Abstract
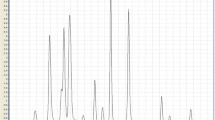
Pheniramine is an over-the-counter antihistamine drug. Its accessibility and low cost made it more popular among drug abusers in Pakistan. In this study, pheniramine was quantified in both conventional and alternative specimens of twenty chronic drug abusers, aged 16–50 years, who were positive for pheniramine in comprehensive toxicological screening for drugs by gas chromatography with mass spectral detection in positive electron impact mode. Pheniramine was extracted from biological specimens using solid phase extraction and liquid chromatography tandem mass spectrometry was employed for quantification. Chromatographic separation was carried out on a Poroshell120EC-18 (2.1 mm × 50 mm × 2.7 µm) column using water-acetonitrile in formic acid (0.1%) mobile phase in gradient elution mode with 500 μL/min flow rate. Positive electrospray ionization mode and multi-reaction monitoring with ion transitions m/z 241.3 → 195.8 and 167.1 for pheniramine and m/z m/z 247.6 → 173.1 for pheniramine-d6 were employed. The quantification method showed good linear ranges of 2–1000 ng/mL in blood, urine, and oral fluid; 2–1000 ng/mg in hair and 5–1000 ng/mg in nail with ≥ 0.985% coefficient of linearity. The retention time of pheniramine was 3.0 ± 0.1 min. The detection and lower quantification limits were 1 ng/mL and 2 ng/mL for blood, urine, oral fluid and hair whereas 2.5 ng/mg and 5 ng/mg for nail, respectively. Mean extraction recovery and ionization suppression ranged 86.3–95.1% and -4.6 to -14.4% in the studied matrices. Intra-day and inter-day precision were 4.1–9.3% and 2.8–11.2%, respectively. Pheniramine levels in specimens of drug abusers were 23–480 ng/mL in blood, 72–735 ng/mL in urine, 25–379 ng/mL in oral fluid, 10–170 ng/mg in hair and 8–86 ng/mg in nail specimens. Alternative specimens are of utmost significance in clinical and medico-legal cases. In this study, authors compared matrix-matched calibration curves to blood calibration curve and obtained results within ± 10%; thereby justifying the use of blood calibration curve for urine, oral fluid, hair, and nail specimens.



Similar content being viewed by others
Availability of data and material
All data generated or analyzed during this study are included in this article.
Abbreviations
- B:
-
Blood
- Cal:
-
Calibration curve
- U:
-
Urine
- OF:
-
Oral fluid
- H:
-
Hair
- N:
-
Nail
References
Baselt RC. Pheniramine. In: Disposition of toxic drugs and chemicals in man. 12th ed. Seal Beach, CA: Biomedical Publications; 2020. p. 1651–2.
Molina DK. Pheniramine. In Handbook of forensic toxicology for medical examiners. 2nd ed. Boca Raton, FL: CRC Press, Taylor & Francis Group; 2019. p. 178.
Querée EA, Dickson SJ, Missen AW, Pannell LK. Therapeutic and toxic levels of pheniramine in biological specimens. J Anal Toxicol. 1979;3(6):253–5.
Moffat AC, Osselton MD, Widdop B, Watts J. Pheniramine. In: Moffat AC, Osselton MD, Widdop B, Watts J, editors. Clarke’s analysis of drugs and poisons, vol. 2. 4th ed. Pharmaceutical Press. U.K.: London; 2011. p. 1902–3.
Gallardo E, Queiroz JA. The role of alternative specimens in toxicological analysis. Biomed Chromatogr. 2008;22(8):795–821.
Baumgartner WA, Hill VA, Blahd WH. Hair analysis for drugs of abuse. In: advances in analytical toxicology. Chicago, IL: Baselt RC edition. Year Book Medical; 1989. p. 298–329.
Pragst F, Balikova MA. State of the art in hair analysis for detection of drug and alcohol abuse. Clinicachimica acta. 2006;370:17–49.
Busardò FP, Gottardi M, Pacifici R, Varì MR, Tini A, Volpe AR, Pichini S. Nails analysis for drugs used in the context of chemsex: a pilot study. J Anal Toxicol. 2020;44(1):69–74.
Cingolani M, Scavella S, Mencarelli R, Mirtella D, Froldi R, Rodriguez D. Simultaneous detection and quantitation of morphine, 6-acetylmorphine, and cocaine in toenails: comparison with hair analysis. J Anal Toxicol. 2004;28(2):128–31.
Cappelle D, De Keukeleire S, Neels H, Been F, De Doncker M, Dom G, van Nuijs AL. Keratinous matrices for the assessment of drugs of abuse consumption: A correlation study between hair and nails. Drug Test Anal. 2018;10(7):1110–8.
Krumbiegel F, Hastedt M, Westendorf L, Niebel A, Methling M, Parr MK, Tsokos M. The use of nails as an alternative matrix for the long-term detection of previous drug intake: validation of sensitive UHPLC-MS/MS methods for the quantification of 76 substances and comparison of analytical results for drugs in nail and hair samples. Forensic Sci Med Pathol. 2016;12:416–34.
Spihler V. Drugs in saliva. In: Moffat AC, Osselton MD, Widdop B, Watts J, editors. Clarke’s analysis of drugs and poisons. 4th edition, Volume 1, Chapter 18. London, UK: Pharmaceutical Press; 2011. p. 308–22.
Lachenmeier K, Musshoff F, Madea B. Determination of opiates and cocaine in hair using automated enzyme immunoassay screening methodologies followed by gas chromatographic–mass spectrometric (GC-MS) confirmation. Forensic Sci Int. 2006;159:189–99.
Jones GR. Postmortem toxicology. In: Moffat AC, Osselton MD, Widdop B, Watts J, editors. Clarke’s analysis of drugs and poisons. 4th edition, Volume 1, Chapter 9. London: Pharmaceutical Press; 2011. p. 160–75.
Meyer MR, Maurer HH. Current status of hyphenated mass spectrometry in studies of the metabolism of drugs of abuse, including doping agents. Anal Bioanal Chem. 2012;402:195–208.
Scientific Working Group for Forensic Toxicology. Scientific Working Group for Forensic Toxicology (SWGTOX) standard practices for method validation in forensic toxicology. J Anal Toxicol. 2013;37(7):452–74.
Society of Hair Testing. Statement of the Society of Hair Testing concerning the examination of drugs in human hair. Forensic Sci Int. 1997;84:3–6.
Pragst F, Balikova MA. State of the art in hair analysis for detection of drug and alcohol abuse. Clin Chem. 2006;370(1–2):17–49.
Kuwayama K, Miyaguchi H, Iwata Y, Kanamori T, Tsujikawa K, Yamamuro T, Segawa H, Inoue H. Time-course measurements of drug concentrations in hair and toenails after single administrations of pharmaceutical products: Time-course measurements of drug concentrations in hair and toenails. Drug Test Anal. 2016;9(4):571–7.
Kronstrand R, Andersson MC, Ahlner J, Larson G. Incorporation of selegiline metabolites into hair after oral selegiline intake. J Anal Toxicol. 2001;25(7):594–601.
Kronstrand R, Förstberg-Peterson S, Kagedal B, Ahlner J, Larson G. Codeine concentration in hair after oral administration is dependent on melanin content. Clin Chem. 1999;45(9):1485–94.
Shen M, Chen H, Xiang P. Determination of opiates in human fingernail-comparison to hair. J Chromatogr B. 2014;967:84–9.
Barbosa J, Faria J, Carvalho F, Pedro M, Queirós O, Moreira R, Dinis-Oliveira RJ. Hair as an alternative matrix in bioanalysis. Bioanalysis. 2013;5(8):895–914.
Palmeri A, Pichini S, Pacifici R, Zuccaro P, Lopez A. Drugs in nails: physiology, pharmacokinetics and forensic toxicology. Clin Pharmacokinet. 2000;38:95–110.
Acknowledgements
Special thanks to faculty of pharmacy, University of Lahore and clinical and Forensic Toxicology Department, Chughtai Healthcare, Pakistan.
Funding
The submitted study did not receive any grant from the funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
All the authors participated equally regarding conceptualization, literature search, analysis, interpretation of results, writing and paper revising.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The permission was obtained from respondents, participating in this study on an individual consent form. All the participants were also informed about the significance and purpose of the study. Furthermore, the study was also approved by the ethics committee of the University of Lahore. All the procedures performed in the study were in accordance with the ethical standards of the University of Lahore, Pakistan.
Consent for publication
Consent has been obtained from all the participants regarding the publication of this study.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Makhdoom, H.S., Abid, A.I., Mujahid, M. et al. Assessment of pheniramine in alternative biological matrices by liquid chromatography tandem mass spectrometry. Forensic Sci Med Pathol (2024). https://doi.org/10.1007/s12024-024-00795-7
Accepted:
Published:
DOI: https://doi.org/10.1007/s12024-024-00795-7