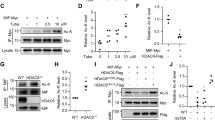
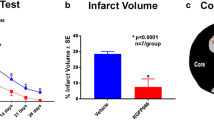
Abstract
This study focuses on understanding the role of c-Myc, a cancer-associated transcription factor, in the penumbra following ischemic stroke. While its involvement in cell death and survival is recognized, its post-translational modifications, particularly acetylation, remain understudied in ischemia models. Investigating these modifications could have significant clinical implications for controlling c-Myc activity in the central nervous system. Although previous studies on c-Myc acetylation have been limited to non-neuronal cells, our research examines its expression in perifocal cells during stroke recovery to explore regulatory mechanisms via acetylation. We found that in peri-infarct neurons, c-Myc is upregulated with acetylation at K148 but not K323 during the acute phase of stroke, with SIRT2 deacetylase primarily affecting K148 acetylation. Molecular dynamics simulations suggest that lysine 148 plays a crucial role in stabilizing c-Myc spatial structure. Increased acetylation at K148 reduces c-Myc compaction, potentially limiting its nuclear penetration, promoting calpain-mediated cleavage, and decreasing nuclear localization. Additionally, cytoplasmic acetylation at K148 may alter c-Myc's interaction with unidentified proteins, potentially influencing its pro-apoptotic effects and promoting cytoplasmic accumulation. Targeting SIRT2 with selective inhibitors could be a promising avenue for future stroke therapy strategies.





Similar content being viewed by others
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
References
Alam, M.S. (2022). Proximity ligation assay (PLA). In L. Del Valle (Ed.), Immunohistochemistry and Immunocytochemistry (Vol. 2422, pp. 191–201). Springer US. https://doi.org/10.1007/978-1-0716-1948-3_13
Ariga, H. (2015). Common Mechanisms of Onset of Cancer and Neurodegenerative Diseases. Biological & Pharmaceutical Bulletin, 38(6), 795–808. https://doi.org/10.1248/bpb.b15-00125
Bauer, P., Hess, B., & Lindahl, E. (2023). GROMACS 2022.5 Manual. https://doi.org/10.5281/zenodo.7586765
Belin, S., Nawabi, H., Wang, C., Tang, S., Latremoliere, A., Warren, P., et al. (2015). Injury-induced decline of intrinsic regenerative ability revealed by quantitative proteomics. Neuron, 86(4), 1000–1014. https://doi.org/10.1016/j.neuron.2015.03.060
Bolte, S., & Cordelières, F. P. (2006). A guided tour into subcellular colocalization analysis in light microscopy. Journal of Microscopy, 224(3), 213–232. https://doi.org/10.1111/j.1365-2818.2006.01706.x
Chen, X., Wales, P., Quinti, L., Zuo, F., Moniot, S., Herisson, F., et al. (2015). The sirtuin-2 inhibitor AK7 is neuroprotective in models of Parkinson’s disease but not amyotrophic lateral sclerosis and cerebral ischemia. PLoS ONE, 10(1), e0116919. https://doi.org/10.1371/journal.pone.0116919
Conacci-Sorrell, M., Ngouenet, C., & Eisenman, R. N. (2010). Myc-Nick: A cytoplasmic cleavage product of myc that promotes α-tubulin acetylation and cell differentiation. Cell, 142(3), 480–493. https://doi.org/10.1016/j.cell.2010.06.037
Dang, C. V. (2012). MYC on the path to cancer. Cell, 149(1), 22–35. https://doi.org/10.1016/j.cell.2012.03.003
Das, S. K., Lewis, B. A., & Levens, D. (2023). MYC: A complex problem. Trends in Cell Biology, 33(3), 235–246. https://doi.org/10.1016/j.tcb.2022.07.006
Demyanenko, S., Dzreyan, V., & Uzdensky, A. (2019). Axotomy-induced changes of the protein profile in the crayfish ventral cord ganglia. Journal of Molecular Neuroscience, 68(4), 667–678. https://doi.org/10.1007/s12031-019-01329-5
Demyanenko, S., Gantsgorn, E., Rodkin, S., & Sharifulina, S. (2020a). Localization and expression of sirtuins 1, 2, 6 and plasticity-related proteins in the recovery period after a photothrombotic stroke in mice. Journal of Stroke and Cerebrovascular Diseases, 29(10), 105152. https://doi.org/10.1016/j.jstrokecerebrovasdis.2020.105152
Demyanenko, S., & Uzdensky, A. (2017). Profiling of signaling proteins in penumbra after focal photothrombotic infarct in the rat brain cortex. Molecular Neurobiology, 54(9), 6839–6856. https://doi.org/10.1007/s12035-016-0191-x
Demyanenko, S. V., Nikul, V. V., & Uzdensky, A. B. (2020b). The neuroprotective effect of the HDAC2/3 inhibitor MI192 on the penumbra after photothrombotic stroke in the mouse brain. Molecular Neurobiology, 57(1), 239–248. https://doi.org/10.1007/s12035-019-01773-9
Dunker, A. K., Silman, I., Uversky, V. N., & Sussman, J. L. (2008). Function and structure of inherently disordered proteins. Current Opinion in Structural Biology, 18(6), 756–764. https://doi.org/10.1016/j.sbi.2008.10.002
Eid, M., Dzreyan, V., & Demyanenko, S. (2022). Sirtuins 1 and 2 in the acute period after photothrombotic stroke: Expression, localization and involvement in apoptosis. Frontiers in Physiology, 13, 782684. https://doi.org/10.3389/fphys.2022.782684
Faiola, F., Liu, X., Lo, S., Pan, S., Zhang, K., Lymar, E., et al. (2005). Dual regulation of c-Myc by p300 via acetylation-dependent control of Myc protein turnover and coactivation of Myc-induced transcription. Molecular and Cellular Biology, 25(23), 10220–10234. https://doi.org/10.1128/MCB.25.23.10220-10234.2005
Farrell, A. S., & Sears, R. C. (2014). MYC degradation. Cold Spring Harbor Perspectives in Medicine, 4(3), a014365–a014365. https://doi.org/10.1101/cshperspect.a014365
Ferrer, I., Friguls, B., Dalfó, E., & Planas, A. M. (2003). Early modifications in the expression of mitogen-activated protein kinase (MAPK/ERK), stress-activated kinases SAPK/JNK and p38, and their phosphorylated substrates following focal cerebral ischemia. Acta Neuropathologica, 105(5), 425–437. https://doi.org/10.1007/s00401-002-0661-2
Guo, J., Parise, R. A., Joseph, E., Egorin, M. J., Lazo, J. S., Prochownik, E. V., & Eiseman, J. L. (2009). Efficacy, pharmacokinetics, tisssue distribution, and metabolism of the Myc-Max disruptor, 10058–F4 [Z, E]-5-[4-ethylbenzylidine]-2-thioxothiazolidin-4-one, in mice. Cancer Chemotherapy and Pharmacology, 63(4), 615–625. https://doi.org/10.1007/s00280-008-0774-y
Guzenko, V. V., Bachurin, S. S., Khaitin, A. M., Dzreyan, V. A., Kalyuzhnaya, Y. N., Bin, H., & Demyanenko, S. V. (2023). Acetylation of p53 in the cerebral cortex after photothrombotic stroke. Translational Stroke Research. https://doi.org/10.1007/s12975-023-01183-z
Jumper, J., Evans, R., Pritzel, A., Green, T., Figurnov, M., Ronneberger, O., et al. (2021). Highly accurate protein structure prediction with AlphaFold. Nature, 596(7873), 583–589. https://doi.org/10.1038/s41586-021-03819-2
Kumar, D., Sharma, N., & Giri, R. (2017). Therapeutic interventions of cancers using intrinsically disordered proteins as drug targets: c-Myc as model system. Cancer Informatics, 16, 117693511769940. https://doi.org/10.1177/1176935117699408
Lee, H.-P., Kudo, W., Zhu, X., Smith, M. A., & Lee, H. (2011). Early induction of c-Myc is associated with neuronal cell death. Neuroscience Letters, 505(2), 124–127. https://doi.org/10.1016/j.neulet.2011.10.004
Ma, J.-J., Ju, X., Xu, R.-J., Wang, W.-H., Luo, Z.-P., Liu, C.-M., et al. (2019). Telomerase reverse transcriptase and p53 regulate mammalian peripheral nervous system and CNS axon regeneration downstream of c-Myc. The Journal of Neuroscience, 39(46), 9107–9118. https://doi.org/10.1523/JNEUROSCI.0419-19.2019
Madden, S. K., De Araujo, A. D., Gerhardt, M., Fairlie, D. P., & Mason, J. M. (2021). Taking the Myc out of cancer: Toward therapeutic strategies to directly inhibit c-Myc. Molecular Cancer, 20(1), 3. https://doi.org/10.1186/s12943-020-01291-6
Manders, E. M., Stap, J., Brakenhoff, G. J., van Driel, R., & Aten, J. A. (1992). Dynamics of three-dimensional replication patterns during the S-phase, analysed by double labelling of DNA and confocal microscopy. Journal of Cell Science, 103(3), 857–862. https://doi.org/10.1242/jcs.103.3.857
Mao, B., Zhao, G., Lv, X., Chen, H.-Z., Xue, Z., Yang, B., et al. (2011). Sirt1 deacetylates c-Myc and promotes c-Myc/Max association. The International Journal of Biochemistry & Cell Biology, 43(11), 1573–1581. https://doi.org/10.1016/j.biocel.2011.07.006
Marinkovic, T., & Marinkovic, D. (2021). Obscure involvement of MYC in neurodegenerative diseases and neuronal repair. Molecular Neurobiology, 58(8), 4169–4177. https://doi.org/10.1007/s12035-021-02406-w
McCloy, R. A., Rogers, S., Caldon, C. E., Lorca, T., Castro, A., & Burgess, A. (2014). Partial inhibition of Cdk1 in G2 phase overrides the SAC and decouples mitotic events. Cell Cycle, 13(9), 1400–1412. https://doi.org/10.4161/cc.28401
Modak, R., Basha, J., Bharathy, N., Maity, K., Mizar, P., Bhat, A. V., et al. (2013). Probing p300/CBP associated factor (PCAF)-dependent pathways with a small molecule inhibitor. ACS Chemical Biology, 8(6), 1311–1323. https://doi.org/10.1021/cb4000597
Moser, M. A., Hagelkruys, A., & Seiser, C. (2014). Transcription and beyond: The role of mammalian class I lysine deacetylases. Chromosoma, 123(1–2), 67–78. https://doi.org/10.1007/s00412-013-0441-x
Nakhate, K. T., Bharne, A. P., Verma, V. S., Aru, D. N., & Kokare, D. M. (2018). Plumbagin ameliorates memory dysfunction in streptozotocin induced Alzheimer’s disease via activation of Nrf2/ARE pathway and inhibition of β-secretase. Biomedicine & Pharmacotherapy, 101, 379–390. https://doi.org/10.1016/j.biopha.2018.02.052
Nebbioso, A., Carafa, V., Conte, M., Tambaro, F. P., Abbondanza, C., Martens, J., et al. (2017). c-Myc modulation and acetylation is a key HDAC inhibitor target in cancer. Clinical Cancer Research, 23(10), 2542–2555. https://doi.org/10.1158/1078-0432.CCR-15-2388
Papamokos, G. V., Tziatzos, G., Papageorgiou, D. G., Georgatos, S. D., Politou, A. S., & Kaxiras, E. (2012). Structural role of RKS motifs in chromatin interactions: A molecular dynamics study of HP1 bound to a variably modified histone tail. Biophysical Journal, 102(8), 1926–1933. https://doi.org/10.1016/j.bpj.2012.03.030
Patel, J. H., Du, Y., Ard, P. G., Phillips, C., Carella, B., Chen, C.-J., et al. (2004). The c-Myc oncoprotein is a substrate of the acetyltransferases hGCN5/PCAF and TIP60. Molecular and Cellular Biology, 24(24), 10826–10834. https://doi.org/10.1128/MCB.24.24.10826-10834.2004
Schemies, J., Sippl, W., & Jung, M. (2009). Histone deacetylase inhibitors that target tubulin. Cancer Letters, 280(2), 222–232. https://doi.org/10.1016/j.canlet.2009.01.040
Tian, C., Kasavajhala, K., Belfon, K. A. A., Raguette, L., Huang, H., Migues, A. N., et al. (2020). ff19SB: Amino-acid-specific protein backbone parameters trained against quantum mechanics energy surfaces in solution. Journal of Chemical Theory and Computation, 16(1), 528–552. https://doi.org/10.1021/acs.jctc.9b00591
Venkateswaran, N., & Conacci-Sorrell, M. (2020). MYC leads the way. Small GTPases, 11(2), 86–94. https://doi.org/10.1080/21541248.2017.1364821
Vervoorts, J., Lüscher-Firzlaff, J., & Lüscher, B. (2006). The ins and outs of MYC regulation by posttranslational mechanisms. Journal of Biological Chemistry, 281(46), 34725–34729. https://doi.org/10.1074/jbc.R600017200
Weinert, B. T., Wagner, S. A., Horn, H., Henriksen, P., Liu, W. R., Olsen, J. V., et al. (2011). Proteome-wide mapping of the Drosophila acetylome demonstrates a high degree of conservation of lysine acetylation. Science Signaling, 4(183), 48. https://doi.org/10.1126/scisignal.2001902
Zhang, M., Pan, Y., Tang, D., Dorfman, R. G., Xu, L., Zhou, Q., et al. (2019). Low levels of pyruvate induced by a positive feedback loop protects cholangiocarcinoma cells from apoptosis. Cell Communication and Signaling, 17(1), 23. https://doi.org/10.1186/s12964-019-0332-8
Zhang, S., Boyd, J., Delaney, K., & Murphy, T. H. (2005). Rapid reversible changes in dendritic spine structure In Vivo gated by the degree of ischemia. The Journal of Neuroscience, 25(22), 5333–5338. https://doi.org/10.1523/JNEUROSCI.1085-05.2005
Funding
This work was supported by the Ministry of Science and Higher Education of the Russian Federation (Grant number FENW-2023–0018).
Author information
Authors and Affiliations
Contributions
Guzenko V.V. contributed toward investigation. Bachurin S.S. contributed toward methodology, investigation, formal analysis, visualization, and writing—original draft. Khaitin A.M. contributed toward investigation, formal analysis, visualization, and writing—review & editing. Dzreyan V.A. contributed toward investigation. Kalyuzhnaya Y.N. contributed toward investigation. Demyanenko S.V. contributed toward investigation, visualization, project administration, supervision, conceptualization, methodology, funding acquisition, and writing—original draft.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guzenko, V.V., Bachurin, S.S., Dzreyan, V.A. et al. Acetylation of c-Myc at Lysine 148 Protects Neurons After Ischemia. Neuromol Med 26, 8 (2024). https://doi.org/10.1007/s12017-024-08777-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12017-024-08777-2