Abstract
Purpose
Primary Hyperparathyroidism (PHPT) is associated with catabolic effects at both trabecular and cortical bone. Mechanical loading is one of the most important natural anabolic stimuli for bone at all ages. The present study was designed to assess the impact of PHPT on vBMD and bone geometry using peripheral quantitative computed tomography (pQCT) at the radius and tibia, sites with similar structural characteristics, but subject to different loading conditions.
Methods
We evaluated the impact of PHPT on bone, by comparing the z-scores of volumetric Bone Mineral Density (vBMD) and indices of bone geometry simultaneously at the tibia and the radius by pQCT, skeletal sites with similar structure, but subject to different loading conditions. Forty-one postmenopausal women with PHPT and 79 controls, comprised the study group.
Results
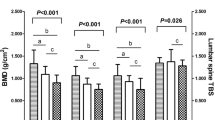
At both trabecular and cortical sites, vBMD and bone geometry indices were significantly lower in patients compared with controls. In patients with PHPT, apart from a lower z-score for total vBMD (p = 0.01) at the radius, there was no other difference between the radius and the tibia at the trabecular sites. On the contrary, at cortical sites, the z-scores of cortical bone mineral content (p = 0.02), cortical vBMD (p = 0.01) and cortical cross-sectional area (p = 0.05) were significantly lower at the radius compared with the tibia, indicating that cortical bone at the weight bearing tibia might be less affected by the catabolic actions of continuous parathyroid hormone (PTH) exposure. PTH levels were positively associated with the difference in z-scores of cort BMD (r = 0.439, p < 0.01) indicating that in more severe cases, as expressed by higher PTH levels, the deleterious effects at the non-weight bearing radius might be accentuated.
Conclusion
We found that in postmenopausal women with PHPT, both trabecular and cortical bone are adversely affected. However, at the weight bearing tibia as compared with the radius, the deleterious effects, especially on cortical bone, seem to be attenuated.
Trial registration number
NCT05426512, 21/06/2022, “retrospectively registered”.




Similar content being viewed by others
References
S.J. Silverberg, B.L. Clarke, M. Peacock et al. Current issues in the presentation of asymptomatic primary hyperparathyroidism: proceedings of the Fourth International Workshop. J. Clin. Endocrinol. Metab. 99(10), 3580–3594 (2014)
E.G. Estell, C.J. Rosen, Emerging insights into the comparative effectiveness of anabolic therapies for osteoporosis. Nat. Rev. Endocrinol. 17(1), 31–46 (2021)
B.C. Silva, J.P. Bilezikian, Parathyroid hormone: anabolic and catabolic actions on the skeleton. Curr. Opin. Pharmacol. 22, 41–50 (2015)
Q. Chen, H. Kaji, M.F. Iu et al. Effects of an excess and a deficiency of endogenous parathyroid hormone on volumetric bone mineral density and bone geometry determined by peripheral quantitative computed tomography in female subjects. J. Clin. Endocrinol. Metab. 88(10), 4655–4658 (2003)
L. van Doorn, P. Lips, J.C. Netelenbos, W.H. Hackeng, Bone histomorphometry and serum concentrations of intact parathyroid hormone (PTH(1-84)) in patients with primary hyperparathyroidism. Bone Miner. 23(3), 233–242 (1993)
D.W. Dempster, M. Parisien, S.J. Silverberg et al. On the mechanism of cancellous bone preservation in postmenopausal women with mild primary hyperparathyroidism. J. Clin. Endocrinol. Metab. 84(5), 1562–1566 (1999)
P. Christiansen, T. Steiniche, H. Brockstedt, L. Mosekilde, I. Hessov, F. Melsen, Primary hyperparathyroidism: iliac crest cortical thickness, structure, and remodeling evaluated by histomorphometric methods. Bone 14(5), 755–762 (1993)
S.J. Silverberg, E. Shane, L. de la Cruz et al. Skeletal disease in primary hyperparathyroidism. J. Bone Min. Res 4(3), 283–291 (1989)
T.D. Vu, X.F. Wang, Q. Wang, N.E. Cusano et al. New insights into the effects of primary hyperparathyroidism on the cortical and trabecular compartments of bone. Bone 55(1), 57–63 (2013)
I. Charopoulos, S. Tournis, G. Trovas et al. Effect of primary hyperparathyroidism on volumetric bone mineral density and bone geometry assessed by peripheral quantitative computed tomography in postmenopausal women. J. Clin. Endocrinol. Metab. 91(5), 1748–1753 (2006)
C. Di Leo, A. Bestetti, A. Bastagli et al. Geometry and bone mass in primary hyperparathyroidism assessed by peripheral Quantitative Computed Tomography (pQCT). La Radiol. Med. 105(3), 171–179 (2003)
P. Vestergaard, C.L. Mollerup, V.G. Frøkjaer, P. Christiansen, M. Blichert-Toft, L. Mosekilde, Cohort study of risk of fracture before and after surgery for primary hyperparathyroidism. BMJ 321(7261), 598–602 (2000)
A.G. Robling, The interaction of biological factors with mechanical signals in bone adaptation: recent developments. Curr. Osteoporos. Rep. 10(2), 126–131 (2012)
J. Pfeilschifter, F. Laukhuf, B. Muller-Beckmann, W.F. Blum, T. Pfister, R. Ziegler, Parathyroid hormone increases the concentration of insulin-like growth factor-I and transforming growth factor beta 1 in rat bone. J. Clin. Investig. 96(2), 767–774 (1995)
J.M. Lean, C.J. Jagger, T.J. Chambers, J.W. Chow, Increased insulin-like growth factor I mRNA expression in rat osteocytes in response to mechanical stimulation. Am. J. Physiol. 268(2 Pt 1), E318–E327 (1995)
K.D. Ryder, R.L. Duncan, Parathyroid hormone modulates the response of osteoblast-like cells to mechanical stimulation. Calcif. Tissue Int 67(3), 241–246 (2000)
J. Delgado-Calle, X. Tu, R. Pacheco-Costa et al. Control of bone anabolism in response to mechanical loading and PTH by distinct mechanisms downstream of the PTH receptor. J. Bone Min. Res 32(3), 522–535 (2017)
Y. Ma, W.S. Jee, Z. Yuan et al. Parathyroid hormone and mechanical usage have a synergistic effect in rat tibial diaphyseal cortical bone. J. Bone Min. Res 14(3), 439–448 (1999)
J.F. Campos, A.G.H. Mierzwa, M. Freitas-Jesus, M. Lazaretti-Castro, K.O. Nonaka, R.D. Reginato, Mechanical vibration associated with intermittent PTH improves bone microarchitecture in ovariectomized rats. J. Clin. Densitom. 23(3), 511–519 (2020)
J.W. Chow, S. Fox, C.J. Jagger, T.J. Chambers, Role for parathyroid hormone in mechanical responsiveness of rat bone. Am. J. Physiol. 274(1), E146–E154 (1998)
H. Hagino, T. Okano, M.P. Akhter, M. Enokida, R. Teshima, Effect of parathyroid hormone on cortical bone response to in vivo external loading of the rat tibia. J. Bone Min. Metab. 19(4), 244–250 (2001)
T. Sugiyama, L.K. Saxon, G. Zaman et al. Mechanical loading enhances the anabolic effects of intermittent parathyroid hormone (1-34) on trabecular and cortical bone in mice. Bone 43(2), 238–248 (2008)
J.E. Adams, Quantitative computed tomography. Eur. J. Radio. 71(3), 415–424 (2009)
T.N. Hangartner, Thresholding technique for accurate analysis of density and geometry in QCT, pQCT and microCT images. J. Musculoskelet. Neuronal Interact. 7(1), 9–16 (2007)
G. Schumann, R. Klauke, F. Canalias et al. IFCC primary reference procedures for the measurement of catalytic activity concentrations of enzymes at 37 °C. Part 9: reference procedure for the measurement of catalytic concentration of alkaline phosphatase International Federation of Clinical Chemistry and Laboratory Medicine (IFCC) Scientific Division, Committee on Reference Systems of Enzymes (C-RSE). Clin. Chem. Lab Med 49(9), 1439–1446 (2011)
S. Tournis, J.D. Antoniou, C.G. Liakou et al. Volumetric bone mineral density and bone geometry assessed by peripheral quantitative computed tomography in women with differentiated thyroid cancer under TSH suppression. Clin. Endocrinol. 82(2), 197–204 (2015)
E.M. Stein, B.C. Silva, S. Boutroy et al. Primary hyperparathyroidism is associated with abnormal cortical and trabecular microstructure and reduced bone stiffness in postmenopausal women. J. Bone Min. Res. 28(5), 1029–1040 (2013)
S. Hansen, J.E. Beck Jensen, L. Rasmussen, E.M. Hauge, K. Brixen, Effects on bone geometry, density, and microarchitecture in the distal radius but not the tibia in women with primary hyperparathyroidism: a case-control study using HR-pQCT. J. Bone Min. Res. 25(9), 1941–1947 (2010)
S.A. Kontulainen, J.D. Johnston, D. Liu, C. Leung, T.R. Oxland, H.A. McKay, Strength indices from pQCT imaging predict up to 85% of variance in bone failure properties at tibial epiphysis and diaphysis. J. Musculoskelet. Neuronal Interact. 8(4), 401–409 (2008)
L.B. Meakin, H. Todd, P.J. Delisser et al. Parathyroid hormone’s enhancement of bones’ osteogenic response to loading is affected by ageing in a dose- and time-dependent manner. Bone 98, 59–67 (2017)
A. Palermo, A.M. Naciu, G. Tabacco, et al. Clinical, biochemical, and radiological profile of normocalcemic primary hyperparathyroidism. J. Clin. Endocrinol. Metab. 105(7), e2609–e2616 (2020)
M.M. Tuna, M. Çalışkan, M. Ünal, et al. Normocalcemic hyperparathyroidism is associated with complications similar to those of hypercalcemic hyperparathyroidism. J. Bone Min. Metab. 34(3), 331–335 (2016)
J.A. Kanis, N.C. Harvey, E. Liu, et al. Primary hyperparathyroidism and fracture probability. Osteoporos Int. 34(3), 489–499 (2023)
E. Cavalier, S. Vasikaran, H.P. Bhattoa, A.C. Heijboer, K. Makris, C.Z. Ulmer, The path to the standardization of PTH: is this a realistic possibility? a position paper of the IFCC C-BM. Clin. Chim. Acta 515, 44–51 (2021)
Author information
Authors and Affiliations
Contributions
All listed authors contributed to the conception, design, acquisition, analysis and interpretation of the data and/or writing of the manuscript and that they all approved the version of the manuscript submitted to “Endocrine”.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethics approval
The protocol was approved by the ethics committee of KAT hospital and all procedures were conducted according to the recommendations of Good Clinical Practice and all other applicable local regulatory and ethical requirements and the Declaration of Helsinki (revised edition, Tokyo 2004).
Consent to participate
Written informed consent was obtained from all patients.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Karlafti, E., Dontas, I., Lambrinoudaki, I. et al. Site specific differences in vBMD and geometry in postmenopausal women with primary hyperparathyroidism. Endocrine 83, 205–213 (2024). https://doi.org/10.1007/s12020-023-03491-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-023-03491-8