelitis (ADEM)/Hyperacute Multiple Sclerosis (MS)
ADEM and MS are autoimmune inflammatory disorders of the central nervous system (CNS). MS is thought to be primarily a Th1-mediated process. The early course in most cases is defined by acute episodes of inflammation with breakdown of the blood–brain barrier. On occasion, the presenting attack is extensive enough to result in severe alteration in level of consciousness and to require critical care. This is variously termed hyperacute, fulminant, or malignant MS. Later stages of the disease are felt to be primarily driven by neurodegeneration, and as such, are less responsive to immunomodulatory therapy [67]. In contrast ADEM is classically a monophasic autoimmune disease of the CNS following viral infection, infection with atypical bacteria, or vaccination [68]. Subtypes include acute hemorrhagic leukoencephalitis (AHL), acute hemorrhagic encephalomyelitis (AHEM), acute necrotizing hemorrhagic leukoencephalitis (ANHLE), and encephalomyeloradiculopathy.
The best studied therapeutic modalities for acute MS exacerbations are corticosteroids and ACTH. A Cochrane review reported six trials with 377 participants, 140 treated with corticosteroids, and 237 with ACTH. Methylprednisolone (500–1,000 mg) was administered daily for 3–5 days with or without a 10–12 day taper. The meta-analysis concluded that both regimens demonstrated a protective effect against further worsening or failure to improve over 5 weeks (OR 0.37, 95% CI: 0.24–0.57). There was a non-significant trend toward greater efficacy of corticosteroid versus ACTH and IV corticosteroid versus oral regimens. No difference was found between 5 and 15 days of treatment [4]. Most experts in MS believe that corticosteroids accelerate recovery from exacerbations but do not otherwise change the natural history of the disease, although a recent small randomized trial enrolling 126 patients found a decrease in relapse rate in patients treated with both interferon beta-1a and methylprednisolone (200 mg/d for 5 days every 4 weeks) compared to those treated with interferon beta-1a alone [69].
In ADEM, corticosteroids are considered first-line therapy. Although there are no RCTs to support their use, such a trial is unlikely given very high response rates in most case series [4]. For example, one adult case series reported improvement in 32 of 39 patients [70]. Delayed development of deep gray matter or brainstem lesions has been associated with corticosteroid failure [71]. The only comparison of different treatment regimens was a retrospective review comparing nonconsecutive but clinically similar patients treated with IV methylprednisolone 30 mg/kg/day × 3–5 days with IV dexamethasone 1 mg/kg/day × 10 days. The final Expanded Disability Status Scale (EDSS) score in the methylprednisolone group was 1 (no disability) versus 3 (mild to moderate disability) in the dexamethasone group (P = 0.029) [72].
A typical treatment regimen for both ADEM and severe MS exacerbations is 1 g of methylprednisolone IV daily for 3–5 days. There is no evidence to support a corticosteroid taper after initial treatment.
Plasmapheresis is utilized in the acute treatment of both MS and ADEM [73–78]. It has been studied in two placebo-controlled trials to date. The first examined plasmapheresis as an adjunctive therapy in patients being treated with ACTH and cyclophosphamide during an acute MS exacerbation. Significant benefit was found at 2 weeks and at 12 months, and the average time to recovery to pre-attack disability was 4 vs. 13 weeks [79]. A second evaluated plasmapheresis in 22 patients with CNS demyelination of any type who had failed corticosteroids. A moderate or greater improvement was seen in 42.1% in the active treatment group vs. 5.9% of controls [80]. Favorable responses were more likely in patients with preserved or brisk reflexes. Seventy-five percent of responders do so within three exchanges or 5 days of treatment [81]. A typical treatment regimen is single volume plasmapheresis daily or every other day for 3–5 exchanges, though prolonged therapy (up to 20 exchanges) has been reported [73–78].
IVIG therapy has shown promising results in uncontrolled investigations in ADEM but does not appear to be effective in MS [82–91]. A prospective non-randomized study in a cohort with ADEM found that involvement of both the central and peripheral nervous system (particularly in severe cases) predicted corticosteroid failure and benefit from IVIG [92]. A second non-randomized prospective study confirmed peripheral nervous system involvement as a predictor of steroid-refractory disease. An IVIG response rate of 53% in corticosteroid non-responders was reported; this response began within 5 days and reached a maximum at 3 weeks in most patients [93]. IVIG as an adjunct to methylprednisolone has been evaluated in two randomized placebo-controlled trials for acute MS exacerbations enrolling a total of 95 patients with negative results [71, 94]. Thus, IVIG may be considered in the treatment of ADEM, especially with peripheral nerve involvement, but does not appear to have a role in treatment of MS. A typical treatment regimen for ADEM consists of 0.4 g/kg/day daily or every other day for 3–5 doses, though single infusions of 2 g/kg have also been reported [82–91, 95].
Approximately 5% of patients with MS have a malignant phenotype characterized by frequent relapse and rapid neurologic deterioration [96]. Many respond to plasmapheresis but some will not. For those that remain refractory, more intense immunosuppression needs to be considered. Currently mitoxantrone is the only FDA approved immunosuppressive for multiple sclerosis. The largest placebo controlled trial of mitoxantrone in rapidly worsening relapsing remitting or secondary progressive MS (n = 194 patients) reported significant benefits with mitoxantrone treatment in terms of change in expanded disability status scale, change in ambulation, adjusted total number of treated relapses, and time to first treated relapse [97]. A recent Cochrane review evaluated four trials with a total of 270 patients. Post-hoc analysis demonstrated a RR of progression at one year of 0.19 (0.06–0.64). 93/270 patients had information available regarding relapse at either 6 months or one year. 68.7% of treated vs. 28.8% of placebo patients were relapse free at 6 months or one year (OR 5.4, 95% CI: 2.2–13.1; P = 0.0002). The study concluded that mitoxantrone was moderately effective at reducing progression and relapse [98]. Mitoxantrone carries a significant risk of cardiotoxicity and for this reason it has been suggested only as a rescue therapy for those patients with relapsing remitting disease with frequent and disabling exacerbations or in patients with rapidly deteriorating secondary progressive disease [99]. Echocardiography should be performed prior to the initiation of therapy and periodically thereafter. Typical dosing consists of an induction phase of 12 mg/m2 monthly for 3 months then every 3 months for two years [99].
Cyclophosphamide is another potential therapy [100] for refractory or fulminant MS, although trials with this agent have yielded conflicting results [101–103]. One of the largest long-term observational studies in severe relapsing-remitting disease included 110 patients and demonstrated a 75% reduction in relapse rate in 70% of patients [104]. A number of smaller case series in malignant or fulminant MS report improvement or stability in up to 75% of treated patients [105–108]. Factors which have been associated with a response to therapy include a rapidly progressive course, gadolinium positive lesions on MRI, young age, <2 years of a progressive course, and absence of primary progressive disease [67]. While several treatment regimens exist, a commonly used protocol involves cyclophosphamide induction with 600 mg/m2 given on days 1, 2, 4, 6, and 8 with 1 g IV methylprednisolone given on days 1–8 [67]. Experience with other acute chemotherapeutic agents is limited and the role of humanized monoclonal antibodies such as natalizumab, alemtuzumab, daclizumab, and rituximab in acute disease remains to be established [100].
For the most severe and treatment-resistant cases a final treatment option is autologous hematopoietic stem cell transplantation (AHSCT) (about 250 reports in MS patients worldwide) [109]. The majority of patients treated with AHSCT have had aggressive secondary progressive disease [109]. In four clinical trials, 73–85.7% of patients were free of progression at 3 years and overall mortality rates were 0–8.2% [110–112].
Meningitis and Encephalitis
Bacterial meningitis is a frequent cause of admission to ICUs and carries a mortality of 10–30% [113]. One meta-analysis found acute corticosteroid treatment initiated concurrently with antibiotic treatment is associated with a reduction in mortality rate from 22 to 12%; the authors recommended dexamethasone 0.6 mg/kg/day IV x 4 days [113]. In children, studies have found conflicting results that are possibly related to patient location (developed/developing world) and accompanying co-morbid conditions (in particular HIV) [114, 115]. The current Infectious Diseases Society of America advocate dexamethasone 0.15 mg/kg IV q 6 h for 2–4 days for all adults with suspected bacterial meningitis given at the time of, or just prior to, the initiation of antibiotic therapy [116]. Since the publication of these guidelines one large RCT failed to show benefit in all adolescents and adults with suspected bacterial meningitis but found a reduction in risk of death at one month (RR 0.43, 95% CI: 0.20–0.94) in microbiologically proven disease, calling into question the timing of antibiotic administration [117]. Evidence for steroids in viral encephalitis is sparse, with one retrospective study finding that steroid administration was associated with better outcome [118]. While one large RCT is ongoing [119] the current Infectious Diseases Society of America guidelines do not endorse the use of corticosteroids in the treatment of viral encephalitis.
Primary Central Nervous System Vasculitis
Primary central nervous system vasculitis (PCNSV) is a rare disorder characterized by inflammation of cerebral blood vessels [120]. Subacute headache and cognitive change followed by infarcts within multiple vascular territories is the most common presentation. The largest case series (n = 101) found that cognitive impairment, infarction, focal neurologic deficit, and large vessel involvement were associated with increased mortality (HR 4.00, 4.39, 3.60, and 7.93, respectively) [120]. In contrast, prominent leptomeningeal enhancement has been associated with a more benign course [121]. Recently, it has been reported that angiographically negative PCNSV is more responsive to immunomodulatory treatment [122]. In children ischemic stroke at onset, large and medium vessel involvement, granulomatous angiitis, and delayed institution of therapy were markers of poor outcome [123]. In contrast neurocognitive dysfunction, multifocal MRI lesions, and distal stenosis were found to be a high risk profile in another pediatric case series. This study also reported that hemiparesis was associated with a non-progressive course [124].
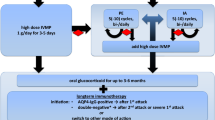
To date, no RCTs have been performed and no treatment guidelines exist [125]. The historical treatment for PCNSV—glucocorticoids and cyclophosphamide—remains the treatment of choice at many institutions [126–130]. With the increasing recognition of patients showing a more benign, non-progressive, corticosteroid responsive course, some authors have recently suggested reserving cyclophosphamide for cases which are either histologically proven or exhibit progression despite corticosteroids (approximately 19%) [130, 131]. While no guidelines exist, the most common treatment regimen is high dose methylprednisolone 1 g/day for 3–7 days followed by oral prednisone. Cyclophosphamide can be administered as a daily oral therapy (2–2.5 mg/kg/day) or more commonly, as pulse IV therapy 500–1,000 mg/m2 of body surface area every other week for 6 weeks then monthly [130]. Therapy is commonly continued for 12 months and has been reported to have a relapse rate of <10% [131, 132].
Two patients have been reported with PCNSV resistant to treatment with corticosteroids and cyclophosphamide therapy that were treated successfully with infliximab and etanercept [133]. There is not currently sufficient evidence to advocate for IVIG or plasmapheresis in PCNSV [131].
Corticosteroid Responsive Encephalitides
Acute autoimmune encephalitis can be categorized as paraneoplastic or non-paraneoplastic. Paraneoplastic encephalitis has been associated with antibodies to a number of antigens including Hu, Yo, Ri, Tr, amphiphysin, CV-2, Ma1, Ma2, mGLuR1, NMDA, and VGKC [134, 135]. Treatment of the underlying malignancy is the cornerstone of therapy and the few small uncontrolled clinical trials evaluating immunomodulatory treatment have shown mixed results. One prospective unblinded study evaluated plasmapheresis plus either cyclophosphamide or specific cancer chemotherapy in 20 patients. Overall, stabilization or modest improvement was seen in 50% without significant differences in efficacy between the treatment arms and no response was seen in patients with severe disability [136]. Several other series, however, have demonstrated substantially lower response rates with IVIG alone [137], the combination of plasmapheresis and cylophosphamide [138, 139], or the combination of IVIG, cyclophosphamide, and corticosteroids [140]. Despite a lack of clear evidence, some experts suggest consideration of plasmapheresis or IVIG, particularly in those with mild or moderate disability [141, 142]. Dosing regimens for plasmapheresis and IVIG are similar to those described for other diseases in this review.
The non-paraneoplastic autoimmune encephalitides are an increasingly recognized group of disorders. Three reasonably well-characterized subtypes are associated with anti-voltage gated potassium channel (VGKC) antibodies, anti-N-methyl-d-aspartate (NMDA) receptor antibodies, and anti-thyroid antibodies.
Both VGKC antibodies and NMDA antibodies are associated with limbic encephalitis characterized by seizures, neuropsychiatric disturbances, and memory loss and both may be paraneoplastic or non-paraneoplastic. Additionally, dyskinesias, autonomic instability, and central hypoventilation are common features of NMDA receptor encephalitis [143]. In uncontrolled clinical trials in patients with VGKC antibodies, dramatic responses to therapy have been noted with methylprednisolone, IVIG and less commonly plasmapheresis [144–147]. About 2/3 of patients with non-paraneoplastic NMDA antibody encephalitis recover with immunotherapy (plasmapheresis, methylprednisolone, IVIG) [139].
It has been suggested by several authors that VGKC-mediated encephalitis represents a more immunoresponsive etiology than other autoimmune encephalopathies [146]. While there are no formal recommendations on treatment of VGKC-mediated and NMDA receptor-associated encephalitis most reported cases have been treated with combinations of IVIG or plasmapheresis either alone or with up to 6 months of oral prednisone [143–145].
Hashimoto’s encephalitis (also referred to as corticosteroid responsive encephalopathy associated with autoimmune thyroiditis (SREAT) or non-vasculitic autoimmune meningoencephalitis (NAIM)) is an autoimmune encephalitis associated with anti-thyroid antibodies [148–151]. Corticosteroid therapy is so effective that some authors use steroid responsiveness as a requirement for the diagnosis [149]. Clinical response is typically seen 4–6 weeks from initiation of therapy [148]. Numerous corticosteroid treatment regimens have been reported, most commonly methylprednisolone 1 g IV daily for 3–5 days, with ongoing oral prednisone at 60–100 mg/day afterward for variable durations [152–159]. In the absence of a response to corticosteroids, a few case reports support the use of IVIG [160, 161] or plasmapheresis [162–164].
Status Epilepticus
The role of autoimmunity in epilepsy and status epilepticus remains unclear [165]. There is a growing body of literature describing possible pathologic antibodies in numerous epilepsy syndromes including antibodies to VGKC, TPO, thyroglobulin, GM1, GluR1, 3, and 4 as well as antiphospholipid antibodies [153, 165, 166]. In addition, the value of immunomodulatory therapies including ACTH, corticosteroids, and to a lesser extent plasmapheresis and IVIG in pediatric epilepsy syndromes such as West syndrome, Lennox-Gastaut, Landau-Kleffner, and Rasmussen encephalitis have been demonstrated by several studies and case series [167–172]. With regard to refractory status epilepticus specifically, there are occasional case reports documenting both treatment success and failure with plasmapheresis [173, 174]. Given the paucity of evidence, unless an autoimmune etiology is strongly suspected, immunomodulatory therapies should be reserved for refractory status epilepticus only in exceptional circumstances.
Controversies and Future Directions
A variety of clinical questions still remain. While high levels of evidence (Table 1) support the use of immunomodulatory therapies in GBS and MG, several details need to be addressed. For example, in GBS, the decision of whether to repeat a course IVIG or plasmapheresis is a common dilemma in patients in whom the initial response is not adequate. In MG, the exact role of corticosteroids (given the propensity for transient symptomatic worsening) has not been elucidated. For ADEM, while case series suggest a benefit for corticosteroids, IVIG, and plasmapheresis, as yet no RCTs have been performed to guide which combinations may be effective. In MS, corticosteroids appear to speed recovery but do not have a substantial disease-modifying effect and there is no consensus regarding severe or treatment refractory exacerbations. In encephalitis, some evidence suggests that patients with viral etiologies (particularly HSV) may benefit from acute high-dose corticosteroid therapy. For many of the other diseases discussed in this review, evidence is limited to small case series.
Over the longer-term, research is likely to focus on more specific therapies. The standard therapies—IVIG, plasmapheresis, and corticosteroids—are all non-specific in their actions. Focused immunomodulation using monoclonal antibodies with specific targets—for example rituximab (CD 20), nataluzimab (α-4-integrin), and a variety of anti-TNF-α antibodies—may ultimately lead to treatments with fewer side effects and increased efficacy. Finally, as the detailed pathogenesis of autoimmune neurological disease is elucidated further, disease-specific treatments will likely become available akin to the progress which has been made in immunoadsorption of circulating AChR antibodies in MG.
References
Broessner G, Helbok R, Lackner P, et al. Survival and long-term functional outcome in 1,155 consecutive neurocritical care patients. Crit Care Med. 2007;35:2025–30.
Gomes JA, Stevens RD, Lewin JJ III, Mirski MA, Bhardwaj A. Glucocorticoid therapy in neurologic critical care. Crit Care Med. 2005;33:1214–24.
Parker BJ, Bruce IN. High dose methylprednisolone therapy for the treatment of severe systemic lupus erythematosus. Lupus. 2007;16:387–93.
Filippini G, Brusaferri F, Sibley WA, et al. Corticosteroids or ACTH for acute exacerbations in multiple sclerosis. Cochrane Database Syst Rev. 2000;001331.
Lindberg C, Andersen O, Lefvert AK. Treatment of myasthenia gravis with methylprednisolone pulse: a double blind study. Acta Neurol Scand. 1998;97:370–3.
Lehmann HC, Hartung HP, Hetzel GR, Stuve O, Kieseier BC. Plasma exchange in neuroimmunological disorders: Part 1: Rationale and treatment of inflammatory central nervous system disorders. Arch Neurol. 2006;63:930–5.
Mokrzycki MH, Kaplan AA. Therapeutic plasma exchange: complications and management. Am J Kidney Dis. 1994;23:817–27.
Keller AJ, Urbaniak SJ. Intensive plasma exchange on the cell separator: effects on serum immunoglobulins and complement components. Br J Haematol. 1978;38:531–40.
Yeh JH, Chiu HC. Plasmapheresis-related hypotension. Artif Organs. 2000;24:705–9.
Gold R, Stangel M, Dalakas MC. Drug Insight: the use of intravenous immunoglobulin in neurology—therapeutic considerations and practical issues. Nat Clin Pract Neurol. 2007;3:36–44.
Kaveri SV, Lacroix-Desmazes S, Bayry J. The antiinflammatory IgG. N Engl J Med. 2008;359:307–9.
Kazatchkine MD, Kaveri SV. Immunomodulation of autoimmune and inflammatory diseases with intravenous immune globulin. N Engl J Med. 2001;345:747–55.
Paran D, Herishanu Y, Elkayam O, Shopin L, Ben-Ami R. Venous and arterial thrombosis following administration of intravenous immunoglobulins. [see comment]. Blood Coagul Fibrinol. 2005;16:313–8.
Ballow M. Safety of IGIV therapy and infusion-related adverse events. Immunol Res. 2007;38:122–32.
Orbach H, Katz U, Sherer Y, Shoenfeld Y. Intravenous immunoglobulin: adverse effects and safe administration. Clin Rev Allergy Immunol. 2005;29:173–84.
Asbury AK, Cornblath DR. Assessment of current diagnostic criteria for Guillain–Barre syndrome [see comment]. Ann Neurol. 1990;27:S21–4.
Hughes RA, Rees JH. Clinical and epidemiologic features of Guillain–Barre syndrome. J Infect Dis. 1997;176:S92–8.
Bosch PE, Smith BE. Disorders of peripheral nerves. In: Neurology in clinical practice. 5th ed. Philadelphia: Butterworth-Heinemann; 2004. p. 2336–2345.
Zochodne DW. Autonomic involvement in Guillain–Barre syndrome: a review. Muscle Nerve. 1994;17:1145–55.
Bernsen RA, de Jager AE, Schmitz PI, van der Meche FG. Residual physical outcome and daily living 3 to 6 years after Guillain–Barre syndrome. Neurology. 1999;53:409–10.
van Doorn PA, Ruts L, Jacobs BC. Clinical features, pathogenesis, and treatment of Guillain–Barre syndrome [see comment]. Lancet Neurol. 2008;7:939–50.
Willison HJ, Yuki N. Peripheral neuropathies and anti-glycolipid antibodies. Brain. 2002;125:2591–625.
Chowdhury D, Arora A. Axonal Guillain–Barre syndrome: a critical review. Acta Neurol Scand. 2001;103:267–77.
Hafer-Macko CE, Sheikh KA, Li CY, et al. Immune attack on the Schwann cell surface in acute inflammatory demyelinating polyneuropathy. Ann Neurol. 1996;39:625–35.
Hafer-Macko C, Hsieh ST, Li CY, et al. Acute motor axonal neuropathy: an antibody-mediated attack on axolemma. Ann Neurol. 1996;40:635–44.
The Guillain–Barre syndrome Study Group. Plasmapheresis and acute Guillain–Barre syndrome. Neurology. 1985;35:1096–104.
French Cooperative Group on Plasma Exchange in Guillain–Barre syndrome. Efficiency of plasma exchange in Guillain–Barre syndrome: role of replacement fluids. Ann Neurol. 1987;22:753–61.
Raphael JC, Chevret S, Hughes RA, Annane D. Plasma exchange for Guillain–Barre syndrome [update of Cochrane Database Syst Rev. 2001;(2):CD001798; PMID: 11406009]. Cochrane Database Syst Rev. 2002;001798.
van der Meche FG, Schmitz PI. A randomized trial comparing intravenous immune globulin and plasma exchange in Guillain–Barre syndrome. Dutch Guillain–Barre Study Group. N Engl J Med. 1992;326:1123–9.
Hughes RA, Raphael JC, Swan AV, van Doorn PA. Intravenous immunoglobulin for Guillain–Barre syndrome [update of Cochrane Database Syst Rev. 2004;(1):CD002063; PMID: 14973982]. Cochrane Database Syst Rev. 2006;002063.
Plasma Exchange/Sandoglobulin Guillain–Barre Syndrome Trial Group. Randomised trial of plasma exchange, intravenous immunoglobulin, and combined treatments in Guillain–Barre syndrome. Lancet. 1997;349:225–30.
Hughes RA, Swan AV, van Koningsveld R, van Doorn PA. Corticosteroids for Guillain–Barre syndrome [update of Cochrane Database Syst Rev. 2000;(3):CD001446; PMID: 10908498]. Cochrane Database Syst Rev. 2006;001446.
Hughes RA, Wijdicks EF, Barohn R, et al. Practice parameter: immunotherapy for Guillain–Barre syndrome: report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology. 2003;61:736–40.
Barohn RJ, Kissel JT, Warmolts JR, Mendell JR. Chronic inflammatory demyelinating polyradiculoneuropathy. Clinical characteristics, course, and recommendations for diagnostic criteria. Arch Neurol. 1989;46:878–84.
McCombe PA, Pollard JD, McLeod JG. Chronic inflammatory demyelinating polyradiculoneuropathy. A clinical and electrophysiological study of 92 cases. Brain. 1987;110:1617–30.
Dyck PJ, O’Brien PC, Oviatt KF, et al. Prednisone improves chronic inflammatory demyelinating polyradiculoneuropathy more than no treatment. Ann Neurol. 1982;11:136–41.
Dyck PJ, Daube J, O’Brien P, et al. Plasma exchange in chronic inflammatory demyelinating polyradiculoneuropathy. N Engl J Med. 1986;314:461–5.
Hahn AF, Bolton CF, Pillay N, et al. Plasma-exchange therapy in chronic inflammatory demyelinating polyneuropathy. A double-blind, sham-controlled, cross-over study. Brain. 1996;119:1055–66.
Eftimov F, Winer JB, Vermeulen M, de Haan R, van Schaik IN. Intravenous immunoglobulin for chronic inflammatory demyelinating polyradiculoneuropathy [update of Cochrane Database Syst Rev. 2002;(2):CD001797; PMID: 12076423]. Cochrane Database Syst Rev. 2009;001797.
Dyck PJ, Litchy WJ, Kratz KM, et al. A plasma exchange versus immune globulin infusion trial in chronic inflammatory demyelinating polyradiculoneuropathy. Ann Neurol. 1994;36:838–45.
Hughes RA, Swan AV, van Doorn PA. Cytotoxic drugs and interferons for chronic inflammatory demyelinating polyradiculoneuropathy [update of Cochrane Database Syst Rev. 2003;(1):CD003280; PMID: 12535467]. Cochrane Database Syst Rev. 2004;003280.
Kelly JJ Jr, Kyle RA, O’Brien PC, Dyck PJ. Prevalence of monoclonal protein in peripheral neuropathy. Neurology. 1981;31:1480–3.
Lunn MP, Nobile-Orazio E. Immunotherapy for IgM anti-myelin-associated glycoprotein paraprotein-associated peripheral neuropathies [update of Cochrane Database Syst Rev. 2003;(1):CD002827; PMID: 12535440]. Cochrane Database Syst Rev. 2006;002827.
Dalakas MC, Rakocevic G, Salajegheh M, et al. Placebo-controlled trial of rituximab in IgM anti-myelin-associated glycoprotein antibody demyelinating neuropathy. Ann Neurol. 2009;65:286–93.
Allen D, Lunn MP, Niermeijer J, Nobile-Orazio E. Treatment for IgG and IgA paraproteinaemic neuropathy. Cochrane Database Syst Rev. 2007;005376.
van Schaik IN, van den Berg LH, de Haan R, Vermeulen M. Intravenous immunoglobulin for multifocal motor neuropathy. Cochrane Database Syst Rev. 2005;004429.
Umapathi T, Hughes RA, Nobile-Orazio E, Leger JM. Immunosuppressant and immunomodulatory treatments for multifocal motor neuropathy [update of Cochrane Database Syst Rev. 2005;(3):CD003217; PMID: 16034892]. Cochrane Database Syst Rev. 2009;003217.
Vrancken AF, Hughes RA, Said G, Wokke JH, Notermans NC. Immunosuppressive treatment for non-systemic vasculitic neuropathy. Cochrane Database Syst Rev. 2007;006050.
Ahmed S, Kirmani JF, Janjua N, et al. An update on myasthenic crisis. Curr Treat Opt Neurol. 2005;7:129–41.
Grob D, Brunner N, Namba T, Pagala M. Lifetime course of myasthenia gravis. Muscle Nerve. 2008;37:141–9.
Lindstrom JM. Acetylcholine receptors and myasthenia. Muscle Nerve. 2000;23:453–77.
Vincent A, Palace J, Hilton-Jones D. Myasthenia gravis. Lancet. 2001;357:2122–8.
Tzartos S, Hochschwender S, Vasquez P, Lindstrom J. Passive transfer of experimental autoimmune myasthenia gravis by monoclonal antibodies to the main immunogenic region of the acetylcholine receptor. J Neuroimmunol. 1987;15:185–94.
McConville J, Farrugia ME, Beeson D, et al. Detection and characterization of MuSK antibodies in seronegative myasthenia gravis. Ann Neurol. 2004;55:580–4.
Berrouschot J, Baumann I, Kalischewski P, Sterker M, Schneider D. Therapy of myasthenic crisis [see comment]. Crit Care Med. 1997;25:1228–35.
Schneider-Gold C, Gajdos P, Toyka KV, Hohlfeld RR. Corticosteroids for myasthenia gravis. Cochrane Database Syst Rev. 2005;002828.
Bromberg MB, Carter O. Corticosteroid use in the treatment of neuromuscular disorders: empirical and evidence-based data. Muscle Nerve. 2004;30:20–37.
Jani-Acsadi A, Lisak RP. Myasthenic crisis: guidelines for prevention and treatment. J Neurol Sci. 2007;261:127–33.
Antozzi C, Gemma M, Regi B, et al. A short plasma exchange protocol is effective in severe myasthenia gravis. J Neurol. 1991;238:103–7.
Yeh JH, Chiu HC. Plasmapheresis in myasthenia gravis. A comparative study of daily versus alternately daily schedule. Acta Neurol Scand. 1999;99:147–51.
Weinstein R. Therapeutic apheresis in neurological disorders. J Clin Apheresis. 2000;15:74–128.
Richman DP, Agius MA. Treatment of autoimmune myasthenia gravis [see comment]. Neurology. 2003;61:1652–61.
Zinman L, Ng E, Bril V. IV immunoglobulin in patients with myasthenia gravis: a randomized controlled trial. Neurology. 2007;68:837–41.
Gajdos P, Chevret S, Clair B, Tranchant C, Chastang C. Clinical trial of plasma exchange and high-dose intravenous immunoglobulin in myasthenia gravis. Myasthenia Gravis Clinical Study Group. Ann Neurol. 1997;41:789–96.
Gajdos P, Chevret S, Toyka K. Intravenous immunoglobulin for myasthenia gravis [update in Cochrane Database Syst Rev. 2008;(1):CD002277; PMID: 18254004][update of Cochrane Database Syst Rev. 2003;(2):CD002277; PMID: 12804431]. Cochrane Database Syst Rev. 2006;002277.
Shibuya N, Sato T, Osame M, Takegami T, Doi S, Kawanami S. Immunoadsorption therapy for myasthenia gravis. J Neurol Neurosurg Psychiatry. 1994;57:578–81.
Weiner HL, Cohen JA. Treatment of multiple sclerosis with cyclophosphamide: critical review of clinical and immunologic effects. Multiple Sclerosis. 2002;8:142–54.
Tenembaum S, Chitnis T, Ness J, Hahn JS, International Pediatric MS Study G. Acute disseminated encephalomyelitis. Neurology. 2007;68:S23–36.
Sorensen PS, Mellgren SI, Svenningsson A, et al. NORdic trial of oral Methylprednisolone as add-on therapy to Interferon beta-1a for treatment of relapsing-remitting Multiple Sclerosis (NORMIMS study): a randomised, placebo-controlled trial. Lancet Neurol. 2009;8:519–29.
Schwarz S, Mohr A, Knauth M, Wildemann B, Storch-Hagenlocher B. Acute disseminated encephalomyelitis: a follow-up study of 40 adult patients. Neurology. 2001;56:1313–8.
Sorensen PS, Haas J, Sellebjerg F, Olsson T, Ravnborg M, TARIMS Study G. IV immunoglobulins as add-on treatment to methylprednisolone for acute relapses in MS. Neurology. 2004;63:2028–33.
Tenembaum S, Chamoles N, Fejerman N. Acute disseminated encephalomyelitis: a long-term follow-up study of 84 pediatric patients. Neurology. 2002;59:1224–31.
Jacquerye P, Ossemann M, Laloux P, Dive A, De Coene B. Acute fulminant multiple sclerosis and plasma exchange. Eur Neurol. 1999;41:174–5.
Mao-Draayer Y, Braff S, Pendlebury W, Panitch H. Treatment of steroid-unresponsive tumefactive demyelinating disease with plasma exchange. Neurology. 2002;59:1074–7.
Rodriguez M, Karnes WE, Bartleson JD, Pineda AA. Plasmapheresis in acute episodes of fulminant CNS inflammatory demyelination. Neurology. 1993;43:1100–4.
RamachandranNair R, Parameswaran M, Girija AS. Acute disseminated encephalomyelitis treated with plasmapheresis. Singapore Med J. 2005;46:561–3.
Kanter DS, Horensky D, Sperling RA, Kaplan JD, Malachowski ME, Churchill WH Jr. Plasmapheresis in fulminant acute disseminated encephalomyelitis. Neurology. 1995;45:824–7.
Ryan LJ, Bowman R, Zantek ND, et al. Use of therapeutic plasma exchange in the management of acute hemorrhagic leukoencephalitis: a case report and review of the literature. Transfusion. 2007;47:981–6.
Weiner HL, Dau PC, Khatri BO, et al. Double-blind study of true vs. sham plasma exchange in patients treated with immunosuppression for acute attacks of multiple sclerosis. Neurology. 1989;39:1143–9.
Weinshenker BG, O’Brien PC, Petterson TM, et al. A randomized trial of plasma exchange in acute central nervous system inflammatory demyelinating disease. Ann Neurol. 1999;46:878–86.
Keegan M, Pineda AA, McClelland RL, Darby CH, Rodriguez M, Weinshenker BG. Plasma exchange for severe attacks of CNS demyelination: predictors of response. Neurology. 2002;58:143–6.
Nishikawa M, Ichiyama T, Hayashi T, Ouchi K, Furukawa S. Intravenous immunoglobulin therapy in acute disseminated encephalomyelitis. Pediatr Neurol. 1999;21:583–6.
Kleiman M, Brunquell P. Acute disseminated encephalomyelitis: response to intravenous immunoglobulin. J Child Neurol. 1995;10:481–3.
Pradhan S, Gupta RP, Shashank S, Pandey N. Intravenous immunoglobulin therapy in acute disseminated encephalomyelitis. J Neurol Sci. 1999;165:56–61.
Hahn JS, Siegler DJ, Enzmann D. Intravenous gammaglobulin therapy in recurrent acute disseminated encephalomyelitis. Neurology. 1996;46:1173–4.
Sahlas DJ, Miller SP, Guerin M, Veilleux M, Francis G. Treatment of acute disseminated encephalomyelitis with intravenous immunoglobulin. Neurology. 2000;54:1370–2.
Finsterer J, Grass R, Stollberger C, Mamoli B. Immunoglobulins in acute, parainfectious, disseminated encephalo-myelitis. Clin Neuropharmacol. 1998;21:258–61.
Markus R, Brew BJ, Turner J, Pell M. Successful outcome with aggressive treatment of acute haemorrhagic leukoencephalitis. J Neurol Neurosurg Psychiatry. 1997;63:551.
Marchioni E, Marinou-Aktipi K, Uggetti C, et al. Effectiveness of intravenous immunoglobulin treatment in adult patients with steroid-resistant monophasic or recurrent acute disseminated encephalomyelitis. J Neurol. 2002;249:100–4.
Fu DC, Montgomery JR. High-dose, rapid-infusion IVIG in postvaccination acute disseminated encephalomyelitis. Neurology. 2008;71:294–5.
Lu RP, Keilson G. Combination regimen of methylprednisolone, IV immunoglobulin, and plasmapheresis early in the treatment of acute disseminated encephalomyelitis. J Clin Apheresis. 2006;21:260–5.
Shahar E, Andraus J, Savitzki D, Pilar G, Zelnik N. Outcome of severe encephalomyelitis in children: effect of high-dose methylprednisolone and immunoglobulins. J Child Neurol. 2002;17:810–4.
Ravaglia S, Piccolo G, Ceroni M, et al. Severe steroid-resistant post-infectious encephalomyelitis: general features and effects of IVIg. J Neurol. 2007;254:1518–23.
Visser LH, Beekman R, Tijssen CC, et al. A randomized, double-blind, placebo-controlled pilot study of i.v. immune globulins in combination with i.v. methylprednisolone in the treatment of relapses in patients with MS. Multiple Sclerosis. 2004;10:89–91.
Kanaheswari Y, Baizura J, Paeds M, Zulfiqar A. Intravenous immunoglobulin in the treatment of acute disseminated encephalomyelitis. Med J Malaysia. 2004;59:103–7.
Kimiskidis V, Sakellari I, Tsimourtou V, et al. Autologous stem-cell transplantation in malignant multiple sclerosis: a case with a favorable long-term outcome. Multiple Sclerosis. 2008;14:278–83.
Hartung HP, Gonsette R, Konig N, Kwiecinski H, Guseo A, Morrissey SP, et al. Mitoxantrone in progressive multiple sclerosis: a placebo-controlled, double-blind, randomised, multicentre trial. Lancet. 2002;360:2018–25.
Martinelli Boneschi F, Rovaris M, Capra R, Comi G. Mitoxantrone for multiple sclerosis. Cochrane Database Syst Rev. 2005;002127.
Gonsette RE. Mitoxantrone in progressive multiple sclerosis: when and how to treat? J Neurol Sci. 2003;206:203–8.
Boster A, Edan G, Frohman E, Javed A, Stuve O, Tselis A, et al. Multiple Sclerosis Clinical Research Center, Department of Neurology, Wayne State University School of Medicine. Intense immunosuppression in patients with rapidly worsening multiple sclerosis: treatment guidelines for the clinician. Lancet Neurol. 2008;7:173–83.
Hauser SL, Dawson DM, Lehrich JR, et al. Intensive immunosuppression in progressive multiple sclerosis. A randomized, three-arm study of high-dose intravenous cyclophosphamide, plasma exchange, and ACTH. N Engl J Med. 1983;308:173–80.
Likosky WH, Fireman B, Elmore R, et al. Intense immunosuppression in chronic progressive multiple sclerosis: the Kaiser study. Journal of Neurol Neurosurg Psychiatry. 1991;54:1055–60.
The Canadian Cooperative Multiple Sclerosis Study Group. The Canadian cooperative trial of cyclophosphamide and plasma exchange in progressive multiple sclerosis. Lancet. 1991;337:441–6.
Gonsette RE, Demonty L, Delmotte P. Intensive immunosuppression with cyclophosphamide in multiple sclerosis. Follow up of 110 patients for 2–6 years. J Neurol. 1977;214:173–81.
Weinstock-Guttman B, Kinkel RP, Cohen JA, et al. Treatment of fulminant multiple sclerosis with intravenous cyclophosphamide. Neurologist. 1997;3:178–85.
Khan OA, Zvartau-Hind M, Caon C, et al. Effect of monthly intravenous cyclophosphamide in rapidly deteriorating multiple sclerosis patients resistant to conventional therapy. Multiple Sclerosis. 2001;7:185–8.
Patti F, Cataldi ML, Nicoletti F, Reggio E, Nicoletti A, Reggio A. Combination of cyclophosphamide and interferon-beta halts progression in patients with rapidly transitional multiple sclerosis. J Neurol Neurosurg Psychiatry. 2001;71:404–7.
Gladstone DE, Zamkoff KW, Krupp L, et al. High-dose cyclophosphamide for moderate to severe refractory multiple sclerosis. Arch Neurol. 2006;63:1388–93.
Blanco Y, Saiz A, Carreras E, Graus F. Autologous haematopoietic-stem-cell transplantation for multiple sclerosis. Lancet Neurol. 2005;4:54–63.
Fassas A, Anagnostopoulos A, Kazis A, et al. Autologous stem cell transplantation in progressive multiple sclerosis—an interim analysis of efficacy. J Clin Immunol. 2000;20:24–30.
Fassas A, Passweg JR, Anagnostopoulos A, Kazis A, Kozak T, Havrdova E, et al. Autoimmune Disease Working Party of the EBMT (European Group for Blood and Marrow Transplantation). Hematopoietic stem cell transplantation for multiple sclerosis. A retrospective multicenter study. J Neurol. 2002;249:1088–97.
Saiz A, Blanco Y, Carreras E, et al. Clinical and MRI outcome after autologous hematopoietic stem cell transplantation in MS. Neurology. 2004;62:282–4.
van de Beek D, de Gans J, McIntyre P, Prasad K. Corticosteroids for acute bacterial meningitis [see comment] [update of Cochrane Database Syst Rev. 2003;(3):CD004405; PMID: 12918010]. Cochrane Database Syst Rev. 2007;004405.
Ozen M, Kanra G, Kara A, et al. Long-term beneficial effects of dexamethasone on intellectual and neuropsychological outcome of children with pneumococcal meningitis. Scand J Infect Dis. 2006;38:104–9.
Scarborough M, Gordon SB, Whitty CJ, et al. Corticosteroids for bacterial meningitis in adults in sub-Saharan Africa. N Engl J Med. 2007;357:2441–50.
Tunkel AR, Hartman BJ, Kaplan SL, et al. Practice guidelines for the management of bacterial meningitis. Clin Infect Dis. 2004;39:1267–84.
Nguyen TH, Tran TH, Thwaites G, et al. Dexamethasone in Vietnamese adolescents and adults with bacterial meningitis. N Engl J Med. 2007;357:2431–40.
Kamei S, Sekizawa T, Shiota H, et al. Evaluation of combination therapy using aciclovir and corticosteroid in adult patients with herpes simplex virus encephalitis. J Neurol Neurosurg Psychiatry. 2005;76:1544–9.
Martinez-Torres F, Menon S, Pritsch M, et al. Protocol for German trial of Acyclovir and corticosteroids in Herpes-simplex-virus-encephalitis (GACHE): a multicenter, multinational, randomized, double-blind, placebo-controlled German, Austrian and Dutch trial [ISRCTN45122933]. BMC Neurol. 2008;8:40.
Salvarani C, Brown RD Jr, Calamia KT, et al. Primary central nervous system vasculitis: analysis of 101 patients. Ann Neurol. 2007;62:442–51.
Salvarani C, Brown RD Jr, Calamia KT, et al. Primary central nervous system vasculitis with prominent leptomeningeal enhancement: a subset with a benign outcome. Arthritis Rheumatism. 2008;58:595–603.
Salvarani C, Brown RD Jr, Calamia KT, et al. Angiography-negative primary central nervous system vasculitis: a syndrome involving small cerebral vessels. Medicine. 2008;87:264–71.
Lanthier S, Lortie A, Michaud J, Laxer R, Jay V, deVeber G. Isolated angiitis of the CNS in children. Neurology. 2001;56:837–42.
Benseler SM, Silverman E, Aviv RI, et al. Primary central nervous system vasculitis in children. Arthritis Rheumatism. 2006;54:1291–7.
Calabrese LH, Molloy ES, Singhal AB. Primary central nervous system vasculitis: progress and questions. Ann Neurol. 2007;62:430–2.
Cupps TR, Moore PM, Fauci AS. Isolated angiitis of the central nervous system. Prospective diagnostic and therapeutic experience. Am J Med. 1983;74:97–105.
Moore PM. Diagnosis and management of isolated angiitis of the central nervous system. Neurology. 1989;39:167–73.
Bitter KJ, Epstein LG, Melin-Aldana H, Curran JG, Miller ML. Cyclophosphamide treatment of primary angiitis of the central nervous system in children: report of 2 cases. J Rheumatol. 2006;33:2078–80.
Volcy M, Toro ME, Uribe CS, Toro G. Primary angiitis of the central nervous system: report of five biopsy-confirmed cases from Colombia. J Neurol Sci. 2004;227:85–9.
Siva A. Vasculitis of the nervous system. J Neurol. 2001;248:451–68.
Hajj-Ali RA, Ghamande S, Calabrese LH, Arroliga AC. Central nervous system vasculitis in the intensive care unit. Crit Care Clin. 2002;18:897–914.
Moore PM, Richardson B. Neurology of the vasculitides and connective tissue diseases. J Neurol Neurosurg Psychiatry. 1998;65:10–22.
Salvarani C, Brown RD Jr, Calamia KT, et al. Efficacy of tumor necrosis factor alpha blockade in primary central nervous system vasculitis resistant to immunosuppressive treatment. Arthritis Rheumatism. 2008;59:291–6.
Darnell RB, Posner JB. Paraneoplastic syndromes involving the nervous system. N Engl J Med. 2003;349:1543–54.
Dalmau J, Tuzun E, Wu HY, et al. Paraneoplastic anti-N-methyl-d-aspartate receptor encephalitis associated with ovarian teratoma. Ann Neurol. 2007;61:25–36.
Vernino S, O’Neill BP, Marks RS, O’Fallon JR, Kimmel DW. Immunomodulatory treatment trial for paraneoplastic neurological disorders. Neuro-oncology. 2004;6:55–62.
Uchuya M, Graus F, Vega F, Rene R, Delattre JY. Intravenous immunoglobulin treatment in paraneoplastic neurological syndromes with antineuronal autoantibodies. J Neurol Neurosurg Psychiatry. 1996;60:388–92.
Graus F, Vega F, Delattre JY, et al. Plasmapheresis and antineoplastic treatment in CNS paraneoplastic syndromes with antineuronal autoantibodies. Neurology. 1992;42:536–40.
Anderson NE, Rosenblum MK, Graus F, Wiley RG, Posner JB. Autoantibodies in paraneoplastic syndromes associated with small-cell lung cancer. Neurology. 1988;38:1391–8.
Keime-Guibert F, Graus F, Fleury A, et al. Treatment of paraneoplastic neurological syndromes with antineuronal antibodies (Anti-Hu, anti-Yo) with a combination of immunoglobulins, cyclophosphamide, and methylprednisolone. J Neurol Neurosurg Psychiatry. 2000;68:479–82.
David YB, Warner E, Levitan M, Sutton DM, Malkin MG, Dalmau JO. Autoimmune paraneoplastic cerebellar degeneration in ovarian carcinoma patients treated with plasmapheresis and immunoglobulin. A case report. Cancer. 1996;78:2153–6.
Ances BM, Vitaliani R, Taylor RA, et al. Treatment-responsive limbic encephalitis identified by neuropil antibodies: MRI and PET correlates. Brain. 2005;128:1764–77.
Dalmau J, Gleichman AJ, Hughes EG, et al. Anti-NMDA-receptor encephalitis: case series and analysis of the effects of antibodies. Lancet Neurol. 2008;7:1091–8.
Vincent A, Buckley C, Schott JM, et al. Potassium channel antibody-associated encephalopathy: a potentially immunotherapy-responsive form of limbic encephalitis. Brain. 2004;127:701–12.
Pozo-Rosich P, Clover L, Saiz A, Vincent A, Graus F. Voltage-gated potassium channel antibodies in limbic encephalitis. Ann Neurol. 2003;54:530–3.
Thieben MJ, Lennon VA, Boeve BF, Aksamit AJ, Keegan M, Vernino S. Potentially reversible autoimmune limbic encephalitis with neuronal potassium channel antibody. Neurology. 2004;62:1177–82.
Buckley C, Oger J, Clover L, et al. Potassium channel antibodies in two patients with reversible limbic encephalitis. Ann Neurol. 2001;50:73–8.
Kothbauer-Margreiter I, Sturzenegger M, Komor J, Baumgartner R, Hess CW. Encephalopathy associated with Hashimoto thyroiditis: diagnosis and treatment. J Neurol. 1996;243:585–93.
Castillo P, Woodruff B, Caselli R, et al. Steroid-responsive encephalopathy associated with autoimmune thyroiditis. Arch Neurol. 2006;63:197–202.
Josephs KA, Rubino FA, Dickson DW. Nonvasculitic autoimmune inflammatory meningoencephalitis. Neuropathology. 2004;24:149–52.
Chong JY, Rowland LP. What’s in a NAIM? Hashimoto encephalopathy, steroid-responsive encephalopathy associated with autoimmune thyroiditis, or nonvasculitic autoimmune meningoencephalitis?[comment]. Arch Neurol. 2006;63:175–6.
Mahad DJ, Staugaitis S, Ruggieri P, et al. Steroid-responsive encephalopathy associated with autoimmune thyroiditis and primary CNS demyelination. J Neurol Sci. 2005;228:3–5.
Ferlazzo E, Raffaele M, Mazzu I, Pisani F. Recurrent status epilepticus as the main feature of Hashimoto’s encephalopathy. Epilepsy Behav. 2006;8:328–30.
Vernino S, Geschwind M, Boeve B. Autoimmune encephalopathies. Neurologist. 2007;13:140–7.
Chen HC, Marsharani U. Hashimoto’s encephalopathy. South Med J. 2000;93:504–6.
Caselli RJ, Boeve BF, Scheithauer BW, O’Duffy JD, Hunder GG. Nonvasculitic autoimmune inflammatory meningoencephalitis (NAIM): a reversible form of encephalopathy. Neurology. 1999;53:1579–81.
Shaw PJ, Walls TJ, Newman PK, Cleland PG, Cartlidge NE. Hashimoto’s encephalopathy: a steroid-responsive disorder associated with high anti-thyroid antibody titers—report of 5 cases. Neurology. 1991;41:228–33.
Chong JY, Rowland LP, Utiger RD. Hashimoto encephalopathy: syndrome or myth? [see comment]. Arch Neurol. 2003;60:164–71.
Mocellin R, Walterfang M, Velakoulis D. Hashimoto’s encephalopathy: epidemiology, pathogenesis and management. CNS Drugs. 2007;21:799–811.
Jacob S, Rajabally YA. Hashimoto’s encephalopathy: steroid resistance and response to intravenous immunoglobulins. J Neurol Neurosurg Psychiatry. 2005;76:455–6.
Wirkowski E, Libman RB, Batash M. Hashimot’s encephalopathy—response to intravenous immunoglobulin. J Stroke Cerebrovasc Dis. 1998;7:265–6.
Boers PM, Colebatch JG. Hashimoto’s encephalopathy responding to plasmapheresis. J Neurol Neurosurg Psychiatry. 2001;70:132.
Hussain NS, Rumbaugh J, Kerr D, Nath A, Hillis AE. Effects of prednisone and plasma exchange on cognitive impairment in Hashimoto encephalopathy. Neurology. 2005;64:165–6.
Nagpal T, Pande S. Hashimoto’s encephalopathy: response to plasma exchange. Neurol India. 2004;52:245–7.
Palace J, Lang B. Epilepsy: an autoimmune disease? J Neurol Neurosurg Psychiatry. 2000;69:711–4.
McKnight K, Jiang Y, Hart Y, et al. Serum antibodies in epilepsy and seizure-associated disorders. Neurology. 2005;65:1730–6.
Verhelst H, Boon P, Buyse G, et al. Steroids in intractable childhood epilepsy: clinical experience and review of the literature. Seizure. 2005;14:412–21.
Sinclair DB. Prednisone therapy in pediatric epilepsy. Pediatr Neurol. 2003;28:194–8.
Okumura A, Tsuji T, Kato T, Natsume J, Negoro T, Watanabe K. ACTH therapy for generalized seizures other than spasms. Seizure. 2006;15:469–75.
Prasad AN, Stafstrom CF, Holmes GL. Alternative epilepsy therapies: the ketogenic diet, immunoglobulins, and steroids. Epilepsia. 1996;37:S81–95.
Bien CG, Granata T, Antozzi C, et al. Pathogenesis, diagnosis and treatment of Rasmussen encephalitis: a European consensus statement. Brain. 2005;128:454–71.
Andrews PI, Dichter MA, Berkovic SF, Newton MR, McNamara JO. Plasmapheresis in Rasmussen’s encephalitis. Neurology. 1996;46:242–6.
Lousa M, Sanchez-Alonso S, Rodriguez-Diaz R, Dalmau J. Status epilepticus with neuron-reactive serum antibodies: response to plasma exchange. Neurology. 2000;54:2163–5.
Nahab F, Heller A, Laroche SM. Focal cortical resection for complex partial status epilepticus due to a paraneoplastic encephalitis. Neurologist. 2008;14:56–9.
Mosca L, Appel LJ, Benjamin EJ, et al. Summary of the American Heart Association’s evidence-based guidelines for cardiovascular disease prevention in women. Arterioscl Thromb Vasc Biol. 2004;24:394–6.
Acknowledgments
Dr. Bhardwaj is supported in part by the U.S. Public Health Service National Institutes of Health Grant NS046379.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
McDaneld, L.M., Fields, J.D., Bourdette, D.N. et al. Immunomodulatory Therapies in Neurologic Critical Care. Neurocrit Care 12, 132–143 (2010). https://doi.org/10.1007/s12028-009-9274-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-009-9274-0