Abstract
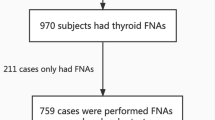
Despite several reports on the association between molecular profiling, aggressive histology, and clinical outcomes, the association between mutation expression and pre-operative cytology is yet to be demonstrated. Therefore, we performed a retrospective, single-center study, including all patients who underwent molecular profiling of thyroid nodules in Bethesda System for Reporting Thyroid Cytopathology (BSRTC) categories III to VI, between 2018 and 2019. Medical records were reviewed to collect demographics, cytology results according to BSRTC, final pathology (presence of malignancy and its type, as well as presence of aggressive features, including extrathyroidal extension, positive neck lymph nodes, and multifocality), and the identified genetic variants stratified by risk levels, according to the 2015 ATA guidelines. We supplemented this analysis with a systematic review to identify the variant distributions across the literature. We included data on 55 nodules from 48 patients for the final analysis. A significant positive correlation was found between BSRTC categories and the mutation risk level, shown by an increase in the intermediate to high-risk mutation rate in the higher BSRTC categories (Rs = 0.660, p ≤ 0.001). A significant positive correlation was also found between mutation risk levels and the presence of malignancy and aggressive tumor features (Rs = 0.637, p < 0.001 and Rs = 0.459, p = 0.006, respectively). This novel positive and significant correlation between BSRTC categories and the mutation risk level provides additional insight to aid clinicians in the interpretation of BSRTC results and may contribute to the discussion of appropriate management of thyroid nodule with patients.





Similar content being viewed by others
Availability of Data and Materials
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request.
References
Ferris RL, Baloch Z, Bernet V, Chen A, Fahey TJ, Ganly I, et al. American Thyroid Association Statement on Surgical Application of Molecular Profiling for Thyroid Nodules: Current Impact on Perioperative Decision Making. Thyroid 2015;25:760–8.
Kumar N, Gupta R, Gupta S. Molecular testing in diagnosis of indeterminate thyroid cytology: Trends and drivers. Diagnostic Cytopathology 2020;dc.24522.
Rossi ED, Pantanowitz L, Faquin WC. The Role of Molecular Testing for the Indeterminate Thyroid FNA. Genes 2019;10:736.
Haugen BR, Alexander EK, Bible KC, Doherty GM, Mandel SJ, Nikiforov YE, et al. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 2016;26:1–133.
Cibas ES, Ali SZ. The 2017 Bethesda System for Reporting Thyroid Cytopathology. Thyroid 2017;27:1341–6.
Krasner JR, Alyouha N, Pusztaszeri M, Forest V-I, Hier MP, Avior G, et al. Molecular mutations as a possible factor for determining extent of thyroid surgery. J of Otolaryngol - Head & Neck Surg 2019;48:51.
Basolo F, Torregrossa L, Giannini R, Miccoli M, Lupi C, Sensi E, et al. Correlation between the BRAF V600E Mutation and Tumor Invasiveness in Papillary Thyroid Carcinomas Smaller than 20 Millimeters: Analysis of 1060 Cases. The Journal of Clinical Endocrinology & Metabolism 2010;95:4197–205.
Elisei R, Ugolini C, Viola D, Lupi C, Biagini A, Giannini R, et al. BRAF(V600E) mutation and outcome of patients with papillary thyroid carcinoma: a. J Clin Endocrinol Metab 2008;93:3943–9.
Liu X, Bishop J, Shan Y, Pai S, Liu D, Murugan AK, et al. Highly prevalent TERT promoter mutations in aggressive thyroid cancers. Endocr Relat Cancer 2013;20:603–10.
Romei C, Ciampi R, Elisei R. A comprehensive overview of the role of the RET proto-oncogene in thyroid carcinoma. Nature Reviews Endocrinology 2016;12:192–202.
Patel SG, Carty SE, McCoy KL, Ohori NP, LeBeau SO, Seethala RR, et al. Preoperative detection of RAS mutation may guide extent of thyroidectomy. Surgery 2017;161:168–75.
Yip L, Nikiforova MN, Carty SE, Yim JH, Stang MT, Tublin MJ, et al. Optimizing surgical treatment of papillary thyroid carcinoma associated with BRAF mutation. Surgery 2009;146:1215–23.
Marti JL, Shaha AR. Molecular Testing for Indeterminate Thyroid Nodules—When the Rubber Meets the Road. JAMA Otolaryngol Head Neck Surg 2019;145:792.
Yip L, Nikiforova MN, Yoo JY, McCoy KL, Stang MT, Armstrong MJ, et al. Tumor genotype determines phenotype and disease-related outcomes in thyroid cancer: a study of 1510 patients. Ann Surg 2015;262:519–25; discussion 524–525.
Chen T, Gilfix BM, Rivera J, Sadeghi N, Richardson K, Hier MP, et al. The Role of the ThyroSeq v3 Molecular Test in the Surgical Management of Thyroid Nodules in the Canadian Public Health Care Setting. Thyroid 2020;thy.2019.0539.
Desai D, Lepe M, Baloch ZW, Mandel SJ. ThyroSeq v3 for Bethesda III and IV: An institutional experience. Cancer Cytopathol 2021;129:164–70.
Steward DL, Carty SE, Sippel RS, Yang SP, Sosa JA, Sipos JA, et al. Performance of a Multigene Genomic Classifier in Thyroid Nodules With Indeterminate Cytology: A Prospective Blinded Multicenter Study. JAMA Oncol 2019;5:204.
Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 2009;6:e1000097.
Kleiman DA, Beninato T, Soni A, Shou Y, Zarnegar R, Fahey TJ. Does Bethesda Category Predict Aggressive Features in Malignant Thyroid Nodules? Ann Surg Oncol 2013;20:3484–90.
Liu X, Medici M, Kwong N, Angell TE, Marqusee E, Kim MI, et al. Bethesda Categorization of Thyroid Nodule Cytology and Prediction of Thyroid Cancer Type and Prognosis. Thyroid 2016;26:256–61.
VanderLaan PA, Marqusee E, Krane JF. Features associated with locoregional spread of papillary carcinoma correlate with diagnostic category in the Bethesda System for reporting thyroid cytopathology. Cancer Cytopathology 2012;120:245–53.
Kimura ET, Nikiforova MN, Zhu Z, Knauf JA, Nikiforov YE, Fagin JA. High prevalence of BRAF mutations in thyroid cancer: genetic evidence for constitutive activation of the RET/PTC-RAS-BRAF signaling pathway in papillary thyroid carcinoma. Cancer Res 2003;63:1454–7.
Hsiao SJ, Nikiforov Y. Molecular approaches to thyroid cancer diagnosis. Endocrine Related Cancer 2014;ERC-14–0166.
Shi C, Cao J, Shi T, Liang M, Ding C, Lv Y, et al. BRAFV600E mutation, BRAF-activated long non-coding RNA and miR-9 expression in papillary thyroid carcinoma, and their association with clinicopathological features. World J Surg Onc 2020;18:145.
Xing M. Prognostic utility of BRAF mutation in papillary thyroid cancer. Molecular and Cellular Endocrinology 2010;321:86–93.
Xing M, Alzahrani AS, Carson KA, Viola D, Elisei R, Bendlova B, et al. Association Between BRAF V600E Mutation and Mortality in Patients With Papillary Thyroid Cancer. JAMA 2013;309:1493.
Crispo F, Notarangelo T, Pietrafesa M, Lettini G, Storto G, Sgambato A, et al. BRAF Inhibitors in Thyroid Cancer: Clinical Impact, Mechanisms of Resistance and Future Perspectives. Cancers (Basel) 2019;11:1388.
Brandler TC, Liu CZ, Cho M, Zhou F, Cangiarella J, Yee-Chang M, et al. Does Noninvasive Follicular Thyroid Neoplasm With Papillary-Like Nuclear Features (NIFTP) Have a Unique Molecular Profile? Am J Clin Pathol 2018;150:451–60.
Pusztaszeri M, Bongiovanni M. The impact of non-invasive follicular thyroid neoplasm with papillary-like nuclear features (NIFTP) on the diagnosis of thyroid nodules. Gland Surg 2019;8:S86–97.
Wong KS, Higgins SE, Marqusee E, Nehs MA, Angell T, Barletta JA. Tall Cell Variant of Papillary Thyroid Carcinoma: Impact of Change in WHO Definition and Molecular Analysis. Endocr Pathol 2019;30:43–8.
Lubitz CC, Economopoulos KP, Pawlak AC, Lynch K, Dias-Santagata D, Faquin WC, et al. Hobnail Variant of Papillary Thyroid Carcinoma: An Institutional Case Series and Molecular Profile. Thyroid 2014;24:958–65.
Jin M, Song DE, Ahn J, Song E, Lee Y-M, Sung T-Y, et al. Genetic Profiles of Aggressive Variants of Papillary Thyroid Carcinomas. Cancers 2021;13:892.
Author information
Authors and Affiliations
Contributions
I.T, I.S, O.C, A.M, and G.A had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. Study concept and design: I.T., I.S., and G.A. Acquisition of data: I.S., A.M., and G.A. Analysis and interpretation of data: I.T., I.S., O.C. Drafting of the manuscript: I.T. and I.S. Critical revision of the manuscript for important intellectual content: O.C. and G.A. Statistical analysis: I.T. and I.S. Study supervision: G.A.
Corresponding author
Ethics declarations
Ethics Approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of Hillel Yaffe Medical Center (approval number 3819).
Statement of Ethics
This clinical trial was conducted in compliance with good clinical practices (GCP) as specified in the Israeli Ministry of Health regulations as well as in the ICH GCP Guidelines.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tessler, I., Shochat, I., Cohen, O. et al. Positive Correlation of Thyroid Nodule Cytology with Molecular Profiling—a Single-Center Experience. Endocr Pathol 32, 480–488 (2021). https://doi.org/10.1007/s12022-021-09680-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12022-021-09680-3