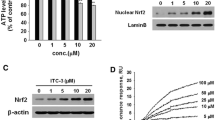
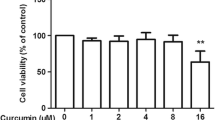
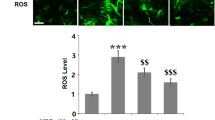
Abstract
Parkinson’s disease (PD) is a neurodegenerative disorder that results in motor impairment due to dopaminergic neuronal loss. The pathology of PD is closely associated with neuroinflammation, which can be characterized by astrocyte activation. Thus, targeting the inflammatory response in astrocytes might provide a novel therapeutic approach. We conducted a luciferase assay on an in-house chemical library to identify compounds with anti-inflammatory effects capable of reducing MPP+-induced NF-κB activity in astrocytes. Among the compounds identified, EI-16004, a novel 3-benzyl-N-phenyl-1H-pyrazole-5-carboxamides, exhibited a significant anti-inflammatory effect by significantly reducing MPP+-induced astrocyte activation. Biochemical analysis and docking simulation indicated that EI-16004 inhibited the MPP+-induced phosphorylation of p65 by attenuating ERK phosphorylation, and EI-16004 reduced pro-inflammatory cytokine and chemokine levels in astrocytes. In vivo studies on the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced PD model in male C57BL/6 mice showed that EI-16004 ameliorated motor impairment and protected against dopaminergic neuronal loss, and EI-16004 effectively mitigated the MPTP-induced astrocyte activation in striatum (STR) and substantia nigra (SN). These results indicate EI-16004 is a potential neuroprotective agent for the prevention and treatment of astrocyte-mediated neuroinflammatory conditions in PD.








Similar content being viewed by others
Data Availability
The data supporting the findings of this study can be made available by contacting the corresponding author upon a reasonable request.
References
Allaman, I., Bélanger, M., & Magistretti, P. J. (2011). Astrocyte-neuron metabolic relationships: For better and for worse. Trends in Neurosciences, 34(2), 76–87. https://doi.org/10.1016/j.tins.2010.12.001.
Cai, L., Gong, Q., Qi, L., Xu, T., Suo, Q., Li, X., et al. (2022). ACT001 attenuates microglia-mediated neuroinflammation after traumatic brain injury via inhibiting AKT/NFκB/NLRP3 pathway. Cell Communication and Signaling, 20(1), 1–23. https://doi.org/10.1186/s12964-022-00862-y.
Chi, H., Barry, S. P., Roth, R. J., Wu, J. J., Jones, E. A., Bennett, A. M., & Flavell, R. A. (2006). Dynamic regulation of pro- and anti-inflammatory cytokines by MAPK phosphatase 1 (MKP-1) in innate immune responses. Proc Natl Acad Sci U S A, 103(7), 2274–2279. https://doi.org/10.1073/pnas.0510965103.
Choi, J. H., Jang, M., Lee, J., Il, Chung, W. S., & Cho, I. H. (2018). Neuroprotective effects of a traditional multi-herbal medicine kyung-ok-ko in an animal model of parkinson’s disease: Inhibition of mapks and nf-kb pathways and activation of keap1-nrf2 pathway. Frontiers in Pharmacology, 9(December), 1–15. https://doi.org/10.3389/fphar.2018.01444.
Colucci-D’Amato, L., Perrone-Capano, C., & Di Porzio, U. (2003). Chronic activation of ERK and neurodegenerative diseases. Bioessays, 25(11), 1085–1095. https://doi.org/10.1002/bies.10355.
Fang, J. Y., & Richardson, B. C. (2005). The MAPK signalling pathways and colorectal cancer. Lancet Oncology, 6(5), 322–327. https://doi.org/10.1016/S1470-2045(05)70168-6.
Germann, U. A., Furey, B. F., Markland, W., Hoover, R. R., Aronov, A. M., Roix, J. J., et al. (2017). Targeting the MAPK signaling pathway in cancer: Promising preclinical activity with the novel selective ERK1/2 inhibitor BVD-523 (ulixertinib). Molecular Cancer Therapeutics, 16(11), 2351–2363. https://doi.org/10.1158/1535-7163.MCT-17-0456.
Ghose, A. K., Herbertz, T., Hudkins, R. L., Dorsey, B. D., & Mallamo, J. P. (2012). Knowledge-based, central nervous system (CNS) lead selection and lead optimization for CNS drug discovery. Acs Chemical Neuroscience, 3(1), 50–68. https://doi.org/10.1021/cn200100h.
Guo, Y., Pan, W., Liu, S., Shen, Z., Xu, Y., & Hu, L. (2020). ERK/MAPK signalling pathway and tumorigenesis (review). Exp Ther Med, 1997–2007. https://doi.org/10.3892/etm.2020.8454.
Hansson, E. (2010). Long-term pain, neuroinflammation and glial activation. Scand J Pain, 1(2), 67–72. https://doi.org/10.1016/j.sjpain.2010.01.002.
Hong, D. G., Lee, S., Kim, J., Yang, S., Lee, M., Ahn, J., et al. (2022). Anti-inflammatory and neuroprotective effects of Morin in an MPTP-Induced Parkinson’s Disease Model. International Journal of Molecular Sciences, 23(18). https://doi.org/10.3390/ijms231810578.
Jankovic, J. (2008). Parkinson’s disease: Clinical features and diagnosis. Journal of Neurology, Neurosurgery and Psychiatry, 79(4), 368–376. https://doi.org/10.1136/jnnp.2007.131045.
Jo, J., Lee, D., Park, Y. H., Choi, H., Han, J., Park, D. H., et al. (2021). Discovery and optimization of novel 3-benzyl-N-phenyl-1H-pyrazole-5-carboxamides as bifunctional antidiabetic agents stimulating both insulin secretion and glucose uptake. European Journal of Medicinal Chemistry, 217, 113325. https://doi.org/10.1016/j.ejmech.2021.113325.
Kaminska, B. (2005). MAPK signalling pathways as molecular targets for anti-inflammatory therapy - from molecular mechanisms to therapeutic benefits. Biochim Biophys Acta Proteins Proteom, 1754(1–2), 253–262. https://doi.org/10.1016/j.bbapap.2005.08.017.
Kempuraj, D., Thangavel, R., Natteru, P. A., Selvakumar, G. P., Saeed, D., Zahoor, H., et al. (2016). Neuroinflammation induces neurodegeneration. J Neurol Neurosurg Spine, 1(1), 1003.
Kirkley, K. S., Popichak, K. A., Hammond, S. L., Davies, C., Hunt, L., & Tjalkens, R. B. (2019). Genetic suppression of IKK2/NF-κB in astrocytes inhibits neuroinflammation and reduces neuronal loss in the MPTP-Probenecid model of Parkinson’s disease. Neurobiology of Diseases, 127(November 2018), 193–209. https://doi.org/10.1016/j.nbd.2019.02.020.
Koelblinger, P., Dornbierer, J., & Dummer, R. (2017). A review of binimetinib for the treatment of mutant cutaneous melanoma. Future Oncology, 13(20), 1–12. https://doi.org/10.2217/fon-2017-0170.
Kwon, H. S., & Koh, S. H. (2020). Neuroinflammation in neurodegenerative disorders: The roles of microglia and astrocytes. Transl Neurodegener, 9(1), 1–12. https://doi.org/10.1186/s40035-020-00221-2.
Latif, S., Jahangeer, M., Maknoon Razia, D., Ashiq, M., Ghaffar, A., Akram, M., et al. (2021). Dopamine in Parkinson’s disease. Clinica Chimica Acta. https://doi.org/10.1016/j.cca.2021.08.009.
Lee, Y., Lee, S., Chang, S. C., & Lee, J. (2019). Significant roles of neuroinflammation in Parkinson’s disease: Therapeutic targets for PD prevention. Archives of Pharmacal Research, 42(5), 416–425. https://doi.org/10.1007/s12272-019-01133-0.
Lee, S., Suh, Y. J., Lee, Y., Yang, S., Hong, D. G., Thirumalai, D., et al. (2021). Anti-inflammatory effects of the novel barbiturate derivative MHY2699 in an mptp‐induced mouse model of parkinson’s disease. Antioxidants, 10(11). https://doi.org/10.3390/antiox10111855.
Lee, S., Hong, D. G., Yang, S., Kim, J., Baek, M., Kim, S., et al. (2022). Anti-inflammatory effect of IKK‐Activated GSK‐3β inhibitory peptide prevented Nigrostriatal Neurodegeneration in the Rodent Model of Parkinson’s Disease. International Journal of Molecular Sciences, 23(2). https://doi.org/10.3390/ijms23020998.
Li, Q., & Barres, B. A. (2018). Microglia and macrophages in brain homeostasis and disease. Nature Reviews Immunology. https://doi.org/10.1038/nri.2017.125.
Li, Y., Chen, N., Wu, C., Lu, Y., Gao, G., Duan, C., et al. (2020). Galectin-1 attenuates neurodegeneration in Parkinson’s disease model by modulating microglial MAPK/IκB/NFκB axis through its carbohydrate-recognition domain. Brain, Behavior, and Immunity, 83, 214–225. https://doi.org/10.1016/j.bbi.2019.10.015.
Liu, T., Zhang, L., Joo, D., & Sun, S. C. (2017). NF-κB signaling in inflammation. Signal Transduct Target Ther, 2(April), https://doi.org/10.1038/sigtrans.2017.23.
Liu, B., Zhang, Y., Yang, Z., Liu, M., Zhang, C., Zhao, Y., & Song, C. (2021). ω-3 DPA protected neurons from Neuroinflammation by balancing Microglia M1/M2 polarizations through inhibiting NF-κB/MAPK p38 signaling and activating Neuron-BDNF-PI3K/AKT pathways. Marine Drugs, 19(11). https://doi.org/10.3390/md19110587.
Ma, X. L., Chen, C., & Yang, J. (2005). Predictive model of blood-brain barrier penetration of organic compounds. Acta Pharmacologica Sinica, 26(4), 500–512. https://doi.org/10.1111/j.1745-7254.2005.00068.x.
Madhi, I., Kim, J. H., Shin, J. E., & Kim, Y. (2021). Ginsenoside re exhibits neuroprotective effects by inhibiting neuroinflammation via CAMK/MAPK/NF-κB signaling in microglia. Molecular Medicine Reports, 24(4), 1–10. https://doi.org/10.3892/mmr.2021.12337.
Marsili, L., Marconi, R., & Colosimo, C. (2017). Treatment Strategies in Early Parkinson’s Disease. Int Rev Neurobiol (1st ed., Vol. 132). Elsevier Inc. https://doi.org/10.1016/bs.irn.2017.01.002.
Mebratu, Y., & Tesfaigzi, Y. (2009). How ERK1/2 activation controls cell proliferation and cell death is subcellular localization the answer? Cell Cycle, 8(8), 1168–1175.
Mishima, K., Inoue, K., & Hayashi, Y. (2002). Overexpression of extracellular-signal regulated kinases on oral squamous cell carcinoma. Oral Oncology, 38(5), 468–474. https://doi.org/10.1016/S1368-8375(01)00104-X.
Novak, M. L., & Koh, T. J. (2013). Macrophage phenotypes during tissue repair. Journal of Leukocyte Biology, 93(6). https://doi.org/10.1189/jlb.1012512.
Oeckinghaus, A., Hayden, M. S., & Ghosh, S. (2011). Crosstalk in NF-κB signaling pathways. Nature Immunology, 12(8), 695–708. https://doi.org/10.1038/ni.2065.
Ohori, M. (2008). ERK inhibitors as a potential new therapy for rheumatoid arthritis. Drug News & Perspectives, 21(5). https://doi.org/10.1358/DNP.2008.21.5.1219006.
Olah, M., Biber, K., Vinet, J., & Boddeke, W. G. M., H (2011). Microglia phenotype diversity. CNS Neurol Disord Drug Targets, 10(1). https://doi.org/10.2174/187152711794488575.
Orihuela, R., McPherson, C. A., & Harry, G. J. (2016). Microglial M1/M2 polarization and metabolic states. British Journal of Pharmacology. https://doi.org/10.1111/bph.13139.
Rai, S. N., Birla, H., Singh, S. S., Zahra, W., Patil, R. R., Jadhav, J. P., et al. (2017). Mucuna pruriens protects against MPTP intoxicated neuroinflammation in Parkinson’s disease through NF-κB/pAKT signaling pathways. Frontiers in Aging Neuroscience, 9(DEC), 1–14. https://doi.org/10.3389/fnagi.2017.00421.
Rai, S. N., Dilnashin, H., Birla, H., Singh, S. S., Zahra, W., Rathore, A. S., et al. (2019). The role of PI3K/Akt and ERK in Neurodegenerative disorders. Neurotoxicity Research, 35(3), 775–795. https://doi.org/10.1007/s12640-019-0003-y.
Shechter, R., Miller, O., Yovel, G., Rosenzweig, N., London, A., Ruckh, J., et al. (2013). Recruitment of beneficial M2 macrophages to injured spinal cord is orchestrated by Remote Brain Choroid Plexus. Immunity, 38(3). https://doi.org/10.1016/j.immuni.2013.02.012.
Sugiura, R., Satoh, R., & Takasaki, T. (2021). Erk: A double-edged sword in cancer. Erk-dependent apoptosis as a potential therapeutic strategy for cancer. Cells, 10(10). https://doi.org/10.3390/cells10102509.
Tang, Y., & Le, W. (2016). Differential roles of M1 and M2 microglia in neurodegenerative diseases. Molecular Neurobiology. https://doi.org/10.1007/s12035-014-9070-5.
Tran, K. A., Cheng, M. Y., Mitra, A., Ogawa, H., Shi, V. Y., Olney, L. P., et al. (2015). MEK inhibitors and their potential in the treatment of advanced melanoma: The advantages of combination therapy. Drug Design, Development and Therapy, 10, 43–52. https://doi.org/10.2147/DDDT.S93545.
Tysnes, O. B., & Storstein, A. (2017). Epidemiology of Parkinson’s disease. Journal of Neural Transmission (Vienna, Austria : 1996), 124(8), 901–905. https://doi.org/10.1007/s00702-017-1686-y.
Vila, M., Jackson-Lewis, V., Guégan, C., Wu, C., Teismann, D., Choi, P., D. K., et al. (2001). The role of glial cells in Parkinson’s disease. Current Opinion in Neurology, 14(4), 483–489. https://doi.org/10.1097/00019052-200108000-00009.
Wager, T. T., Chandrasekaran, R. Y., Hou, X., Troutman, M. D., Verhoest, P. R., Villalobos, A., & Will, Y. (2010). Defining desirable central nervous system drug space through the alignment of molecular properties, in vitro ADME, and safety attributes. Acs Chemical Neuroscience, 1(6), 420–434. https://doi.org/10.1021/cn100007x.
Yang, Q. qiao, & Zhou, J. (2019). Neuroinflammation in the central nervous system: Symphony of glial cells. Glia, 67(6), 1017–1035. https://doi.org/10.1002/glia.23571.
Funding
This work was supported by the National Research Foundation of Korea (NRF) (Grant no. NRF-2021R1A2C1010091) and by the 2022 BK21 FOUR Program of Pusan National University.
Author information
Authors and Affiliations
Contributions
Jaehoon Kim: Methodology, Validation, Formal analysis, Investigation, Writing-original draft, Visualization. Seulah Lee: Investigation, Visualization, Validation. Dong Geun Hong: Investigation, Visualization. Seonguk Yang: Investigation, Visualization. Cong So Tran: Resources. Jinsook Kwak: Resources. Min-Ju Kim: Software, Formal analysis, Validation. Thenmozhi Rajarathinam: Formal analysis, Resources. Ki Wung Chung: Data curation, Visualization. Young-Suk Jung: Data curation, Visualization. Akihito Ishigami: Data curation, Visualization. Seung-Cheol Chang: Data curation, Visualization. Haeseung Lee: Software, Data curation, Resources. Hwayoung Yun: Data curation, Resources. Jaewon Lee: Conceptualization, Writing-review & editing, Supervision, Project administration, Funding acquisition.
Corresponding author
Ethics declarations
Conflict of Interest
The authors have no potential conflict of interest to declare.
Ethics Approval
The animal protocol employed in this study underwent prior review and received approval from the Animal Care Committee of Pusan National University Institutional Animal Care Committee (PNU-IACUC) under approval number PNU-2021-0232.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kim, J., Lee, S., Hong, D.G. et al. Amelioration of Astrocyte-Mediated Neuroinflammation by EI-16004 Confers Neuroprotection in an MPTP-induced Parkinson’s Disease Model. Neuromol Med 26, 1 (2024). https://doi.org/10.1007/s12017-023-08769-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12017-023-08769-8