Abstract
Cerebral ischemic stroke is one of the foremost global causes of death and disability. Due to inadequate knowledge in its sequential disease mechanisms, therapeutic efforts to mitigate acute ischemia-induced brain injury are limited. Recent studies have implicated epigenetic mechanisms, mostly histone lysine acetylation/deacetylation, in ischemia-induced neural damage and death. However, the role of lysine methylation/demethylation, another prevalent epigenetic mechanism in cerebral ischemia has not undergone comprehensive investigation, except a few recent reports, including those from our research cohort. Considering the impact of sex on post-stroke outcomes, we studied both male and female mice to elucidate molecular details using our recently developed Internal Carotid Artery Occlusion (ICAO) model, which induces mild to moderate cerebral ischemia, primarily affecting the striatum and ventral hippocampus. Here, we demonstrate for the first time that female mice exhibit faster recovery than male mice following ICAO, evaluated through neurological deficit score and motor coordination assessment. Furthermore, our investigation unveiled that dysregulated histone lysine demethylases (KDMs), particularly kdm4b/jmjd2b are responsible for the sex-specific variance in the modulation of inflammatory genes. Building upon our prior reportage blocking KDMs by DMOG (Dimethyloxalylglycine) and thus preventing the attenuation in H3k9me2 reduced the post-ICAO transcript levels of the inflammatory molecules and neural damage, our present study delved into investigating the differential role of H3k9me2 in the regulation of pro-inflammatory genes in female vis-à-vis male mice underlying ICAO-induced neural damage and recovery. Overall, our results reveal the important role of epigenetic mark H3k9me2 in mediating sex-specific sequential events in inflammatory response, elicited post-ICAO.
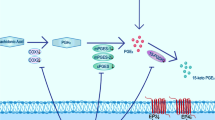
Graphical Abstract







Similar content being viewed by others
Data Availability
The data that support the findings are available with the corresponding author, may be available on reasonable request.
References
Aanerud, J., Borghammer, P., Rodell, A., Jonsdottir, K. Y., & Gjedde, A. (2017). Sex differences of human cortical blood flow and energy metabolism. Journal of Cerebral Blood Flow & Metabolism, 37(7), 2433–2440.
Abdu, H., & Seyoum, G. (2022). Sex differences in stroke risk factors, clinical profiles, and in-hospital outcomes among stroke patients admitted to the medical ward of dessie comprehensive specialized hospital, Northeast Ethiopia. Degenerative Neurological and Neuromuscular Disease, 12, 133–144.
Alisch, J. S. R., Khattar, N., Kim, R. W., Cortina, L. E., Rejimon, A. C., Qian, W., Ferrucci, L., Resnick, S. M., Spencer, R. G., & Bouhrara, M. (2021). Sex and age-related differences in cerebral blood flow investigated using pseudo-continuous arterial spin labeling magnetic resonance imaging. Aging (albany NY), 13(4), 4911.
Aspey, B. S., Taylor, F. L., Terruli, M., & Harrison, M. J. G. (2000). Temporary middle cerebral artery occlusion in the rat: Consistent protocol for a model of stroke and reperfusion. Neuropathology and Applied Neurobiology, 26(3), 232–242.
Bertogliat, M. J., Morris-Blanco, K. C., & Vemuganti, R. (2020). Epigenetic mechanisms of neurodegenerative diseases and acute brain injury. Neurochemistry International, 133, 104642.
Bouët, V., Freret, T., Toutain, J., Divoux, D., Boulouard, M., & Schumann-Bard, P. (2007). Sensorimotor and cognitive deficits after transient middle cerebral artery occlusion in the mouse. Experimental Neurology, 203(2), 555–567.
Chakravarty, S., Jhelum, P., Bhat, U. A., Rajan, W. D., Maitra, S., Pathak, S. S., Patel, A. B., & Kumar, A. (2017). Insights into the epigenetic mechanisms involving histone lysine methylation and demethylation in ischemia induced damage and repair has therapeutic implication. Biochimica Et Biophysica Acta Molecular Basis of Disease, 1863(1), 152–164.
Chen, W., Sun, Y., Liu, K., & Sun, X. (2014). Autophagy: A double-edged sword for neuronal survival after cerebral ischemia. Neural Regeneration Research, 9(12), 1210.
Chisholm, N. C., Henderson, M. L., Selvamani, A., Park, M. J., Dindot, S., Miranda, R. C., & Sohrabji, F. (2015). Histone methylation patterns in astrocytes are influenced by age following ischemia. Epigenetics, 10(2), 142–152.
Covington, H. E., Maze, I., Sun, H. S., Bomze, H. M., DeMaio, K. D., Wu, E. Y., Dietz, D. M., et al. (2011). A role for repressive histone methylation in cocaine-induced vulnerability to stress. Neuron, 71(4), 656–670.
Das, T., Soren, K., Yerasi, M., Kumar, A., & Chakravarty, S. (2019). Revealing sex-specific molecular changes in hypoxia-ischemia induced neural damage and subsequent recovery using Zebrafish model. Neuroscience Letters, 712, 134492.
Dell’Aversana, C., Lepore, I., & Altucci, L. (2012). HDAC modulation and cell death in the clinic. Experimental Cell Research, 318(11), 1229–1244.
Doerfler, W. (2008). In pursuit of the first recognized epigenetic signal––DNA methylation: A 1976 to 2008 synopsis. Epigenetics, 3(3), 125–133.
Donnan, G. A., Fisher, M., Macleod, M., & Davis, S. M. (2008). Stroke. Lancet, 371(9624), 1612–1623.
Dyken, M. L., Klatte, E., Kolar, O. J., & Spurgeon, C. (1974). Complete occlusion of common or internal carotid arteries: Clinical significance. Archives of Neurology, 30(5), 343–346.
Endres, M., Meisel, A., Biniszkiewicz, D., Namura, S., Prass, K., Ruscher, K., Lipski, A., Jaenisch, R., Moskowitz, M. A., & Dirnagl, U. (2000). DNA methyltransferase contributes to delayed ischemic brain injury. Journal of Neuroscience, 20(9), 3175–3181.
Faraco, G., Pancani, T., Formentini, L., Mascagni, P., Fossati, G., Leoni, F., Moroni, F., & Chiarugi, A. (2006). Pharmacological inhibition of histone deacetylases by suberoylanilide hydroxamic acid specifically alters gene expression and reduces ischemic injury in the mouse brain. Molecular Pharmacology, 70(6), 1876–1884.
Fisher, M. (1954). Occlusion of the carotid arteries: Further experiences. AMA Archives of Neurology & Psychiatry, 72(2), 187–204.
Flaherty, M. L., Flemming, K. D., McClelland, R., Jorgensen, N. W., & Brown, R. D. (2004). Population-based study of symptomatic internal carotid artery occlusion: Incidence and long-term follow-up. Stroke, 35(8), e349–e352.
Formisano, L., Noh, K.-M., Miyawaki, T., Mashiko, T., Bennett, M. V. L., & Suzanne Zukin, R. (2007). Ischemic insults promote epigenetic reprogramming of μ opioid receptor expression in hippocampal neurons. Proceedings of the National Academy of Sciences United State of America, 104(10), 4170–4175.
Harder, D. R., Alkayed, N. J., Lange, A. R., Gebremedhin, D., & Roman, R. J. (1998). Functional hyperemia in the brain: Hypothesis for astrocyte-derived vasodilator metabolites. Stroke, 29(1), 229–234.
Hebbes, T. R., Thorne, A. W., & Crane-Robinson, C. (1988). A direct link between core histone acetylation and transcriptionally active chromatin. The EMBO Journal, 7(5), 1395–1402.
Jayaraj, R. L., Azimullah, S., Beiram, R., Jalal, F. Y., & Rosenberg, G. A. (2019). Neuroinflammation: Friend and foe for ischemic stroke. Journal of Neuroinflammation, 16(1), 1–24.
Jhelum, P., Mydhili Radhakrishnan, A. R., Paul, S., Dey, S. K., Kamle, A., Kumar, A., Sharma, A., & Chakravarty, S. (2022). Neuroprotective and proneurogenic effects of glucosamine in an internal carotid artery occlusion model of ischemia. NeuroMolecular Medicine. https://doi.org/10.1007/s12017-021-08697-5
Jhelum, P., Wahul, A. B., Kamle, A., Kumawat, S., Kumar, A., Bhutani, K. K., Tripathi, S. M., & Chakravarty, S. (2017). Sameerpannag ras mixture (SRM) improved neurobehavioral deficits following acute ischemic stroke by attenuating neuroinflammatory response. Journal of Ethnopharmacology, 197, 147–156.
Jørgensen, H. S., Nakayama, H., Raaschou, H. O., & Olsen, T. S. (1999). Stroke: Neurologic and functional recovery the Copenhagen stroke study. Physical Medicine and Rehabilitation Clinics of North America, 10(4), 887–906.
Karisetty, B. C., Joshi, P. C., Kumar, A., & Chakravarty, S. (2017). Sex differences in the effect of chronic mild stress on mouse prefrontal cortical BDNF levels: A role of major ovarian hormones. Neuroscience, 356, 89–101.
Kim, H. J., Rowe, M., Ren, M., Hong, J.-S., Chen, P.-S., & Chuang, D.-M. (2007). Histone deacetylase inhibitors exhibit anti-inflammatory and neuroprotective effects in a rat permanent ischemic model of stroke: Multiple mechanisms of action. Journal of Pharmacology and Experimental Therapeutics, 321(3), 892–901.
Klöting, I., Nitschke, C., & van den Brandt, J. (2003). Impact of genetic profiles on experimental studies: Outbred versus wild rats. Toxicology and Applied Pharmacology, 189(1), 68–71.
Krupinski, J., Carrera, C., Muiño, E., Torres, N., Al-Baradie, R., Cullell, N., & Fernandez-Cadenas, I. (2018). DNA methylation in stroke. Update of latest advances. Computational and Structural Biotechnology Journal, 16, 1–5.
Kumar, A., Choi, K.-H., Renthal, W., Tsankova, N. M., Theobald, D. E. H., Truong, H.-T., Russo, S. J., et al. (2005). Chromatin remodeling is a key mechanism underlying cocaine-induced plasticity in striatum. Neuron, 48(2), 303–314.
Leng, X., Fang, H., Leung, T. W. H., Mao, C., Miao, Z., Liu, L., Wong, K. S., & Liebeskind, D. S. (2016). Impact of collaterals on the efficacy and safety of endovascular treatment in acute ischaemic stroke: A systematic review and meta-analysis. Journal of Neurology, Neurosurgery & Psychiatry, 87(5), 537–544.
Lu, X., Jian Zhang, Y., Ding, J. W., & Chen, G. (2022). Novel therapeutic strategies for ischemic stroke: Recent insights into autophagy. Oxidative Medicine and Cellular Longevity, 2022, 1–15.
Manwani, B., Bentivegna, K., Benashski, S. E., Venna, V. R., Yan, X., Arnold, A. P., & McCullough, L. D. (2015). Sex differences in ischemic stroke sensitivity are influenced by gonadal hormones, not by sex chromosome complement. Journal of Cerebral Blood Flow & Metabolism, 35(2), 221–229.
Marks, P. A. (2010). Histone deacetylase inhibitors: a chemical genetics approach to understanding cellular functions. Biochimica Et Biophysica Acta Gene Regulatory Mechanisms, 1799, 717–725.
McAuley, M. A. (1995). Rodent models of focal ischemia. Cerebrovascular and Brain Metabolism Reviews, 7(2), 153–180.
Mead, G. E., Shingler, H., Farrell, A., O’neill, P. A., & Mccollum, C. N. (1998). Carotid disease in acute stroke. Age and Ageing, 27(6), 677–682.
Mhairi, M. I. (1992). New models of focal cerebral ischaemia. British Journal of Clinical Pharmacology, 34(4), 302–308.
Moskowitz, M. A., Lo, E. H., & Iadecola, C. (2010). The science of stroke: Mechanisms in search of treatments. Neuron, 67(2), 181–198.
Narlikar, G. J., Fan, H.-Y., & Kingston, R. E. (2002). Cooperation between complexes that regulate chromatin structure and transcription. Cell, 108(4), 475–487.
Patrizz, A. N., Moruno-Manchon, J. F., O’Keefe, L. M., Doran, S. J., Patel, A. R., Venna, V. R., Tsvetkov, A. S., Li, J., & McCullough, L. D. (2021). Sex-specific differences in autophagic responses to experimental ischemic stroke. Cells, 10(7), 1825.
Ren, M., Leng, Y., Jeong, MiRa., Leeds, P. R., & Chuang, D.-M. (2004). Valproic acid reduces brain damage induced by transient focal cerebral ischemia in rats: Potential roles of histone deacetylase inhibition and heat shock protein induction. Journal of Neurochemistry, 89(6), 1358–1367.
Roy-O’Reilly, M., & McCullough, L. D. (2018). Age and sex are critical factors in ischemic stroke pathology. Endocrinology, 159(8), 3120–3131.
Schweizer, S., Harms, C., Lerch, H., Flynn, J., Hecht, J., Yildirim, F., Meisel, A., & Märschenz, S. (2015). Inhibition of histone methyltransferases SUV39H1 and G9a leads to neuroprotection in an in vitro model of cerebral ischemia. Journal of Cerebral Blood Flow & Metabolism, 35(10), 1640–1647.
Shigeno, T., Teasdale, G. M., McCulloch, J., & Graham, D. I. (1985). Recirculation model following MCA occlusion in rats: Cerebral blood flow, cerebrovascular permeability, and brain edema. Journal of Neurosurgery, 63(2), 272–277.
Song, T.-J., Jeong, Y., Park, J., & Gwak, H. S. (2020). Risk factors and prevention of stroke. Cerebrovascular Diseases, 49(1), 1–149.
Stenzel-Poore, M. P., Stevens, S. L., King, J. S., & Simon, R. P. (2007). Preconditioning reprograms the response to ischemic injury and primes the emergence of unique endogenous neuroprotective phenotypes: A speculative synthesis. Stroke, 38(2), 680–685.
Sudlow, C. L. M., & Warlow, C. P. (1997). Comparable studies of the incidence of stroke and its pathological types: Results from an international collaboration. Stroke, 28(3), 491–499.
Sundt, T. M., Grant, W. C., & Garcia, J. H. (1969). Restoration of middle cerebral artery flow in experimental infarction. Journal of Neurosurgery, 31(3), 311–322.
Szadvári, I., Ostatníková, D., & Durdiaková, J. B. (2023). Sex differences matter: Males and females are equal but not the same. Physiology & Behavior, 259, 114038.
Tamura, A., Graham, D. I., McCulloch, J., & Teasdale, G. M. (1981). Focal cerebral ischaemia in the rat: Description of technique and early neuropathological consequences following middle cerebral artery occlusion. Journal of Cerebral Blood Flow & Metabolism, 1(1), 53–60.
Ten, V., & Galkin, A. (2019). Mechanism of mitochondrial complex I damage in brain ischemia/reperfusion injury: A hypothesis. Molecular and Cellular Neuroscience, 100, 103408.
Thanvi, B., & Robinson, T. (2007). Complete occlusion of extracranial internal carotid artery: Clinical features, pathophysiology, diagnosis and management. Postgraduate Medical Journal, 83(976), 95–99.
Thiele, B. L., Young, J. V., Chikos, P. M., Hirsch, J. H., & Strandness, D. E. (1980). Correlation of arteriographic findings and symptoms in cerebrovascular disease. Neurology, 30(10), 1041–1041.
Trotman-Lucas, M., & Gibson, C. L. (2021). A review of experimental models of focal cerebral ischemia focusing on the middle cerebral artery occlusion model. F1000Research, 10, 242.
Tsankova, N. M., Berton, O., Renthal, W., Kumar, A., Neve, R. L., & Nestler, E. J. (2006). Sustained hippocampal chromatin regulation in a mouse model of depression and antidepressant action. Nature Neuroscience, 9(4), 519–525.
Tsankova, N. M., Kumar, A., & Nestler, E. J. (2004). Histone modifications at gene promoter regions in rat hippocampus after acute and chronic electroconvulsive seizures. Journal of Neuroscience, 24(24), 5603–5610.
Tsankova, N., Renthal, W., Kumar, A., & Nestler, E. J. (2007). Epigenetic regulation in psychiatric disorders. Nature Reviews Neuroscience, 8(5), 355–367.
Wahul, A. B., Joshi, P. C., Kumar, A., & Chakravarty, S. (2018). Transient global cerebral ischemia differentially affects cortex, striatum and hippocampus in bilateral common carotid arterial occlusion (BCCAo) mouse model. Journal of Chemical Neuroanatomy, 92, 1–15.
Wilkinson, M. B., Xiao, G., Kumar, A., LaPlant, Q., Renthal, W., Sikder, D., Kodadek, T. J., & Nestler, E. J. (2009). Imipramine treatment and resiliency exhibit similar chromatin regulation in the mouse nucleus accumbens in depression models. Journal of Neuroscience, 29(24), 7820–7832.
Zampino, M., Yuzhakova, M., Hansen, J., McKinney, R. D., Goldspink, P. H., Geenen, D. L., & Buttrick, P. M. (2006). Sex-related dimorphic response of HIF-1α expression in myocardial ischemia. American Journal of Physiology-Heart and Circulatory Physiology, 291(2), H957–H964.
Zhang, Y.-Z., Zhang, Q.-H., Ye, H., Zhang, Y., Luo, Y.-M., Ji, X.-M., & Ying-Ying, Su. (2010). Distribution of lysine-specific demethylase 1 in the brain of rat and its response in transient global cerebral ischemia. Neuroscience Research, 68(1), 66–72.
Zhao, H., Han, Z., Ji, X., & Luo, Y. (2016). Epigenetic regulation of oxidative stress in ischemic stroke. Aging and Disease, 7(3), 295.
Zhong, X., Sun, Y., Yajun, Lu., & Lei, Xu. (2023). Immunomodulatory role of estrogen in ischemic stroke: Neuroinflammation and effect of sex. Frontiers in Immunology, 14, 1164258.
Zwemer, C. F., Song, M. Y., Carello, K. A., & D’Alecy, L. G. (2007). Strain differences in response to acute hypoxia: CD-1 versus C57BL/6J mice. Journal of Applied Physiology, 102(1), 286–293.
Acknowledgements
This research was initiated by the Council of Scientific and Industrial Research (CSIR), India network projects [(BSC0103-UNDO) to SC and AK] and later supported by ICMR grant [5/4-5/3/17/Neuro/2022-NCD-1 to SC]. MR, VV, PBT, SP and KS wish to acknowledge CSIR India, Department of Biotechnology (DBT) and DST-INSPIRE for their doctoral fellowships, respectively. In addition, the authors would like to specially acknowledge B. Jyothilakshmi of the Centre for Cellular and Molecular Biology (CCMB), Hyderabad, for the maintenance and care of animals throughout the study period. KIM Department of CSIR-IICT is greatly acknowledged for generating institutional publication number IICT/Pubs/2023/186.
Author information
Authors and Affiliations
Contributions
SC and AK conceived the study. SC and AK designed and supervised all the experiments. The experiments performed by MR, VV, SA, PB, SP, KS, and SC, SC and AK arranged the resources and analyzed the results. MR, SC, and AK. wrote the original draft of the manuscript and made the edits to make the final version. All authors reviewed and edited the manuscript for their part.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Radhakrishnan, M., Vijay, V., Supraja Acharya, B. et al. Uncovering Sex-Specific Epigenetic Regulatory Mechanism Involving H3k9me2 in Neural Inflammation, Damage, and Recovery in the Internal Carotid Artery Occlusion Mouse Model. Neuromol Med 26, 3 (2024). https://doi.org/10.1007/s12017-023-08768-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12017-023-08768-9