Abstract
Purpose
The identification of somatic mutations in cancer specimens enables detection of molecular markers for personalized treatment. We recently developed a novel molecular assay and evaluated its clinical performance as an ancillary molecular method for indeterminate thyroid nodule cytology. Herein we describe the analytical validation of the novel targeted next-generation sequencing (NGS) assay in thyroid samples from different sources.
Methods
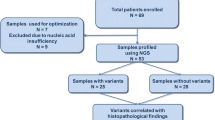
We present validation data of a novel NGS-based panel on 463 thyroid samples, including 310 fine-needle aspiration (FNA) specimens from different sources (FNA collected in preservative solution, liquid-based, and stained smears), 10 fresh frozen, and 143 formalin-fixed paraffin-embedded (FFPE) thyroid tissue specimens. Sequencing performance in the different samples was evaluated along with reproducibility, repeatability, minimum nucleic acid input to detect variants, and analytical sensitivity of the assay.
Results
All thyroid samples achieved high sequencing performance, with a mean base coverage depth ranging from 2228 × (in liquid-based FNA) to 3661 × (in FNA stained smears), and coverage uniformity ranging from 86% (in FFPE) to 95% (in FNA collected in preservative solution), with all target regions covered above the minimum depth required to call a variant (500×). The minimum nucleic acid input was 1 ng. Analytic sensitivity for mutation detection was 2–5% mutant allele frequency.
Conclusions
This validation study of a novel NGS-based assay for thyroid nodules demonstrated that the assay can be reliably used on multiple thyroid sample types, including FNA from different sources and FF and FFPE thyroid samples, thus providing a robust and reliable assay to genotype thyroid nodules, which may improve thyroid cancer diagnosis and care.

Similar content being viewed by others
References
C.D.Seib, J.A.Sosa, Evolving understanding of the epidemiology of thyroid cancer. Endocrinol. Metab. Clin. N. Am 48, 23–35 (2019)
S. Filetti, C. Durante, D. Hartl, S. Leboulleux, L.D. Locati, K. Newbold, M.G. Papotti, A. Berruti, Thyroid cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 30, 1856–1883 (2019). https://doi.org/10.1093/annonc/mdz400
D. Tumino, G. Grani, M. Di Stefano, M. Di Mauro, M. Scutari, T. Rago, L. Fugazzola, M.G. Castagna, F. Maino, Nodular thyroid disease in the era of precision medicine. Front. Endocrinol. (Lausanne) 10, 907–916 (2020). https://doi.org/10.3389/fendo.2019.00907
M. Bongiovanni, A. Spitale, W.C. Faquin, L. Mazzucchelli, Z.W. Baloch, The bethesda system for reporting thyroid cytopathology: a meta-analysis. Acta Cytol. 56, 333–339 (2012). https://doi.org/10.1159/000339959
A. Prete, P. Borges. de Souza, S. Censi, M. Muzza, N. Nucci, M. Sponziello, Update on fundamental mechanisms of thyroid Cancer. Front. Endocrinol. (Lausanne) 11, 102–111 (2020). https://doi.org/10.3389/fendo.2020.00102
Y.E. Nikiforov, Role of molecular markers in thyroid nodule management: then and now. Endocr. Pract. 23, 979–988 (2017). https://doi.org/10.4158/EP171805.RA
N. Agrawal, R. Akbani, B.A. Aksoy, A. Ally, H. Arachchi, S.L. Asa, J.T. Auman, M. Balasundaram, S. Balu, S.B. Baylin, M. Behera, B. Bernard, R. Beroukhim, J.A. Bishop, A.D. Black, T. Bodenheimer, L. Boice, M.S. Bootwalla, J. Bowen, R. Bowlby, C.A. Bristow, R. Brookens, D. Brooks, R. Bryant, E. Buda, Y.S.N. Butterfield, T. Carling, R. Carlsen, S.L. Carter, S.E. Carty, T.A. Chan, A.Y. Chen, A.D. Cherniack, D. Cheung, L. Chin, J. Cho, A. Chu, E. Chuah, K. Cibulskis, G. Ciriello, A. Clarke, G.L. Clayman, L. Cope, J.A. Copland, K. Covington, L. Danilova, T. Davidsen, J.A. Demchok, D. DiCara, N. Dhalla, R. Dhir, S.S. Dookran, G. Dresdner, J. Eldridge, G. Eley, A.K. El-Naggar, S. Eng, J.A. Fagin, T. Fennell, R.L. Ferris, S. Fisher, S. Frazer, J. Frick, S.B. Gabriel, I. Ganly, J. Gao, L.A. Garraway, J.M. Gastier-Foster, G. Getz, N. Gehlenborg, R. Ghossein, R.A. Gibbs, T.J. Giordano, K. Gomez-Hernandez, J. Grimsby, B. Gross, R. Guin, A. Hadjipanayis, H.A. Harper, D.N. Hayes, D.I. Heiman, J.G. Herman, K.A. Hoadley, M. Hofree, R.A. Holt, A.P. Hoyle, F.W. Huang, M. Huang, C.M. Hutter, T. Ideker, L. Iype, A. Jacobsen, S.R. Jefferys, C.D. Jones, S.J.M. Jones, K. Kasaian, E. Kebebew, F.R. Khuri, J. Kim, R. Kramer, R. Kreisberg, R. Kucherlapati, D.J. Kwiatkowski, M. Ladanyi, P.H. Lai, P.W. Laird, E. Lander, M.S. Lawrence, D. Lee, E. Lee, S. Lee, W. Lee, K.M. Leraas, T.M. Lichtenberg, L. Lichtenstein, P. Lin, S. Ling, J. Liu, W. Liu, Y. Liu, V.A. LiVolsi, Y. Lu, Y. Ma, H.S. Mahadeshwar, M.A. Marra, M. Mayo, D.G. McFadden, S. Meng, M. Meyerson, P.A. Mieczkowski, M. Miller, G. Mills, R.A. Moore, L.E. Mose, A.J. Mungall, B.A. Murray, Y.E. Nikiforov, M.S. Noble, A.I. Ojesina, T.K. Owonikoko, B.A. Ozenberger, A. Pantazi, M. Parfenov, P.J. Park, J.S. Parker, E.O. Paull, C.S. Pedamallu, C.M. Perou, J.F. Prins, A. Protopopov, S.S. Ramalingam, N.C. Ramirez, R. Ramirez, B.J. Raphael, W.K. Rathmell, X. Ren, S.M. Reynolds, E. Rheinbay, M.D. Ringel, M. Rivera, J. Roach, A.G. Robertson, M.W. Rosenberg, M. Rosenthal, S. Sadeghi, G. Saksena, C. Sander, N. Santoso, J.E. Schein, N. Schultz, S.E. Schumacher, R.R. Seethala, J. Seidman, Y. Senbabaoglu, S. Seth, S. Sharpe, K.R.M. Shaw, J.P. Shen, R. Shen, S. Sherman, M. Sheth, Y. Shi, I. Shmulevich, G.L. Sica, J.V. Simons, R. Sinha, P. Sipahimalani, R.C. Smallridge, H.J. Sofia, M.G. Soloway, X. Song, C. Sougnez, C. Stewart, P. Stojanov, J.M. Stuart, S.O. Sumer, Y. Sun, B. Tabak, A. Tam, D. Tan, J. Tang, R. Tarnuzzer, B.S. Taylor, N. Thiessen, L. Thorne, V. Thorsson, R.M. Tuttle, C.B. Umbricht, D.J. Van Den Berg, F. Vandin, U. Veluvolu, R.G.W. Verhaak, M. Vinco, D. Voet, V. Walter, Z. Wang, S. Waring, P.M. Weinberger, N. Weinhold, J.N. Weinstein, D.J. Weisenberger, D. Wheeler, M.D. Wilkerson, J. Wilson, M. Williams, D.A. Winer, L. Wise, J. Wu, L. Xi, A.W. Xu, L. Yang, L. Yang, T.I. Zack, M.A. Zeiger, D. Zeng, J.C. Zenklusen, N. Zhao, H. Zhang, J. Zhang, J. Zhang, W. Zhang, E. Zmuda, L. Zou, Integrated genomic characterization of papillary thyroid carcinoma. Cell. 159, 676–690 (2014). https://doi.org/10.1016/j.cell.2014.09.050
I. Landa, T. Ibrahimpasic, L. Boucai, R. Sinha, J.A. Knauf, R.H. Shah, S. Dogan, J.C. Ricarte-Filho, G.P. Krishnamoorthy, B. Xu, N. Schultz, M.F. Berger, C. Sander, B.S. Taylor, R. Ghossein, I. Ganly, J.A. Fagin, Genomic and transcriptomic hallmarks of poorly-differentiated and anaplastic thyroid cancers. J. Clin. Investig. 126, 1052–1066 (2016). https://doi.org/10.1172/JCI85271
M. Sponziello, S. Benvenuti, A. Gentile, V. Pecce, F. Rosignolo, A.R. Virzì, M. Milan, P.M. Comoglio, E. Londin, P. Fortina, A. Barnabei, M. Appetecchia, F. Marandino, D. Russo, S. Filetti, C. Durante, A. Verrienti, Whole exome sequencing identifies a germline MET mutation in two siblings with hereditary wild-type RET medullary thyroid cancer. Hum. Mutat. 39, 371–377 (2018). https://doi.org/10.1002/humu.23378
F. Rosignolo, L. Memeo, F. Monzani, C. Colarossi, V. Pecce, A. Verrienti, C. Durante, G. Grani, L. Lamartina, S. Forte, D. Martinetti, D. Giuffrida, D. Russo, F. Basolo, S. Filetti, M. Sponziello, MicroRNA-based molecular classification of papillary thyroid carcinoma. Int. J. Oncol. 50, 1767–1777 (2017). https://doi.org/10.3892/ijo.2017.3960
F. Rosignolo, M. Sponziello, L. Giacomelli, D. Russo, V. Pecce, M. Biffoni, R. Bellantone, C.P. Lombardi, L. Lamartina, G. Grani, C. Durante, S. Filetti, A. Verrienti, Identification of thyroid-associated serum microRNA profiles and their potential use in thyroid cancer follow-up. J. Endocr. Soc. 1, 3–13 (2017). https://doi.org/10.1210/js.2016-1032
M. Sponziello, C. Brunelli, A. Verrienti, G. Grani, V. Pecce, L. Abballe, V. Ramundo, G. Damante, D. Russo, C.P. Lombardi, C. Durante, E.D. Rossi, P. Straccia, G. Fadda, S. Filetti. Performance of a dual-component molecular assay in cytologically indeterminate thyroid nodules. Endocrine. (2020). https://doi.org/10.1007/s12020-020-02271-y
P. Straccia, C. Brunelli, E.D. Rossi, P. Lanza, M. Martini, T. Musarra, C.P. Lombardi, A. Pontecorvi, G. Fadda, The immunocytochemical expression of VE‐1 (BRAFV600E‐related) antibody identifies the aggressive variants of papillary thyroid carcinoma on liquid‐based cytology. Cytopathology 30, 460–467 (2019). https://doi.org/10.1111/cyt.12690
I. Landa, N. Pozdeyev, C. Korch, L.A. Marlow, R.C. Smallridge, J.A. Copland, Y.C. Henderson, S.Y. Lai, G.L. Clayman, N. Onoda, A.C. Tan, M.E.R. Garcia-Rendueles, J.A. Knauf, B.R. Haugen, J.A. Fagin, R.E. Schweppe, Comprehensive genetic characterization of human thyroid cancer cell lines: a validated panel for preclinical studies. Clin. Cancer Res. 25, 3141–3151 (2019). https://doi.org/10.1158/1078-0432.CCR-18-2953
V. Maggisano, M. Celano, G.E. Lombardo, S.M. Lepore, M. Sponziello, F. Rosignolo, A. Verrienti, F. Baldan, E. Puxeddu, C. Durante, S. Filetti, G. Damante, D. Russo, S. Bulotta, Silencing of hTERT blocks growth and migration of anaplastic thyroid cancer cells. Mol. Cell. Endocrinol. 448, 34–40 (2017). https://doi.org/10.1016/j.mce.2017.03.007
M. Sponziello, G. Silvestri, A. Verrienti, A. Perna, F. Rosignolo, C. Brunelli, V. Pecce, E.D. Rossi, C.P. Lombardi, C. Durante, S. Filetti, G. Fadda, A novel nonsense EIF1AX mutation identified in a thyroid nodule histologically diagnosed as oncocytic carcinoma. Endocrine 62, 492–495 (2018). https://doi.org/10.1007/s12020-018-1611-7
L.J. Jennings, M.E. Arcila, C. Corless, S. Kamel-Reid, I.M. Lubin, J. Pfeifer, R.L. Temple-Smolkin, K.V. Voelkerding, M.N. Nikiforova, Guidelines for validation of next-generation sequencing–based oncology panels: a joint consensus recommendation of the association for Molecular Pathology and College of American Pathologists. J. Mol. Diagn. 19, 341–365 (2017). https://doi.org/10.1016/j.jmoldx.2017.01.011
S. Roy-Chowdhuri, P. Pisapia, M. Salto-Tellez, S. Savic, M. Nacchio, D. de Biase, G. Tallini, G. Troncone, F. Schmitt, Invited review—next-generation sequencing: a modern tool in cytopathology. Virchows Arch. 475, 3–11 (2019). https://doi.org/10.1007/s00428-019-02559-z
S. Roy-Chowdhuri, J. Stewart, Preanalytic variables in cytology: lessons learned from next-generation sequencing—the MD Anderson experience. Arch. Pathol. Lab. Med. 140, 1191–1199 (2016). https://doi.org/10.5858/arpa.2016-0117-RA
L. Lamartina, G. Grani, C. Durante, S. Filetti, Recent advances in managing differentiated thyroid cancer. F1000Res 7, 86 (2018). https://doi.org/10.12688/f1000research.12811.1
L. Lamartina, G. Grani, C. Durante, I. Borget, S. Filetti, M. Schlumberger, Follow-up of differentiated thyroid cancer—what should (and what should not) be done. Nat. Rev. Endocrinol. 14, 538–551 (2018). https://doi.org/10.1038/s41574-018-0068-3
S. Kim, C. Park, Y. Ji, D.G. Kim, H. Bae, M. van Vrancken, D.H. Kim, K.M. Kim, Deamination effects in formalin-fixed, paraffin-embedded tissue samples in the era of precision medicine. J. Mol. Diagn. 19, 137–146 (2017). https://doi.org/10.1016/j.jmoldx.2016.09.006
M. Serizawa, T. Yokota, A. Hosokawa, K. Kusafuka, T. Sugiyama, Y. Tsubosa, H. Yasui, T. Nakajima, Y. Koh, The efficacy of uracil DNA glycosylase pretreatment in amplicon-based massively parallel sequencing with DNA extracted from archived formalin-fixed paraffin-embedded esophageal cancer tissues. Cancer Genet. 208, 415–427 (2015). https://doi.org/10.1016/j.cancergen.2015.05.001
Funding
The study was supported by “Sapienza” University of Rome (grant RP11916B8919C921 to M.S.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures involving humans were in accordance with the ethical standards of the institutional and/or national research committee (Ethics Committee of Azienda Universitaria Policlinico Umberto I, Rome, Italy; Prot. 806/16, Rif. 4233) and with the 1964 Helsinki Declaration and its later amendments.
Informed consent
Written informed consent was obtained from all subjects included in the study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Verrienti, A., Pecce, V., Abballe, L. et al. Analytical validation of a novel targeted next-generation sequencing assay for mutation detection in thyroid nodule aspirates and tissue. Endocrine 69, 451–455 (2020). https://doi.org/10.1007/s12020-020-02372-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-020-02372-8