Abstract
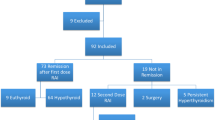
Iodine is beneficial against Graves’ thyrotoxicosis, though its effects are short-lived. However, its long-term effectiveness as an initial therapy has not been fully elucidated. Here, we compared the effects of potassium iodine (KI) and methimazole (MMI) in Graves’ thyrotoxicosis and on thyrotropin receptor antibody (TRAb) levels. Between 2008 and 2011, 293 patients with untreated Graves’ disease visited the outpatient clinic of Juntendo University. Of these, 227 patients were treated with MMI and 30 treated with KI as the initial therapy. To compare the effects of KI and MMI, we identified patients with similar probabilities of receiving MMI or KI using propensity score (PS) analysis based on the observed clinical features. PS matching created 20 matched pairs of patients with Graves’ disease treated with MMI and KI. The baseline characteristics of post-matched patients treated with MMI were comparable to those treated with KI (FT3; 7.16 ± 2.30, 6.56 ± 1.85 pg/ml, FT4; 2.57 ± 0.79, 2.49 ± 0.70 ng/dl, respectively). The initial dose of MMI was 14.0 ± 8.2 mg/day and that of KI was 53.6 ± 11.7 mg/day. Three patients of the KI group did not respond to the monotherapy, requiring the inclusion of antithyroid drugs. One patient on MMI developed moderate skin eruption, but continued the treatment. Patients who continued the initial treatment showed significant and comparable reductions in FT4, FT3 and TRAb by MMI as well as by KI at the end of 12-month treatment. Although patients were limited to mild untreated Graves’ disease thyrotoxicosis, KI offers a possible alternative initial treatment for this condition.

Similar content being viewed by others
Abbreviations
- GD:
-
Graves’ disease
- FT3:
-
Free triiodothyronine
- FT4:
-
Free thyroxine
- KI:
-
Potassium iodide
- MMI:
-
Methimazole
- PS:
-
Propensity score
- STA-PSV:
-
Mean peak systolic velocity of superior thyroid artery
- TSH:
-
Thyrotropin
- TRAb:
-
Thyrotropin receptor antibody
References
D.S. Cooper, Antithyroid drugs. N. Engl. J. Med. 352, 905–917 (2005)
S.M. McLachlan, Y. Nagayama, B. Rapoport, Insight into Graves’ hyperthyroidism from animal models. Endocr. Rev. 26, 800–832 (2005)
T.H. Totterman, F.A. Karlsson, M. Bengtsson, I. Mendel-Hartvig, Induction of circulating activated suppressor-like T cells by methimazole therapy for Graves’ disease. N. Engl. J. Med. 316, 15–22 (1987)
N. Mitsiades, V. Poulaki, S. Tseleni-Balafouta, G.P. Chrousos, D.A. Koutras, Fas ligand expression in thyroid follicular cells from patients with thionamide-treated Graves’ disease. Thyroid 10, 527–532 (2000)
M. Meyer-Gessner, G. Benker, T. Olbricht, R. Windeck, K. Cissewski, C. Reiners, D. Reinwein, Side effects of antithyroid therapy of hyperthyroidism. A study of 1256 continuously treated patients. Dtsch. Med. Wochenschr. 114, 166–171 (1989)
Y.F. Liaw, M.J. Huang, K.D. Fan, K.L. Li, S.S. Wu, T.J. Chen, Hepatic injury during propylthiouracil therapy in patients with hyperthyroidism. A cohort study. Ann Intern. Med. 118, 424–428 (1993)
L. Wartofsky, B.J. Ransil, S.H. Ingbar, Inhibition by iodine of the release of thyroxine from the thyroid glands of patients with thyrotoxicosis. J. Clin. Invest. 49, 78–86 (1970)
P.H. Eng, G.R. Cardona, S.L. Fang, Escape from the acute Wolff-Chaikoff effect is associated with a decrease in thyroid sodium/iodide symporter messenger ribonucleic acid and protein. Endocrinology 140, 3404–3410 (1999)
L.J. Degroot, M.A. Greer, The effect of stable iodide on thyroid secretion in man. Metabolism 5, 682–696 (1956)
J. Wolff, I.L. Chaikoff, Plasma inorganic iodide as a homeostatic regulator of thyroid function. J. Biol. Chem. 174, 555–564 (1948)
S. Nagataki, K. Shizume, K. Nakao, Effect of iodide on thyroidal iodine turnover in hyperthyroid subjects. J. Clin. Endocrinol. Metab. 30, 469–478 (1970)
C.H. Emerson, A.J. Anderson, W.J. Howard, R.D. Utiger, Serum thyroxine and triiodothyronine concentrations during iodide treatment of hyperthyroidism. J. Clin. Endocrinol. Metab. 40, 33–36 (1975)
G. Robuschi, A. Manfredi, M. Salvi, E. Gardini, M. Montermini, L. d’Amato, E. Borciani, L. Negrotti, A. Gnudi, E. Roti, Effect of sodium ipodate and iodide on free T4 and free T3 concentrations in patients with Graves’ disease. J. Endocrinol. Invest. 9, 287–291 (1986)
G. Philippou, D.A. Koutras, G. Piperingos, A. Souvatzoglou, S.D. Moulopoulos, The effect of iodide on serum thyroid hormone levels in normal persons, in hyperthyroid patients, and in hypothyroid patients on thyroxine replacement. Clin. Endocrinol. (Oxf.) 36, 573–578 (1992)
C.M. Feek, J.S. Sawers, W.J. Irvine, G.J. Beckett, W.A. Ratcliffe, A.D. Toft, Combination of potassium iodide and propranolol in preparation of patients with Graves’ disease for thyroid surgery. N. Engl. J. Med. 302, 883–885 (1980)
E. Roti, G. Robuschi, E. Gardini, M. Montermini, M. Salvi, A. Manfredi, A. Gnudi, L.E. Braverman, Comparison of methimazole, methimazole and sodium ipodate, and methimazole and saturated solution of potassium iodide in the early treatment of hyperthyroid Graves’ disease. Clin Endocrinol. (Oxf.) 28, 305–314 (1988)
K. Takata, N. Amino, S. Kubota, I. Sasaki, E. Nishihara, T. Kudo, M. Ito, S. Fukata, A. Miyauchi, Benefit of short-term iodide supplementation to antithyroid drug treatment of thyrotoxicosis due to Graves’ disease. Clin. Endocrinol. (Oxf.) 72, 845–850 (2010)
E. Roti, E. Gardini, R. Minelli, L. Bianconi, M. Salvi, G. Gavaruzzi, L.E. Braverman, Effects of chronic iodine administration on thyroid status in euthyroid subjects previously treated with antithyroid drugs for Graves’ hyperthyroidism. J. Clin. Endocrinol. Metab. 76, 928–932 (1993)
E. Roti, G. Robuschi, A. Manfredi, L. D’Amato, E. Gardini, M. Salvi, M. Montermini, A.L. Barlli, A. Gnudi, L.E. Braverman, Comparative effects of sodium ipodate and iodide on serum thyroid hormone concentrations in patients with Graves’ disease. Clin. Endocrinol. (Oxf.) 22, 489–496 (1985)
T.T. Tan, P. Morat, M.L. Ng, B.A. Khalid, Effects of Lugol’s solution on thyroid function in normals and patients with untreated thyrotoxicosis. Clin. Endocrinol. (Oxf.) 30, 645–649 (1989)
G. Philippou, G. Piperingos, A. Souvatzoglou, D.A. Koutras, S.D. Moulopoulos, Treatment of hyperthyroidism with potassium iodide. Exp. Clin. Endocrinol. 97, 308–311 (1991)
T. Uchida, K. Takeno, M. Goto, R. Kanno, S. Kubo, S. Takahashi, K. Azuma, K. Sakai, Y. Fujitani, T. Hirose, R. Kawamori, H. Watada, Superior thyroid artery mean peak systolic velocity for the diagnosis of thyrotoxicosis in Japanese patients. Endocr. J. 57, 439–443 (2010)
P.C. Austin, Balance diagnostics for comparing the distribution of baseline covariates between treatment groups in propensity-score matched samples. Stat. Med. 28, 3083–3107 (2009)
T. Kasai, K. Miyauchi, N. Kubota, K. Kajimoto, A. Amano, H. Daida, Probucol therapy improves long-term (>10-year) survival after complete revascularization: a propensity analysis. Atherosclerosis 220, 463–469 (2012)
T.T. Zava, D.T. Zava, Assessment of Japanese iodine intake based on seaweed consumption in Japan: a literature-based analysis. Thyroid Res. 4, 14 (2011)
B.R. Shah, A. Laupacis, J.E. Hux, P.C. Austin, Propensity score methods gave similar results to traditional regression modeling in observational studies: a systematic review. J. Clin. Epidemiol. 58, 550–559 (2005)
Acknowledgments
The authors thank Noriko Iijima and Emi Ito for the excellent technical assistance.
Conflicts of interest
TK has research funds from Philips-Respironics, Teijin Home Healthcare, Fukuda Denshi. TM has research funds from Eli Lilly, Takeda Pharmaceutical Co., MSD. AK has received lecture fees from Takeda Pharmaceutical Co., Sanofi-Aventis. YF has lecture fees from Novartis Pharmaceuticals, Eli Lilly and research funds from Eli Lilly, Takeda Pharmaceutical Co., MSD. HW has received lecture fees from Boehringer Ingelheim, Sanofi-Aventis, Ono Pharmaceutical Co., Novo Nordisk Pharma, Novartis Pharmaceuticals, Eli Lilly, Sanwakagaku Kenkyusho, Daiichi Sankyo Inc., Takeda Pharmaceutical Co., MSD, Dainippon Sumitomo Pharm., Kowa Co. and research funds from Boehringer Ingelheim, Pfizer, Mochida Pharmaceutical Co. Sanofi-Aventis, Novo Nordisk Pharma, Novartis Pharmaceuticals, Sanwakagaku Kenkyusho, Terumo Corp. Eli Lilly, Mitsubishi Tanabe Pharma, Daiichi Sankyo Inc., Takeda Pharmaceutical Co., MSD, Shionogi, Pharma, Dainippon Sumitomo Pharma, Kissei Pharma, and Astrazeneca.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Uchida, T., Goto, H., Kasai, T. et al. Therapeutic effectiveness of potassium iodine in drug-naïve patients with Graves’ disease: a single-center experience. Endocrine 47, 506–511 (2014). https://doi.org/10.1007/s12020-014-0171-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-014-0171-8