Abstract
Non-specific innate and antigen-specific adaptive immunological memories are vital evolutionary adaptations that confer long-lasting protection against a wide range of pathogens. Adaptive memory is established by memory T and B lymphocytes following the recognition of an antigen. On the other hand, innate immune memory, also called trained immunity, is imprinted in innate cells such as macrophages and natural killer cells through epigenetic and metabolic reprogramming. However, these mechanisms of memory generation and maintenance are compromised as organisms age. Almost all immune cell types, both mature cells and their progenitors, go through age-related changes concerning numbers and functions. The aging immune system renders the elderly highly susceptible to infections and incapable of mounting a proper immune response upon vaccinations. Besides the increased infectious burden, older individuals also have heightened risks of metabolic and neurodegenerative diseases, which have an immunological component. This review discusses how immune function, particularly the establishment and maintenance of innate and adaptive immunological memory, regulates and is regulated by epigenetics, metabolic processes, gut microbiota, and the central nervous system throughout life, with a focus on old age. We explain in-depth how epigenetics and cellular metabolism impact immune cell function and contribute or resist the aging process. Microbiota is intimately linked with the immune system of the human host, and therefore, plays an important role in immunological memory during both homeostasis and aging. The brain, which is not an immune-isolated organ despite former opinion, interacts with the peripheral immune cells, and the aging of both systems influences the health of each other. With all these in mind, we aimed to present a comprehensive view of the aging immune system and its consequences, especially in terms of immunological memory. The review also details the mechanisms of promising anti-aging interventions and highlights a few, namely, caloric restriction, physical exercise, metformin, and resveratrol, that impact multiple facets of the aging process, including the regulation of innate and adaptive immune memory. We propose that understanding aging as a complex phenomenon, with the immune system at the center role interacting with all the other tissues and systems, would allow for more effective anti-aging strategies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Human beings, like all organisms, inevitably age and die. Even if science eventually cracked the code for immortality, that would not end the need to understand the mechanisms of aging and the efforts to slow or revert it. If anything, it will be even more critical to maintain the health of all cells and organs throughout a long life. Tackling aging is always a worthwhile effort to improve the quality of live for the middle-aged and elderly populations, especially since the human population over 60 years of age is expected to reach two billion by 2050 [1].
Infectious diseases of the elderly, especially in low-income countries, represent a significant social and economic burden. The immune system undergoes numerous changes as humans age, leaving older individuals more prone to disease [2]. The age-related dysregulations in the immune system are collectively referred to as “immunosenescence” and include accumulating tissue damage, a low-grade chronic systemic inflammation termed “inflammaging,” impaired immune cell function, inadequate response to vaccination, and increased vulnerability to infections [3].
The importance of immune memory has perhaps never been more evident than during the ongoing coronavirus disease 2019 (COVID-19) pandemic, which disproportionately affected the elderly population due to the altered functionality of their immune system [4]. Thanks to the outstanding collaborative effort of governments and scientists, 7 vaccines generating effective immune response and protection against the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) have been authorized for emergency use by World Health Organization (WHO)–recognized authorities as of June 2021, and many more are in use with authorizations by national regulatory agencies [5]. Due to the increased vulnerability of the elderly, they are the priority group in COVID-19 vaccination rollouts.
Besides the morbidities caused by infection, the elderly also present an increased incidence of metabolic diseases such as type 2 diabetes and obesity [6], and neurodegenerative disorders such as Alzheimer’s and Parkinson’s diseases [7]. However, the development of these age-related conditions is not separated from their aging immunity. All systems and organs exchange signals with and are influenced by the immune system. Combining all the accumulating insights from different lines of research is critical to drawing up a comprehensive view of aging.
In this review focusing on immune memory, we first outline how memory is developed and maintained. Next, we delve into metabolic and epigenetic mechanisms, their roles in immune memory, how they change with age, and the implications for age-related pathologies. As two examples of the far-reaching impacts of an aging immune system, we highlight the interplay of immune memory with the gut microbiota and the brain. We end the review by presenting the current preventative and therapeutic strategies against aging, approaching from the alternative points of view of epigenetic modulation, metabolic intervention, microbiota reconstitution, and neuroprotection.
Adaptive Immune Memory
Infections have been one of the primary selective forces throughout evolution, so immunological memory has evolved to ensure survival when an organism is exposed to a pathogen that it encountered before [8]. Until the discovery of non-specific innate immune memory in the last decade, the antigen-specific memory established by T and B lymphocytes have been getting all the credit for long-term protection against pathogens.
T Cells: Thymus-Derived Troops of Immunity
Immunological memory against infections and tumors requires the intervention of T cells. T cells can recognize both self and non-self antigens through their T cell receptors (TCRs) and mount self-tolerance or immunological memory. Different subsets of T cells include naïve T cells that recognize new antigens and memory T cells that are formed upon former exposure to antigen and assure long-lasting immunity.
T Cell Development
T cells derive from the hematopoietic stem cells (HSCs) in the bone marrow but mature in the thymus. Most mature T cells reside in lymphoid tissues, but they are ubiquitously present throughout the body. After lymphoid progenitors migrate from bone marrow to the thymus, TCR gene rearrangement occurs, and CD4+ CD8+ double-positive cells expressing both co-receptors are generated. Then, these cells undergo positive selection based on TCR-antigen interactions and differentiate into naïve single positive CD4+ helper or CD8+ cytotoxic T cells, which are released into the periphery [9].
Most of our knowledge on T cell development originates from mouse studies. However, there are substantial differences between mice and humans. For instance, although the peripheral naïve T cell pool is almost exclusively provided by the thymus in mice, humans primarily sustain it by peripheral cell division [10].
When a naïve cell recognizes an antigen presented by antigen-presenting cells (APCs) such as dendritic cells (DCs) and macrophages, they proliferate and develop into effector cells that can clear the source of the antigen, likely a pathogen. A small portion of these effector cells later become memory cells to establish long-term immunity that can last multiple decades, while the rest die by apoptosis [11]. Early in life, before exposure to many antigens, naïve T cells constitute most of the T cell pool [12]. Meanwhile, regulatory T cells (Treg) are critical for the development of tolerance for innocuous antigens in the environment [13].
Around 5% of all adult CD4+ T cells are Tregs that are able to suppress the immune response [12]. Tregs are produced in the thymus but can also derive from peripheral naïve T cells by acquiring Forkhead Box P3 (FOXP3) expression in response to environmental cues [13]. Recently, Tregs were shown to acquire memory characteristics, mostly against self-antigens, to prevent unwanted inflammation [14].
Memory T cells are divided into three subtypes which are central memory (TCM), effector memory (TEM), and stem cell memory (TSCM). Compared to TEMs, TCMs have more proliferation capacity and are closer to naïve T cells in gene expression profiles [15]. TEMs can perform effector functions such as cytokine production. TSCMs are a stem cell-like, less differentiated cell type with high self-renewal capacity and the ability to differentiate into effector T cells, TEMs, or TCMs [16]. Following TCR stimulation, they are able to secrete interferon gamma (IFN-γ) and interleukin 2 (IL-2). The long-lasting, multipotent TSCMs might help protect the organism against infections later in life when thymic output is low.
Although 90–95% of the effector T cells die after an infection resolves, a population of terminally differentiated effector cells regaining the naïve T cell marker CD45RA, termed TEMRA cells, remain in circulation. These senescent-like cells have defects in telomerase expression and proliferation; however, they are capable of cytokine production and cytotoxicity, unlike exhausted cells [17].
In many tissues such as lungs, intestines, and spleen, TEMs are the predominant T cell type [18, 19]. Moreover, discrete tissue-resident memory T cell populations (TRM) are identified with enhanced expression of adhesion markers and homing receptors, lower proliferative capacity, and higher production ability of pro-inflammatory and anti-inflammatory cytokines [20]. They can quickly react upon tissue injury or infection while also restricting the inflammatory damage. Establishing TRMs is a promising approach to consider in vaccine design, boosting and prolonging vaccine-mediated protection [21,22,23,24].
Effects of Aging on T Cells
Lineage differentiation dynamics of HSCs in the bone marrow are altered with age. They skew towards myeloid differentiation, leading to lower numbers of lymphoid cells in the elderly [25]. HSCs also accumulate DNA damage throughout life and differentiate into leukocytes with chronic DNA damage response [26]. This triggers cellular senescence, which contributes to chronic inflammation by inducing a senescence-associated secretory phenotype (SASP), impacting neighboring immune and non-immune cell types. Another way that DNA damage can contribute to inflammation is the activation of DNA-dependent protein kinase catalytic subunits (DNA-PKcs) that can promote NFκB and inflammasome activity [27, 28].
Involution of the thymus is one of the critical age-dependent changes in the immune system [29]. It is an evolutionarily conserved phenomenon in all vertebrates, starting before puberty, where the total mass, volume, and cellular content of the thymus shrink [30]. Thymic activity does not entirely cease, at least until the sixth decade of life, but thymopoiesis strikingly decreases with age [31, 32]. Thymic epithelial cells gradually lose the ability to produce IL-7, which is crucial to support thymopoiesis [33, 34]. Low thymic output in the elderly is associated with increased vulnerability to infections [35]. In a young adult, the thymus provides around 16% of the naïve T cell pool, the rest of which derives from peripheral proliferation [36]. In the elderly, this number falls below 1%, causing them to entirely rely on the proliferation of existing naïve T cells.
The decline in the number of naïve T cells and accumulation of terminally differentiated cells are two of the hallmarks of T cell aging [36]. CD4+ and CD8+ naïve cell pools, although more markedly for CD8+ T cells, contract in the elderly. Maintenance of naïve T cells through peripheral proliferation is more successful for CD4+ T cells, but CD8+ T cells are largely lost. Interestingly, while this is mostly the case in cytomegalovirus (CMV) + individuals in women, it is observed in men irrespective of the CMV status [37]. Also, CMV + individuals of both sexes have a higher proportion of late-differentiated senescent T cells than CMV individuals.
Chronic CMV infection affects most adults, with an 83% global seroprevalence rate [38]. Even though it usually does not cause active symptoms and is mainly unrecognized, CMV presence significantly shapes the T cell compartments and accelerates immunosenescence. Accumulation of terminally differentiated T cell types such as TEMs and TEMRAs occurs faster in CMV + individuals throughout their lifespan [39]. Expansion of CD8+ TEMRA cells is related to impaired antibody production upon influenza vaccination in the elderly [40]. Latent CMV infection is also associated with inadequate CD4+ T cell response against influenza antigens [41]. Moreover, CMV positivity is associated with a higher risk of all-cause mortality [42]. Of note, CMV + young adults displayed higher antibody responses to influenza vaccination, compared to CMV − young individuals [43]. In the early stages of the infection, CMV might be potentiating immune responses before the accumulation of CMV-induced senescent cells pass a certain threshold and causes functional impairments.
Not just the numbers but also the receptor diversity of naïve T cells are compromised in aged organisms. Naïve T cells of a young adult carry around 100 million different TCR sequences; however, this repertoire diversity is reduced up to tenfold in the elderly [44]. Moreover, memory T cells experience a narrowing of TCR repertoires [45], and the proliferative capacity of senescent T cells following TCR engagement is defective [46]. Activated CD8+ cells of elderly individuals also produce lower levels of cytotoxins such as granzyme B and perforin [47]. On the other hand, CD4+ naïve T cells of the elderly seem to maintain their differentiation and subsequent cytokine production capacities [48].
Lastly, differentiation of non-Treg cells into Tregs and proliferation of existing Tregs can maintain the Treg pools throughout life, despite reduced thymic output with aging. However, the balance between T cell subsets is altered: as in other T cell types, the naïve subset declines with age while memory Tregs increase [49].
B Cells: Bone Marrow-Born Battlers
B cells are a vital part of the adaptive immune memory. They have several immunological functions, including antibody and cytokine production, antigen presentation, and regulation of T cell responses [50]. Most vaccines mainly target and rely on B cell activation by inducing long-lived plasma and memory B cell proliferation[51]. However, aging affects the functional capacity of existing B cell subsets drastically, which is evident from the susceptibility to diseases and poor vaccine responses [52].
B Cell Development
B cells continuously arise from the hematopoietic stem cells (HSCs) and develop in the bone marrow (BM) [53]. HSCs generate multipotent progenitors that eventually diverge to common lymphoid progenitors (CLPs). Certain environmental cues, transcription factors (TFs), cytokines, and chemokines lead CLPs to differentiate into B-cell lineage. Following differentiation, cells undergo a rearrangement in the variable regions of the immunoglobulin (Ig) genes and start to express B-cell receptors (BCRs) and IL-7 receptor (IL-7R) [54]. Each B cell has a unique BCR with a different specificity to antigens.
B cells that finish their developmental process in the bone marrow are called transitional (TR) B cells. They make 4% of all B lymphocytes in healthy individuals [55] and are found in several places, including the bone marrow, peripheral blood, and secondary lymphoid tissues. Transitional B cells become either marginal zone (MZ) or mature follicular (FO) cells partly based on the strength of their BCR signaling. Cells with more robust signaling tend to develop into follicular type, while weaker signaling drives them to be MZ cells [56]. FO B cells have a broad immunoglobulin repertoire and are located in the follicles close to T cell zones [57]. Therefore, they are suited for getting T-cell help and becoming short-lived plasma cells. On the other hand, MZ B cells can get activated easier than FO B cells, which quickly allow them to produce immunoglobulin M (IgM) or induce class switching without T-cell help [58].
The third naïve B cell subset is B-1 cells, which are considered part of the innate immune system [59, 60]. Apart from the other B cell subsets developed in the bone marrow, B-1 B cells originate from a distinct progenitor in the fetal bone marrow [61]. They are mainly found in peritoneal and pleural cavities; however, low numbers can also be located in secondary lymphoid organs. During an infection, they act by producing non-specific antibodies that are crucial for early defense [62, 63].
Advancing age alters the entire course of B cell development, the abundance of distinct B cell subsets, and their function. Furthermore, a B cell subset emerging with increasing age influences immune responses in the elderly.
Effects of Aging on B Cell Development
B cell development and the influence of old age in this process are extensively studied in mice. First of all, the differentiation capacity of long-term HSCs (LT-HSCs) reduces with advanced age [64]. The genes driving lymphoid cell differentiation and function are downregulated in LT-HSCs, while the genes mediating myeloid cell development are upregulated. Numbers and percentages of early B-cell lineage progenitors decrease as C57BL/6 mice age [65]. Furthermore, these populations exhibit declined IL-7 responsiveness, indicating an impaired B lymphopoiesis.
Following progenitor differentiation, the development of B cells in the bone marrow is also influenced by aging. In different groups of old mice, a severe decrease with more than 80% loss of pre-B cells and 50% loss of pro-B cells, or a moderate decrease with 20–80% loss of pre-B cells were observed [66]. TFs regulating B cell development are altered by age, influencing the abundance of developing B cells [66,67,68]. Among them, the E2A gene encodes for two proteins, E47 and E12. Transcription and DNA-binding capacity of E47 were shown to decline in aged mice [66]. As E47 is a vital TF in B cell development during the pro- to pre-B cell stage [69], lower numbers of pre- and pro-B cells in old mice could partly be explained by the decreased function and expression of E47. PAX5 is another TF regulating early B-cell development that is lower in the elderly [70]. Lastly, BCR expression and diversity are altered upon aging [71, 72], although a study suggested that the changes were not evident until 70 years of age [73].
The Emergence of Age-Associated B Cells
In 2011, a new subset of B cells was described in aged mice [74, 75]. This mature B cell population is named age-associated B cells (ABCs) since it progressively accumulates with increasing age. The origins of ABCs are not exactly known; however, differentiated FO, MZ, and B-1 cells are thought to contribute to the heterogeneous ABC pool [76]. Although studies define ABCs using different markers, they agree that ABCs are mature B cells with memory characteristics. Unlike the other B cell subtypes, ABCs express the transcription factor T-bet and a unique surface marker combination [77]. Therefore, their activation requirements, functions, and survival conditions are remarkably different. BCR engagement induces FO and MZ B cell proliferation, while Toll-like receptor 9 (TLR9) or TLR7 signaling with or without BCR ligation drives proliferation in ABCs [76]. In vitro studies showed that TLR stimulation leads to IL-10 and IFNγ production from ABCs, and an in vivo study reported that they also produce tumor necrosis factor alpha (TNFα) [78].
ABCs are engaged in both protective and autoreactive immune responses, although their protective role seems scarce. Furthermore, they are linked with autoinflammatory and autoimmune diseases, such as systemic lupus erythematosus and rheumatoid arthritis [75, 79, 80], making ABCs a potential underlying reason for the increased incidence of autoimmune diseases in the elderly.
ABCs contribute to immune dysfunctions observed during the aging process. For instance, TNFα produced by ABCs has direct and indirect effects on pro-B cell numbers: ABCs directly induce pro-B cell apoptosis and lead to their loss by altering the bone marrow microenvironment [78]. Besides, increased abundance of ABCs was significantly correlated with the loss of B cell precursors in the bone marrow of aged mice.
ABCs express considerably high major histocompatibility complex II (MHC-II), CD80, and CD86 compared to FO B cells; therefore, they are better inducers of T cell activation and antigen presentation [81]. However, the same study associated these properties of ABCs with autoimmune diseases in an autoimmune-prone mice strain. Besides, considering that they make the bone marrow environment more inflammatory via the production of TNFα and robustly produce IL-6 and IFNγ upon TLR7 and TLR9 engagement [74, 78], it is plausible to propose that ABCs contribute to inflammaging.
Lastly, a study reported that humoral response depends more on TLR signaling and less on CD4+ T cell help due to decreased FO B cells and increased ABCs in aged mice [82]. This eventually resulted in impaired production of IgG and long-lived plasma cells.
Abundance and Functions of B Cells in the Elderly
Several studies reported a decrease of mature B cell subsets in humans with aging, although the extent of these changes varies depending on the subsets, experimental approaches, and cohorts of people [53, 83, 84]. For instance, Muggen et al. reported that numbers and relative abundance of several B cell subsets including transitional B cells, memory cells, and plasmablasts reduced with aging, particularly in individuals older than 70 years old [73]. Plasma and memory B cell percentages in the circulation and bone marrow decline, while naïve and immature B cells remain relatively stable in older people [85]. The abundance of B-1 cells, along with their ability to produce IgM, decreases with age [63]. A study found significantly low switched memory B cells, but high naïve and double-negative memory B cells in people over 65 years of age compared to younger adults [86]. The authors concluded that double-negative or so-called late-exhausted memory B cells express senescence markers and are associated with poor immune responses against influenza vaccine. Of note, switched memory B cells play a role in antibody production upon re-infection, generating a rapid response compared to naïve B cells [84]; therefore, a lower abundance of switched memory B cells is another evidence of impaired humoral immune response in the elderly.
Not only the numbers but also functions of B cells are diminished with aging. Poor antibody responses in the elderly after influenza vaccination are due to low binding and neutralization capacity of antibodies, decreased class switch recombination, hypermutations of the antibody variable regions, and higher abundance of inflammatory B cells [87, 88]. Besides, antigen-specific antibody production decreases with age, while self-reactive antibodies become more abundant, rendering old individuals more susceptible to develop autoimmune diseases [89]. All these defects in the humoral immune response lead to increased susceptibility to diseases and reduced efficiency of vaccines [90].
Trained Immunity: a De facto Innate Immune Memory
Although immune memory had been attributed only to the adaptive immune system for a long time, growing evidence consistently shows the existence of memory-like characteristics in innate immune cells [91,92,93,94]. Certain infections, vaccinations, or molecules can reprogram innate immune cell types to exhibit increased responsiveness against a secondary insult. This phenomenon is termed trained immunity and mediated through extensive epigenetic and metabolic changes.
Over the last couple of years, innate immune cells, including monocytes [95], natural killer (NK) cells [96], innate lymphoid cells (ILCs) [97], DCs [98], and neutrophils [99], have been reported to exhibit trained immunity response. As innate immune cells can only recognize microbial patterns via their pattern recognition receptors (PRRs), their memory-like response is not specific to pathogens but can work against a wide range of antigens. Thus far, vaccines, such as the tuberculosis vaccine Bacillus-Calmette Guérin (BCG) [100], measles [101], and oral polio vaccine [102]; microbes/microbial patterns, e.g., β-glucan [91], Candida albicans; oxidized low-density lipoprotein (oxLDL) [103]; and metabolites such as fumarate [104] have been reported to induce heterologous protection through trained immunity.
Epidemiological studies reporting decreased all-cause mortality after certain vaccinations suggested the existence of an innate immune memory [105]. The existence of trained immunity was first depicted in monocytes with an in vitro model and in vivo in mice, where C. albicans and β-glucan induced enhanced cytokine productions after the second microbial stimulation [91]. In parallel, BCG vaccination was reported to induce higher TNFα and IL-1β production against unrelated pathogens, even 3 months after the vaccination [100]. Further research demonstrated that trained immunity could persist up to 1 year and possibly even longer [106]. Considering that monocytes have a half-life around 1–2 days in the circulation [107], the programming of progenitor cells could be involved in sustaining the memory-like phenotype. Indeed, β-glucan administration leads to the expansion of myeloid lineage progenitors in the bone marrow of mice [108]. Increased myelopoiesis is associated with upregulated IL-1β and granulocyte–macrophage colony-stimulating factor (GM-CSF) signaling, besides alterations in glucose and cholesterol metabolism. Another mouse study demonstrated increased myelopoiesis following BCG vaccination, which is associated with enhanced protection against M. tuberculosis infection [109]. These findings align with a recent study on humans, showing that BCG vaccination leads to the upregulation of myeloid and granulocyte-lineage genes in HSCs [110].
Trained Immunity in the Elderly
Low-grade chronic inflammation occurring in the elderly is associated with poor innate and adaptive immune responses [111]. Koeken et al. recently reported that BCG vaccination reduces systemic inflammation, and a lower abundance of circulating inflammatory proteins at baseline is correlated with trained immunity response 3 months after vaccination in males [112]. Therefore, BCG vaccination could alleviate inflammaging while providing non-specific protection via trained immunity induction in the elderly. On the other hand, since the cell differentiation capacity of HSCs in the bone marrow changes and is skewed toward myelopoiesis with aging, inducing trained immunity could lead to unfavorable outcomes by further expanding the myeloid cell production in older people.
Nevertheless, a double-blinded placebo-controlled clinical trial demonstrated that trained immunity could be safely induced in the elderly by BCG vaccination, evident from the increased cytokine production compared to the participants who received placebo [113]. Remarkably, the trial showed that BCG prolongs the time until an infection and reduces the risk of all new infections and respiratory infections by 45% and 79% compared to the placebo group, respectively. In line with this, other trials reported a decrease in acute upper respiratory tract infections and pneumonia in older people vaccinated with BCG [114, 115]. However, more research is needed to explore the strength and longevity of trained immunity responses in older individuals compared to adults.
BCG’s ability to confer protection against heterologous infections has attracted a lot of attention during the COVID-19 pandemic, which disproportionally affects the elderly. BCG is being tested in more than 20 randomized control trials to investigate if it has a protective effect against SARS-CoV-2 infection [116]. Promisingly, a recently published study from Greece reported 68% risk reduction for COVID-19 6 months after BCG vaccination [117]. Another study revealed that even an early history of BCG vaccination is associated with decreased incidence and symptoms of COVID-19 among healthcare workers [118]. Therefore, induction of trained immunity by BCG vaccination may be utilized as a preventive measure against COVID-19, especially in the vulnerable elderly group.
Aging as a Multisystem Malady
Aging leaves no part of the body unscathed. Besides tissue-specific damage occurring with advanced age, the aging immune system impacts many other systems and processes. Even the organs that were once thought to be devoid of immune cells, such as the brain, are now known to harbor tissue-resident immune cells and interact extensively with the peripheral immune system. The last few decades have also witnessed a boom in research on the microbiota, the collection of up to 100 trillion microorganisms residing in human bodies, mainly in the gut [119]. The microbiota has close interactions with the host immune system and is also prone to age-related disruptions.
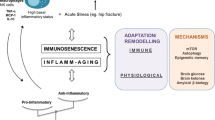
In the following chapters, we discuss the interplay of microbiota and the brain with the aging immune system, mainly focusing on immune memory. We especially approach this body of research from a metabolic perspective, describing various cellular metabolic programs and their impact on immune memory in aging and age-related diseases. Additionally, we point out the role of epigenetic regulation underlying all the topics discussed. By providing such a comprehensive view, visualized in Fig. 1, we aim to strengthen the notion of aging as a multisystem problem and accordingly inform counteractive efforts.
The far-reaching effects of the aging immune system. Age-related changes in immune cells include genomic instability, epigenetic modifications, altered cellular metabolism, and cellular senescence. An aged and impaired immune system has broad consequences, affecting many tissues and systems of the body. Gut microbiota and the central nervous system are profoundly impacted by and, in turn, regulate the immune system
Interplay of Metabolism and Immune Memory
Metabolism and metabolic inflammation are key processes that both influence and get influenced by aging. Metabolic diseases such as type 2 diabetes mellitus, cardiovascular diseases, and obesity are also considered age-related diseases. These conditions are accompanied by chronic inflammation, termed metaflammation, which is driven by nutrient excess. Although the triggers might vary, the mechanisms underlying metaflammation and inflammaging are quite similar. Mitochondrial dysfunction, accumulation of senescent cells and cellular debris, and hyperactivation of innate immune responses, such as inflammasome, contribute to both processes [120]. Therefore, it is crucial to understand the interplay between cellular aging, metabolism, and inflammation in chronological aging and age-related metabolic diseases to revert them.
T Cell Metabolism
Quiescent T cells mainly use catabolic processes, while activated cells rely on anabolic processes to support protein production and proliferation. Cells need to activate a critical serine/threonine kinase, mammalian target of rapamycin (mTOR), to induce anabolic pathways [121]. While driving growth and proliferation, mTOR also upregulates glucose transport and glycolysis. Glycolysis is one of the main pathways to generate energy. Although it is not energetically efficient — only 2 adenosine triphosphate (ATP) molecules can be generated from one glucose molecule — it generates energy very rapidly, which is of use for active and proliferating T cells [122]. Processing of glucose yields ATP, NADH, and pyruvate. Pyruvate is then converted to lactate and exported as lactic acid in the case of glycolysis or otherwise transported to mitochondria for oxidative phosphorylation (OXPHOS).
OXPHOS is a much more efficient bioenergetic pathway, producing 36 ATP molecules from every glucose molecule [123]. In this case, pyruvate is converted to acetyl-CoA and enters the tricarboxylic acid cycle (TCA cycle), which is coupled to the electron transport chain (TCA) through electron donors NADH and FADH2. TCA cycle can be replenished by amino acids and oxidation of fatty acids. Fatty acid oxidation (FAO) is mainly used by cells with low energy demands and plays a critical role in CD8+ memory and CD4+ Treg development [124]. Activated T cells upregulate their glutamine uptake and perform glutaminolysis to yield α-ketoglutarate, which enters the TCA cycle.
Additionally, TCA cycle metabolites can regulate immune functions in ways other than energy production. For instance, acetyl-CoA acts as the key cofactor for histone acetylation [125]. In activated T cells, acetyl-CoA is required for IFNγ production through histone acetylation [126]. Acetyl-CoA also contributes to the acetylation of mitochondrial proteins [127], which has vast functional consequences for both innate and adaptive immune cells [128].
Quiescent naïve T cells meet their energy needs with OXPHOS [129]. IL-7 and TCR signaling are essential for their metabolic regulation and survival [130, 131]. When T cells are activated, an immediate need for energy occurs for effector functions and biomass generation. The cells upregulate transporters like glucose transporter 1 (GLUT1) and engage in aerobic glycolysis, promoting cytokine production through pathways, such as the phosphoinositide 3-kinase (PI3K)-AKT-mTOR axis and mitogen-activated protein kinase (MAPK) signaling [132]. The glycolytic switch is required for the effector functions, e.g., IFNγ production but not essential for proliferation [133]. OXPHOS can also be utilized for proliferation and survival purposes. Although activated T cells functionally rely on glycolysis, OXPHOS is certainly not dispensable: when OXPHOS is inhibited with oligomycin, T cell activation and proliferation are blocked [133].
Although they rely on OXPHOS and FAO in the resting state, memory T cells need to respond quickly and efficiently upon antigen encounter. Therefore, they can shift to glycolysis quicker than naïve T cells [134]. Greater mitochondrial mass and a strong mitochondrial spare respiratory capacity have been linked to this bioenergetic advantage [135, 136]. Additionally, mitochondrial fusion is essential for the development and function of memory T cells [137].
Impact of Aging on T Cell Metabolism
Increased p38 MAPK activity is one of the characteristics of senescent T cells. Inhibiting p38 improves telomerase activity, proliferation, autophagy, and mitochondrial fitness, in an mTOR-independent way [17]. MAPK inhibition also enhances T cell and antibody responses in influenza-vaccinated old mice [138].
Patients with gain-of-function mutations in PI3K have depleted naïve T cells but an accumulation of senescent effector cells, just like in the elderly [139]. Inhibiting mTOR activity with rapamycin treatment partially restores the senescent phenotype in these patients. Therefore, overactive PI3K/AKT/mTOR signaling is suggested as one of the drivers of T cell senescence.
Aged naïve T cells have higher mitochondrial mass, but interestingly, less mitochondrial respiratory capacity, possibly due to transcriptional downregulation of respiratory chain genes [140]. Furthermore, enzymes of one-carbon metabolism are deficient in aged naïve T cells, and supplementation with formate and glycine, one-carbon metabolism metabolites, improves cell survival and activation [141].
Autophagy is important for the generation of T cell memory, and induction of autophagy by spermidine improves CD8+ T cell responses against influenza vaccination in aged mice [142]. CD4+ memory T cells of the elderly display upregulated oxidative phosphorylation, reactive oxygen species (ROS) production, and fatty acid oxidation [143]. They also have a higher expression of Sirtuin 1 (SIRT1), a NAD-dependent deacetylase, compared to younger cells. SIRT1 and AMPK, two important nutrient-sensing molecules and negative regulators of mTOR, positively influence each other [144]. In contrast to CD4+ memory cells, aging-associated terminally differentiated memory CD8+CD28− T cells have a high glycolytic capacity, which is linked to their downregulated SIRT1 expression [145].
CD8+ TEMRA cells have a higher expression of glycolysis and glutaminolysis-related genes and a larger ATP pool compared to naïve and EM cells [146]. Despite upregulated glycolytic transcription in TEMRA cells, basal glycolysis levels are similar to naïve and EM cells. Like EM cells, TEMRA cells can quickly increase glycolysis and OXPHOS upon activation [146]. In terms of function, TEMRA cells are capable of cytotoxicity and cytokine production, despite their senescent state and impaired mitochondrial function [17, 36].
Long-term CMV infection, known to promote immunosenescence, also alters the cellular metabolism of T cells, increasing glucose uptake, promoting glycolysis, restructuring lipid rafts, and disturbing cholesterol metabolism [147, 148]. In addition, chronic inflammation due to lifelong CMV infection disrupts pancreatic β-cells and increases the risk for type 2 diabetes in the elderly [149].
B Cell Metabolism
The metabolic pathways that regulate T cells are also essential for B cell function, although there has not been much research on B cell metabolism. When a B cell is activated upon antigen recognition by the BCR and T cell help, it activates PI3K/AKT/mTOR signaling [150]. Just like activated T cells, activated B cells need rapid energy production to increase biomass and proliferate. As a result, glucose and glutamine uptake increase, along with oxygen consumption, OXPHOS, and mitochondrial remodeling [151]. OXPHOS and glutamine-fueling of the TCA cycle have been suggested as the critical bioenergetic pathways for B cell growth and function, while glucose was dispensable [152].
A study showed that activated B cells have more mitochondria but similar amounts of mitochondrial DNA, indicating that fission of naïve B cell mitochondria with multiple nucleoids, rather than mitochondrial replication, occurs upon activation [152]. Another study suggested that mitochondrial remodeling and ROS levels determine the fate of activated B cells. Cells with increased mitochondrial mass and higher ROS levels upon activation are destined for class switch recombination, whereas cells with decreased mitochondrial mass undergo plasma cell differentiation [153].
The energy needs of activated B cells in GCs frequently shift [154]. In the hypoxic light zone, cells consume less oxygen and are more glycolytic. mTORC1 is not necessary for the regulation of glycolysis here, but it is critical, together with c-Myc, for the positive selection of the cells and migration to the dark zone for proliferation and somatic hypermutation [155, 156].
Upon GC maturation, when a cell differentiates into memory B cell, the metabolic state becomes more quiescent with dominant OXPHOS. However, rapid re-activation of mTORC1 and glycolysis is possible for later differentiation into antibody-producing plasmablasts [157]. Furthermore, memory B cells have high basal autophagy, which is essential for their survival until antigen encounter [158, 159].
GCs also output long-lasting plasma cells, which can produce thousands of antibodies per second. This, naturally, is highly energy demanding. mTORC1 is essential for plasma cell generation and antibody synthesis [160]. Plasma cells have high levels of glucose uptake, but most of the glucose is used for protein glycosylation [161]. Still, survival and antibody production of plasma cells were impaired when the glucose transporter Glut1 was deleted [162]. Also, mitochondrial import of pyruvate, provided by glycolysis, is critical for the long-term maintenance of plasma cells [161].
Finally, tissue-resident B1 B cells are more active in glycolysis and OXPHOS than other B cells, the classical antibody-producing and memory B cells. In addition, autophagy is critical for the mitochondrial function and self-renewal of B1 cells [163].
Impact of Aging on B Cell Metabolism
There is less literature on how B cell metabolism is regulated and impacts function as organisms age. A study showed that antibody-secreting B cells of aged individuals had lower SIRT1 expression, and higher SIRT1 levels were associated with better antibody response to multiple influenza virus strains [164]. Also, naïve and activated B cells of the elderly had slightly less glycolytic capacity and a more striking reduction in OXPHOS. In mice, aged B cells had similar glycolysis and OXPHOS rates as young counterparts but could not further enhance OXPHOS upon stimulation [165]. However, the cells were able to upregulate glycolysis to meet their energy need.
Leptin, a pro-inflammatory hormone secreted by adipocytes, is higher in the circulation of obese individuals [166]. Among non-obese people, leptin concentrations are strikingly more elevated in the elderly [167]. Leptin abundance in the serum is also positively associated with frailty [168]. After exposure to leptin, B cells from young lean individuals exhibit a similar profile as B cells of older lean and young obese individuals regarding the transcriptional profile and antibody secretion [167]. Leptin also decreases influenza-specific antibody production from B cells in vitro. Obesity is known to impair B cell responses to vaccination, and studies suggest that leptin might be partially responsible for this [169].
Additionally, post-transcriptional glycosylation of antibodies modulates their function, and altered glycosylation patterns have been linked to aging [170, 171]. β4-Galactosyltransferase activity increases with age [172], which would have functional consequences, although yet unexplored.
Metabolism in Trained Immunity
Metabolic reprogramming is one of the key mechanisms underlying trained immunity (also known as innate immune memory), along with chromatin remodeling. In fact, metabolic changes can drive epigenetic changes since certain metabolites, e.g., acetyl-CoA, can regulate epigenetic enzymes [173]. Fumarate is one example of TCA metabolites driving epigenetic changes. It can induce trained immunity on its own, and its accumulation during this process induces trimethylation of histone 3 lysine 4 at the promoters of IL-6 and TNFα [104]. This is due to fumarate inhibiting the activity of lysine-specific histone demethylase KDM5.
The AKT/mTOR/HIF1α pathway is the most critical pathway for inducing aerobic glycolysis in β-glucan-trained monocytes [174]. Contrary to β-glucan-induced trained immunity, BCG upregulates not just glycolysis but also OXPHOS [175]. Glutaminolysis and cholesterol synthesis are other crucial metabolic pathways for β-glucan-induced trained immunity [104]. Interrupting these pathways blocks these processes in vitro and in vivo. BCG also induces glutaminolysis, and glutamine availability is important for the trained response [175].
Synthesis of cholesterol itself is not essential for trained immunity but rather the accumulation of the intermediate mevalonate is. Blocking mevalonate generation inhibits trained immunity, while mevalonate alone can induce trained immunity in monocytes through the activation of insulin-like growth factor 1 (IGF1) receptor and mTOR [176]. Furthermore, the changes in glycolysis and mevalonate pathways are observed not only in monocytes but also in HSPCs [108].
oxLDL, a non-microbial inducer of innate immune memory, upregulates both glycolysis and oxygen consumption, and high glucose availability further enhances the trained immunity response [103]. Similarly, catecholamine-induced trained immunity is accompanied by increased glycolysis and oxygen consumption. Of note, the particular metabolic rewiring might differ for different inducers of innate immune memory. For instance, stimulation with aldosterone is not associated with elevated glycolysis or OXPHOS but is dependent on fatty acid synthesis [177].
As of yet, trained immunity responses and associated metabolic states have not been characterized in the context of aging. However, several ongoing large-scale studies of BCG vaccination in the elderly would soon shed light on the effects of BCG-induced trained immunity on the metabolism of aged immune cells (NCT04537663, NCT04417335).
Role of Epigenetic Alterations in Immune Memory
Epigenetic changes include histone modifications and DNA methylation that regulate the way a gene works. These modifications are dynamic and affect all cells and tissues throughout life. Environment and lifestyle, as well as aging, can lead to dramatic epigenetic alterations. For the purpose of this review, we will focus on how age-dependent epigenetic modifications alter innate and adaptive immune memory.
DNA Methylation In Adaptive Immunity
DNA methylation is the most abundant epigenetic modification that occurs by transferring a methyl group to the 5th carbon of the cytosine [178]. DNA methylation does not always indicate a lower gene expression; however, methylation in gene promoters is generally associated with poor TF binding and reduced transcription [179]. Biological sex, genetic background, environmental factors, and age affect the DNA methylation profile [180]. Among these factors, age-dependent methylation is very well-characterized. Remarkably, different mathematical models are developed to predict the biological age based on the methylation levels of certain CpG sites from various tissues or cells [180,181,182].
Advancing age is associated with a progressive loss of methylation marks on DNA [183], although abnormal hypermethylation patterns are also observed in some gene promoters [184]. Changes in the methylation landscape are linked to various age-related diseases as well as dysfunctions in the immune system. For instance, age-related macular degeneration, a disease resulting in irreversible blindness in the elderly, has been correlated with the loss of methylation in the promoter region of the IL17RC gene, leading to increased IL17RC protein levels in the blood [185].
A growing number of studies indicate that DNA methylation plays a significant role in the adaptive immune system’s functioning. Age-related functional changes in immune cells, such as decreased self-renewal capacity, defects in cell differentiation, and skewed differentiation towards myeloid cell production in the elderly, are strongly correlated with epigenetic modifications occurring in HSCs during aging [186]. Murine studies show that gene expression of HSCs is regulated via hyper- and hypomethylation of certain DNA regions, affecting the capacity of those cells to differentiate [187]. Expression of DNA methyltransferases in HSCs are lower in aged mice [186]. Also, HSCs of mice with decreased DNA methyltransferase activity fail to efficiently differentiate into lymphoid progeny [188]. These studies reveal that DNA methylation is essential to fine-tuning the differentiation capacity of HSCs and therefore the proper activity of the innate and adaptive immune system. Epigenetic modifications also modulate the function of HSCs during aging, which will be elaborated in “Histone Modifications.”
Several studies report age-dependent methylation changes in T cells. A study analyzing CD4+ and CD8+ T cell methylome profiles in young individuals and the elderly found that 48,876 and 12,275 CpG sites were differentially methylated in CD8+ T and CD4+ T cells, respectively [189]. Moreover, the methylation profile of CD8+ T cells was strongly associated with aging and inversely correlated with genes linked to T cell differentiation and immune response, suggesting a possible link between weakened T cell responses and age-related alterations in DNA methylation.
The age-associated methylation profile of CD4+ T cells is characterized by hypermethylation of CpG sites enriched in the polycomb repressive complex 2 (PRC2) genes and hypomethylation of CpG sites enriched in enhancer regions [190, 191]. Of note, the PRC2 proteins regulate histone methylation, cell differentiation, and proliferation [192]. These methylation patterns identified by Dozmorov and colleagues were highly similar to the methylation and transcriptomic profile of T cells from lupus patients. Lupus erythematosus, an autoimmune disease leading to autoreactive T cells, is characterized by defects in the MAPK signaling pathway and increased mTOR activity resulting from altered methylation patterns [193]. Therefore, the authors suggested that the age-dependent methylation profile of naïve CD4+ T cells might render the elderly susceptible to autoimmune diseases, such as lupus, though this remains to be formally demonstrated.
Loss of CD28 co-stimulatory protein in CD4+ T cells is one of the well-characterized aging marks, leading to impaired T cell activation and differentiation. Comparison of methylation profiles of CD28+ and CD28null T cells revealed 296 differentially methylated genes associated with poor TCR signaling and cytotoxic response [194]. Furthermore, the expression of the genes involved in inflammasome activation was higher in CD28null T cells, suggesting that these cells have a higher pre-activation state. Another study reported that increased methylation at the BACH2 locus of the CD4+ T cells in the middle and old age groups results in lower BACH2 expression [195]. BACH2 has a regulatory role in immune responses, modulating CD4+ T cell differentiation and controlling inflammation [196]. Overall, alterations in the DNA methylation patterns contribute to CD4+ T cells becoming more inflammatory in the elderly.
A few studies shed light on the DNA methylation profile of B cells during activation and diseases [197,198,199,200]; however, whether B cells are affected by age-dependent methylation changes is yet to be known.
Histone Modifications in Adaptive Immunity
N-terminal histone tails are targets for post-translational enzymatic modifications including acetylation, methylation, phosphorylation, ubiquitylation, and sumoylation [201]; however, this review will focus on methylation and acetylation, which are the most well-characterized alterations regulating histone structure. Methyl groups are added to the histone by histone methyltransferases and removed by histone demethylases [202]. The trimethylation of histone 3 lysine 4 (H3K4me3), histone 3 lysine 36 (H3K36), and histone 3 lysine 79 (H3K79) are linked to open and actively transcribed regions [203]. On the other hand, mono-methylation of histone 3 lysine 9 (H3K9me), histone 3 lysine 27 (H3K27me), and histone 4 lysine 20 (H4K20me) is associated with closed and inactive chromatin regions. Furthermore, histone acetylation is associated with loosened chromatin structure and increased gene transcription [204]. Histone acetyltransferases catalyze lysine acetylation, whereas histone deacetylases (HDACs) reverse the modification [205]. Post-translational modifications of histones do not only influence the accessibility and transcription of genes but also modulate alternative splicing, DNA replication, and repair [206].
Histones and epigenetic marks on histones undergo transitions with aging. HSCs from old mice have more H3K4me3 and H3K27me3 peaks compared to young HSCs [186]. In addition, expression of FLT3, one of the regulators of CLPs, was decreased due to H3K27me3 in the old HSCs, suggesting a link between poor lymphoid differentiation potential of HSCs in the elderly. An extensive study performed in young and old monozygotic twins showed that chromatin modifications during aging are non-heritable [207]. Moreover, histone modification profiles are, to some extent, homogenous in young individuals and heterogeneous among elderly subjects. Heterogeneity in histone modifications was observed between individuals and also cell types in the elderly.
Epigenetic changes are one of the underlying causes of the major defects seen in CD8+ T cells of the elderly. More closed chromatin regions are observed in the enhancer and promoter regions of the genes related to T cells signaling in the elderly compared to the young [208]. Furthermore, IL-7R, in the memory CD8+ T cells, is one of the top genes related to multiple closed chromatin peaks in the elderly. As IL-7 ensures homeostasis and maintenance of T and B cells, poor IL-7 signaling in the elderly might be an underlying reason of impaired adaptive immune response [209]. Furthermore, naïve CD8+ cells in the elderly have lower chromatin accessibility at the gene promoters associated with poor nuclear respiratory factor 1 (NRF1) binding [140]. Considering the role of NRF1 in oxidative phosphorylation, decreased chromatin activity might partially explain the impaired CD8+ T cell metabolism in the elderly [210]. Other significant findings of the study are that open chromatin regions are associated with a memory cell profile, and accessibility of the promoters is diminished in aged individuals.
As mentioned in the DNA methylation section, an age-associated decrease in BACH2 expression is observed in CD4+ T cells. Another mechanism leading to lower BACH2 gene transcription is due to Menin deficiency observed in immune senescence [211]. Menin induces BACH2 expression by binding to its locus and maintaining histone acetylation. Decreased binding of Menin to BACH2 locus and subsequently reduced BACH2 expression contributes to immunosenescence in CD4+ T cells.
A study investigating the epigenetic changes in B cell precursors in old and young mice associated these alterations with gene expressions [212]. It revealed that aged pre-B cells exhibit a loss of H3K4me3 at the promoter site of insulin receptor substrate 1 (IRS1), which is associated with lower transcription. As insulin signaling is necessary for the development of B cells in the bone marrow [213], decreased insulin growth factor (IGF) signaling might lead to defects in B cell development.
Epigenetic Reprogramming as a Hallmark of Trained Immunity
A distinct epigenetic profile regulates trained immunity responses following the first insult. As a result of certain infections or stimulations, primed cells undergo an epigenetic reprogramming that allows them to respond stronger upon a heterologous infection by facilitating the transcription of genes related to inflammation and metabolism [106].
H3K4me3 is the first characterized epigenetic mark in monocytes after β-glucan treatment [91]. Further analysis revealed that H3K4me3 peaks are enriched at the promoter sites of TNF, IL6, IL18, DECTIN1, and MYD88 genes, indicating that gene transcriptions are more active in these regions. In addition, increased H3K27ac is a well-characterized histone mark in trained cells, promoting glycolysis and PI3K/AKT pathway activation [174, 214]. Besides the enrichment in H3K4me3 and H3K27ac, decreased H3K9me3 was found in the promoters of genes related to cytokine production and glycolysis [175]. Since H3K9me3 is a repressive mark, reduced trimethylation suggests the presence of open chromatin regions. These studies show that trained immunity responses are modulated by epigenetic modifications that facilitate enhanced cytokine responses and specific metabolic changes. Trained cells share a common epigenetic profile; however, different stimuli could lead to minor unique epigenetic alterations.
Infections and certain stimulations leave marks on the DNA methylation profile, as well as histones, of innate immune cells [215]. Studies demonstrate the role of DNA methylation in anti-mycobacterium response following BCG vaccination, discriminating responders from non-responders [216, 217]. Responders to BCG vaccination were characterized by reduced DNA methylation at the promoters of inflammatory genes [216]. However, whether DNA (de)methylation plays a direct role in the development of non-specific protective responses is still being investigated.
As in adults, trained immunity is modulated by histone modifications in the elderly. Giamarellos-Bourboulis and colleagues recently showed that increased cytokine production upon BCG vaccination in the elderly was accompanied by acetylation of H3K27 at the promoter regions of TNF and IL6 genes [113]. However, further studies are warranted to compare the epigenetic differences following innate immune memory development between adults and older individuals and explore how aging influences epigenetic marks in the context of trained immunity.
Gut Microbiota Modulating Immune Memory
Aging causes changes throughout the whole body of humans, and trillions of microbes living there are no exemption. The composition and diversity of gut microbiota dynamically shift in infancy, remain relatively stable during adulthood, and start to decline with old age [218].
Interactions of Microbiota and the Adaptive Immune System
The gut microbiota has essential roles in educating the adaptive immune system by inducing a certain level of immune response and fine-tuning the inflammation. For instance, Bacteroides fragilis, a commensal in the gut, enhances and regulates CD4+ T cell differentiation into T helper 1 (Th1) and Th2 [219]. In the presence of gut bacteria and TGFβ, naïve CD4+ T cells become Tregs, producing IL-10 to maintain immune homeostasis. On the other hand, Tregs and Th17 cells in the lymphoid follicles of the gut induce B cell class switching, resulting in IgA secretion [220, 221]. Microbiota-associated IgA, IgM, and IgG secretion from B cells also occurs via TLR signaling activation without T cell help [222].
The adaptive immune system can limit the inflammatory response against commensal gut microbes mediated by the innate immune system. IgA produced by B cells is explained as a part of sustainable host-microbe interaction, controlling the inflammatory response against beneficial microorganisms [223]. Besides, intestinal Treg cells express TCRs for intestinal antigens, such as metabolic products and commensals, while other Tregs in the body express TCRs for self-antigens [224]. In this way, intestinal Tregs suppress immune responses against intestinal antigens and play an immunoregulatory role in the guts.
How microbiota strikingly shapes the adaptive immune system development was also demonstrated in germ-free mice: the lack of microbial species in the gut is characterized by defects in secondary lymphoid tissue development [225], low IgA production [226], and reduced Th17 cells and Tregs [227]. It should be noted that short-chain fatty acids (SCFAs) produced by microbial species in the gut greatly contribute to the immune system development and responses [228].
A healthy gut microbiota composition is important in protecting individuals from diseases. As an example, IL-10 secreting IgA + plasma cells and plasmablasts originating in the gut confer resistance to experimental autoimmune encephalomyelitis induced in mice [229]. Another study reported that gut microbiota protects against respiratory infections induced by S. pneumoniae and K. pneumoniae by inducing GM-CSF and IL-17A secretion [230].
The Role of Dysbiosis in Aging
The incidence of gut dysbiosis, the imbalance of microbial species, increases with age and is associated with numerous health problems [231]. However, it is unclear whether cellular and molecular alterations of the immune cells during aging affect the composition and functioning of the gut microbiota, or if age-related dysbiosis triggers defective immune responses. It is likely that both are concurrently true, but a better understanding of the gut microbiota-immune system interactions is necessary to resolve this question.
As individuals age, a decline in certain beneficial bacterial species, such as Bifidobacterium, is replaced by the growth of pathogenic species, i.e., Enterobacteriaceae [232]. A decrease in Firmicutes and increase in Proteobacteria are also reported in older people [233]. Besides, gut dysbiosis is associated with several age-related diseases, including obesity [234], type 2 diabetes [235], Alzheimer’s disease [236], and increased incidence of infections [237,238,239]. The risk of developing cancer is also higher in the elderly due to dysbiosis-associated chronic inflammation, debilitated phagocytosis of senescent and dormant tumor cells, and impaired activation of tumor-specific CD8+ T cells [240].
Dysbiosis was also proposed to be a major reason for various age-associated pathologies and premature death in older individuals by triggering excess inflammation and several complications, including leaky gut and diminished functions of gastrointestinal tract [228]. In line with this, a particular composition and diversity of microbial species is correlated with health, fitness, and increased survival in the elderly [241, 242]. A recent study revealed that healthy elderly experience a particular drift in their microbiota composition, while this drift is missing in the frail elderly [242]. Furthermore, having high Bacteroides abundance during aging correlates with decreased survival rate over the 4-year follow-up. Another recent work with 15 years of follow-up reported that Enterobacteriaceae abundance was significantly linked with deaths related to gastrointestinal and respiratory causes in the elderly [243].
Dysbiosis can lead to defects in intestinal barrier integrity, which results in the translocation of bacterial species to the host tissues. Those bacteria create inflammation through the recruitment of neutrophils and differentiated Th17 cells [244]. For example, translocation of a gram-positive pathobiont E. gallinarum that results from defects in the gut barrier induces Th17 response and autoantibody production [245].
Akkermansia is a beneficial commensal shown to protect the gut barrier integrity [228] and enhance antibody and T cell responses [246]. Loss of Akkermansia is associated with insulin resistance in aged non-human primates and mice [247]. Decreased butyrate and Akkermansia abundance increase gut leakage, which in turn increases pro-inflammatory responses.
A human study, on the other hand, reported that Akkermansia is more abundant in the elderly [248]. Furthermore, Akkermansia was significantly correlated with serum IgA and CD8+ T cells and negatively correlated with CD4+ T cells in older people. Bacteroidetes, which are less abundant in the elderly, were positively correlated with serum IgG levels and CD4+ T cell abundance in the middle age group. In conclusion, this study highlights the relationship between the adaptive immune system and gut microbiota composition, although the direct link between them is missing.
Microbiota also affects disease course and vaccine responses in the elderly. Even though the antiviral therapy for human immunodeficiency virus (HIV) is successful and increases the life expectancy of patients, older HIV + people suffer more from comorbidities compared to HIV − elderly. HIV + elderly have less CD4+ T cells and more CD8+ T cells than HIV individuals older than 55 [249]. In addition, the abundance of Prevotella in the gut is significantly higher in the individuals with low CD4+ T cell counts. Prevotella was previously associated with cardiovascular diseases [250], but how it interacts with the immune system is not yet clear.
Age-dependent alterations in gut microbiota are likely to contribute to poor immune responses after vaccinations [251]. Some studies reported that probiotics supplements increase the antibody titers after influenza vaccine in the elderly [252,253,254,255], whereas a few studies showed limited or no effect [87, 256, 257]. Variations in the results could be due to multiple factors, including the sample size, type of probiotics, and delivery route. Nevertheless, studies strongly suggest that imbalances in microbiota cause impaired immune responses, and restoring the healthy composition might be beneficial for a better vaccine response in the elderly.
Innate Immune Memory Induction by Gut Microbiota
As the adaptive immune cells, members of the innate immune system closely interact with the gut microbiota. A few studies suggest that microbiota could regulate immune memory development by priming or tolerizing the cells with microbial antigens and SCFAs. For instance, β-glucan, a fungal cell wall component, and BCG act through Dectin-1 and NOD2 signaling pathways, respectively [91, 100]. Since Dectin-1 and Nod-like receptors (NLRs) are found on various cell types in the intestines, including non-immune cells, it is plausible to propose that these cells develop immune memory due to their exposure to the gut microbiome. Supporting this argument, peptidoglycan fragments derived from gut microbiota were shown to prime the innate immune system, promoting the killing capacity of neutrophils [258].
Furthermore, gut microbiota was shown to induce myelopoiesis to protect mice against infection [259], similar to the increase in the number of myeloid progenitors in the bone marrow of mice following trained immunity induction by β-glucan administration [108]. Other microbiota-derived components, such as lipopolysaccharide (LPS), flagellin, and β-glucan, might also be able to induce trained immunity in the guts, although the dose of the stimuli is critical for immune memory or tolerance response [260].
As mentioned before, trained immunity is mediated by extensive metabolic and epigenetic programming. Molecules and metabolites produced by commensal gut microbes and microbes themselves are able to induce such changes in both innate and adaptive immune cells [261]. For example, despite causing an increase in the anti-microbial activity, butyrate produced by gut microbes have effects opposite to trained immunity in macrophages, possibly stemming from decreased mTOR activity and inhibition of HDAC3 [262].
It is important to note that non-immune cells, e.g., fibroblasts [263], epithelial cells [264], and intestinal stromal cells (ISCs) [265] are also capable of forming immune memory, showing increased responsiveness after secondary infection. It was shown that ISCs could clear infection more rapidly during a secondary related or unrelated infection, indicating the presence of immune memory [266]. Therefore, non-immune cells also contribute to the homeostasis between gut microbes and the immune system.
Considering the strong links between gut microbiota and induction of innate immune memory, it would be conceivable to hypothesize that trained immunity response could be dysregulated by the dysbiosis in the elderly. Poor trained immunity response could render the elderly more susceptible to infections, while exuberant response might contribute to disease pathogenesis. However, more research is needed to understand how age-related changes in microbiota affect innate immune memory.
Cross talk Between the Immune System and the Brain
Aging causes a great deal of deterioration in the central nervous system (CNS) through DNA damage, accumulation of waste products, oxidative stress, disturbed energy homeostasis, and impaired function [267]. The brain and the rest of the CNS are not immunologically isolated, as once thought: there is extensive cross talk between the immune system and the CNS. Brain homeostasis and regeneration depend on a robust immune system [268]. Therefore, deterioration of the immune system with old age contributes to and escalates brain aging and neurodegenerative diseases.
In the CNS parenchyma, the resident immune cell type is the microglia, which originates from primitive macrophage progenitors in the yolk sac early in development [269]. Microglia are extremely important for the maintenance of a healthy brain. They perform immunosurveillance, respond to infections, orchestrate the communication with the circulating immune system, regulate neurons, and other cell types in the brain, phagocytose cellular debris, misfolded proteins, toxic products, and even synapses [270]. Microglia are altered by aging and contribute to age-related neurodegenerative diseases [271]. Their phagocytic capacity is reduced with advancing age, and they contribute to a state of chronic low-grade inflammation. Due to this review’s focus on immune memory, we will not go into detail on microglia and instead focus on the role of adaptive immunity and trained immunity in the context of brain aging.
The blood–brain barrier (BBB) largely prevents the infiltration of immune cells into the brain. However, certain immune cell types are present in the cerebrospinal fluid (CSF) and the blood-CSF barrier at the choroid plexus (CP) [272]. CP, located in the brain’s ventricles, is a CSF-producing epithelial cell network with embedded capillaries. T cells are present in CP, and they regulate immune cell trafficking into CSF by IFNγ-dependent activation of CP epithelium [273].
Immune cells contribute to neuronal survival and neurogenesis during homeostasis, upon injury, or under neurodegenerative conditions [272]. Damage to the CNS induces a protective T cell response that prevents neuronal loss [274]. CD4+ lymphocytes play the most prominent role in this “neuroprotective immunity.”
Neuroprotective T Cell Immunity
CP harbors CD4+ T cells with an effector-memory phenotype that recognize CNS-specific self-antigens [275]. These cells can receive signals from circulation through the epithelium and the CNS through the CSF and orchestrate an integrated response to maintain brain homeostasis [276]. Astrocytes, a cell type that helps maintain synapses and the BBB, among various other functions, assume a neuroprotective phenotype and reduce neuronal apoptosis when co-cultured with T cells [277]. During spinal cord injury, CNS-specific autoreactive T cells migrate to the injury site, inhibit cyst formation, and contribute to the preservation of axons [278].
In T cell-deficient mice, the proliferation of progenitor cells is reduced, leading to lower numbers of new neurons, while neurogenesis is boosted in transgenic mice with excess CNS-specific autoreactive T cells [268]. Supplementation of the T-cell-derived cytokine IFNγ can enhance neurogenesis in old mice with Alzheimer’s disease [279]. CNS-specific T cells are also critical for spatial learning and memory. In immunodeficient mice, spatial memory is impaired but can be restored with reconstitution of immune cells even in aged mice [280]. In models of the motor neuron disease amyotrophic lateral sclerosis (ALS), T cell deficiency accelerates the disease, while reconstitution promotes neuroprotection and delays disease progression [281,282,283]. However, of note, T cells contribute to the death of dopaminergic neurons in mouse models of Parkinson’s disease [284].
One mechanism through which T cells improve brain maintenance is the regulation of brain-derived neurotrophic factor (BDNF). BDNF signaling via tropomyosin receptor kinase B (TrkB) plays wide-ranging roles, for example, in adult neurogenesis [285], memory formation and retrieval [286, 287], and is regulated by anti-depressant treatments [288]. BDNF levels are lower in T cell-deficient mice [268]. BDNF is associated with depressive behavior and immunization of mice with a myelin-derived peptide, generating CNS-specific immunity, restores BDNF levels, improves neurogenesis, and reduces depressive behavior [289]. Furthermore, healthy stress response in mice is associated with T cell trafficking in the brain and BDNF levels. Anxious behavior caused by stress is also reduced by immunization with a myelin-derived peptide [290]. Apart from neurons and microglia, T cells themselves are shown to secrete BDNF [291].
Tregs are also shown to be protective and delay disease progression in ALS by reducing microglial activation [292]. In models of Alzheimer’s disease, Treg transplantation enhances cognitive abilities and reduces amyloid plaques [293]. Moreover, a lower Treg/Th17 ratio is correlated with more severe disease in patients with multiple sclerosis, a debilitating autoimmune disease affecting neurons [294].
Although an over-exuberant immune response would impair brain function, a fine-tuned T cell immunity is clearly vital for healthy brain homeostasis and recovery from injury. Any intervention targeting this phenomenon must be carefully controlled to avoid inflammatory damage; however, the insights into adaptive immunity’s role in brain health open up new avenues to counter brain injury or age-related neurodegenerative diseases.
Trained Immunity in Microglia
Recent studies suggest that innate immune memory can be induced in microglial cells. One study found epigenetic reprogramming in microglia present for at least 6 months upon systemic LPS administration [295]. Interestingly, while a single LPS injection induced a trained phenotype in microglia, repeated LPS injection led to the induction of tolerance. Similarly, low-dose TNFα administration was also found to induce microglia training. In a mouse model of Alzheimer’s disease, trained immunity exacerbated the disease while tolerance alleviated it. A recent study confirmed the finding of LPS-induced training and demonstrated that systemic β-glucan administration could also induce trained immunity in microglia [296]. However, the trained phenotype of microglia was only observed two days after the priming and was no longer present at day 7, possibly indicating a lack of sustained epigenetic reprogramming. Therefore, it is worthwhile to investigate the strength and persistence of training with different doses and different injection regimens.
The Aging Brain
Many brain functions deteriorate with aging, with some even starting to decline after the third decade of life [297]. The impaired functions include processing speed, problem-solving, fluid reasoning, perceptual abilities, verbal fluency, and working memory. However, the impairments do not necessarily correlate with chronological age. It is rather an outcome of increased maintenance demand through the accumulation of damage and the inability of the immune system to monitor the brain to meet these demands. Of course, aging contributes to both the demand and the incapacity of the immune system through the mechanisms discussed earlier.
Aged microglia develop a pro-inflammatory phenotype [298]. Following a head injury or infection, they produce an excessive amount of pro-inflammatory cytokines for a longer time compared to a healthy young brain [299]. This inflammatory state leads to inhibited neurogenesis [300, 301]. A pro-inflammatory environment also inhibits modulators of long-term memory such as BDNF and activity-dependent cytoskeletal-associated protein and causes memory dysfunction [299]. Circulating BDNF levels decrease with age in humans, and brain levels are shown to decline in rodent models [302], which might reflect the age-associated drop in T cell numbers and function.
Aging is also associated with increased recruitment of effector memory CD8+ T cells to the CP and the meninges — the membranes covering the brain [303]. These cells were shown to impair microglial function during homeostasis but enhance pro-inflammatory cytokine production upon injury. Moreover, Treg numbers are elevated in elderly individuals; however, their migratory capacity and function are likely impaired since they are not able to control neurodegeneration. For instance, Tregs of multiple sclerosis patients have less immunosuppressive capacity and are unable to survive in sclerotic lesions in the brain [304].
In the case of chronic inflammation, while innate immune cells typically display tolerance leading to lower cytokine production, microglia acquire a primed to exhibit a more inflammatory phenotype, accelerating cognitive decline [305]. In addition, high levels of circulating TNFα observed in aged organisms might also cause damage by inducing trained immunity in microglia, as discussed above. Therefore, a well-balanced innate immunity is as essential for the healthy maintenance of the brain as adaptive immunity.
Tackling Immune Aging From All Angles
Efforts to slow or revert aging are far from scarce. However, the outcome measures assessed by most studies are restricted in the sense that they do not offer mechanistic insights or focus on specific processes. Yet, some exciting interventions, including caloric restriction, metformin, and physical exercise, interfere with aging on multiple levels encompassing immunity, metabolism, epigenetics, microbiota, and the nervous system (Fig. 2). The following chapters discuss different ways to tackle the aging problem and detail the mechanisms of the most promising anti-aging treatments.
Promising anti-aging interventions that target multiple facets of the aging process. Metformin delays stem cell aging, improves mitochondrial function, prevents telomere shortening, reverses age-related epigenetic modifications, and reduces gut leakiness and dysbiosis. Physical exercise, even if initiated late in life, improves immune cell numbers and functions, restores mitochondrial metabolism, prevents cellular senescence, counteracts cognitive decline, and reduces risks for neurodegenerative diseases. Resveratrol, available in grapes and red wine, acts as an antioxidant, extends lifespan in various model organisms, attenuates systemic inflammation, and slows epigenetic aging. Caloric restriction by 20–40% enhances lifespan and reduces all-cause mortality in non-human primates, delays epigenetic aging, restores gut microbiota, and slows cognitive decline. Cellular mechanisms shared by these treatments include limitation of the mTOR/AKT axis and activation of AMPK and SIRT1
Metabolic Interventions
For most of the human evolution, nutrients were scarce, and a great deal of physical activity was required to obtain them. Thus, humans evolved to adapt to those conditions. Our current sedentary lifestyle with an overabundance of nutrients is proposed to cause the high prevalence of metabolic diseases, such as obesity, diabetes, and cardiovascular disease [306]. Furthermore, age is a risk factor for these conditions, as mentioned before, and immunosenescence has a lot in common with metabolic disease profiles. Therefore, focusing on metabolic interventions is a sensible approach to tackle aging and metabolic disorders simultaneously. Caloric restriction (CR) and exercise, bringing us closer to the ancestral conditions, take the lead in this line of research.
CR refers to a reduction of total calory intake by 20–40%. From yeasts to non-primates, CR has repeatedly been shown to enhance lifespan [307]. In rhesus monkeys, CR starting from young adulthood reduced the risk of mortality related to age-related causes by threefold and all-cause mortality by 1.8-fold [308]. In another study, CR decreased the incidence of diabetes, cancer, and cardiovascular disease while also delaying disease onset [309]. A contrasting study reported no improvement in survival, although the incidence of cancer and diabetes was reduced [310].
In a randomized controlled trial of 218 non-obese people, a 2-year CR diet reduced circulating TNFα levels and strikingly decreased cardiometabolic risk markers, such as cholesterol and triglycerides, without any intervention-related adverse effects [311]. So far, there is no human study reporting a significant effect of CR on longevity. Large and extensive studies with genetically diverse populations are needed to solidify the promise of CR in humans.
Various metabolic impacts of CR include downregulation of mTOR and insulin signaling and activation of SIRT1, which all have broad implications on immune cell function [312]. CR is shown to delay T cell senescence in rhesus monkeys [313]. Furthermore, CD4+ and CD8+ naïve T cell pools were expanded, and thymic output and T cell proliferation were increased, but IFNγ production by CD8 + cells was reduced after CR. Although reducing the amount of calories taken seems to reverse age-induced metabolic changes and improve health and longevity, it is important to note that a few studies in rodents reported an impaired adaptive response and increased mortality against influenza A and West Nile viruses in elderly animals after CR [314, 315]. However, a recent mouse study revealed protective effects of CR against M. tuberculosis infection. This effect was related to metabolic shift characterized by mTOR inhibition but enhanced glycolysis and reduced FAO, along with increased autophagy [316]. mTOR inhibitor rapamycin acted synergistically with CR and further enhanced autophagy, leading to more efficient inhibition of M. tuberculosis.
Similar to CR, exercise is promising to interfere with immunosenescence. Regularly exercising older women had better NK and T cell functions compared to age-matched sedentary women [317]. Naïve T cell numbers and thymic output were higher in physically active elderly, similar to young adults, compared to sedentary ones [318]. They also had lower circulating IL-6 and higher IL-7, which is essential for T cell development. However, senescent CD8+ T cell numbers did not differ between groups. After an 8-week training program, immune cells of elderly adults displayed enhanced autophagy and downregulated NLRP3 inflammasome [319]. Exercise also improved mitophagy and mitochondrial biogenesis in skeletal muscle cells and immune cells alike, restoring the cellular metabolic status impaired by aging [320].
Apart from lifestyle interventions, chemical metabolic regulators are also investigated for their anti-aging potential. Metformin, safely used in humans for more than 60 years for its glucose-lowering effect, attenuates age-associated hallmarks through a plethora of mechanisms. These include activation of AMPK, inhibition of mTORC1, improved mitochondrial biogenesis, downregulation of insulin/IGF1 signaling, and activation of SIRT1 [321]. Furthermore, metformin delays stem cell aging and reduces telomere shortening. Overall, it seems to act on all hallmarks of aging. A large clinical trial of more than 3000 individuals aged 65–79 is currently being planned to assess the anti-aging potential of metformin (https://www.afar.org/tame-trial).
Everolimus, another mTOR inhibitor, attenuated immunosenescence and improved antibody responses to influenza vaccination in the elderly [322]. Even though most immune cell subsets were not altered in this study, T cells positive for programmed cell death protein 1 (PD-1), a marker of exhaustion, were markedly reduced. A follow-up study with 264 elderly subjects reported upregulated antiviral expression, improved response to influenza vaccination, and overall fewer infections [323].
SIRT1 activation is another approach to tackle immunosenescence. It is known to improve B cell proliferation and function, and therefore could help improve antibody responses declining with age [324]. SIRT1 can modulate metabolic pathways through protein and histone deacetylation [325]. Targets of SIRT1 include NF-κB, hypoxia-inducible factor 1-alpha (HIF1α), and FOXO transcription factors. Moreover, SIRT1 activation potentiates BCG-induced trained immunity response [326]. Despite mouse studies with SIRT1-activators showing delayed age-related phenotypes and increased lifespan [327, 328], there is no evidence suggesting that SIRT1 is associated with longevity in humans [329].
Resveratrol, a polyphenol compound found in red wine, is a potent activator of SIRT1 [330]. It is also shown to activate AMPK, therefore repressing mTOR signaling [331]. Apart from in vitro studies and inflammatory disease models displaying resveratrol’s antioxidant and anti-inflammatory activity [332], several mice studies reveal its antiviral capacity [333, 334]. In terms of longevity, studies failed to report a significant lifespan extension by resveratrol in healthy mice [327, 335]. However, in mice fed with a high-calorie diet, resveratrol shifted the transcriptional profile towards that of standard-fed mice [336]. It also improved insulin sensitivity and increased survival. Similar results were observed in rhesus monkeys on a high-fat, high-sugar diet [337]. Thirty-day supplementation of obese men with resveratrol induced metabolic changes through the AMPK-SIRT1 axis and reduced systemic inflammation, glucose, and triglyceride levels [338]. However, a similar study did not report any beneficial effects of resveratrol [339].
Overall, there are highly promising therapeutic approaches targeting metabolic pathways underlying immunosenescence and age-associated metabolic diseases. However, large-scale randomized control trials in humans are needed to see whether these exciting observations in non-human primates and smaller model organisms are translatable for human use.
Strategies Modulating Epigenetics
Epigenetic interventions have been employed for several age-related diseases, e.g., cancer, diabetes, and Alzheimer’s disease; however, only a few studies specifically target age-dependent changes in the epigenetic structure [340]. Instead, metabolic interventions employed to halt immunoaging also work by altering the age-associated epigenetic landscape. Resveratrol, CR, and metformin are three promising therapy options reconfiguring age-related DNA methylation and histone modifications in the elderly.
An intriguing study revealed that regenerating the thymus resulted in a 2.5-year younger epigenetic age [341]. Participants between 51 and 65 years of age received a 1-year treatment with recombinant human growth hormone, dehydroepiandrosterone (DHEA), which is a steroid hormone precursor, and metformin. The treatment led to restored functional thymic mass, changes in the immune cell subsets, and cytokine production, as well as altered epigenetic profile, which was associated with younger age.
Rhesus monkeys, who were exposed to 40% caloric restriction, were late to display the methylation changes found in the older monkeys [342]. Although this study does not provide direct evidence of a longer lifespan associated with delayed methylation drift, it suggests that CR could be used to slow down the aging process. In line with this, improving the lifespan of mice with resveratrol or CR resulted in slower epigenetic aging [343]. Life-long CR has also shown to prevent age-related DNA methylation changes in the brain, providing neuroprotection [344].
A few studies explain how CR could affect epigenetics. These mechanisms include decreased histone acetylation mediated by increased SIRT1 expression, higher DNA methyltransferase (DNMT) activity, and hypermethylation of specific regulatory genes, such as Ras [340]. Similarly, metformin acts on epigenetic marks via activating SIRT1 and inhibiting HDACs [345]. To our knowledge, there is no research investigating the effects of CR on aging-related epigenetic alterations, possibly due to the limitations of implementing such long-term interventions on humans.
Potential Treatments Targeting Microbiota
Since gut microbiota regulates host metabolism, anti-aging interventions targeting metabolism inevitably affect the gut microbiota. As an example, besides acting on metabolic pathways, metformin modulates the gut microbiota. A study investigating the effects of metformin in obese and aged mice found a decrease in IL-1β and IL-6 in the epididymal fat, which was associated with changes in the gut microbes [346]. Furthermore, type 2 diabetes patients who take metformin had a higher abundance of Akkermansia in their guts [347], which was correlated with lower bacterial translocation and risk of dysbiosis [348]. In line with these, metformin reduced age-related leaky gut and inflammation in mice [349].
Another treatment strategy to halt immunoaging by targeting the microbiota is the use of pro- and prebiotics. Probiotics are supplements containing live microorganisms, while prebiotics is substrates that microorganisms can utilize for a living [350]. Although there is conflicting evidence, studies suggest that regular probiotics use can modulate the diversity and abundance of the gut microbes, decreasing the incidence of dysbiosis [351, 352]. Probiotics are associated with improved immune responses evident from increased B and T cell counts, enhanced NK cell activity [353] and higher IgA production against influenza virus in older individuals [354]. Furthermore, supplementation with probiotics helped reduce the growth of opportunistic bacteria Clostridium difficile among the elderly [355]. Contrary to these findings, a meta-analysis of 10 randomized controlled studies showed no beneficial effect of probiotics on decreasing inflammatory cytokine production [356].
The combination of probiotics with prebiotics, i.e., synbiotics, also has beneficial effects, like probiotics supplementation. Two months of treatment in elderly individuals with a synbiotic formula significantly improved the metabolic syndrome parameters in circulation and decreased inflammatory proteins, such as TNFα and C-reactive protein [357]. A double-blind 4-week symbiotic treatment study reported an increase in Bifidobacteria, Actinobacteria, Firmicutes, and the metabolite butyrate in the treatment group compared to placebo, while Proteobacteria and pro-inflammatory cytokines were lower [358].
Caloric restriction could be another treatment strategy to improve cognitive functions, metabolic parameters, and gut microbiota in the elderly. CR slowed the cognitive decline in a mouse model of Alzheimer’s disease, associated with increased Bacteroides in the guts. Aged mice receiving 30% fewer calories for 2 months displayed significant shifts in their microbiota towards a more balanced composition similar to that of young mice [359]. Lifelong CR induced more extensive changes in the microbiota, reduced the concentration of inflammatory peptides, and increased the lifespan of mice [360]. However, a recent study revealed that severe CR, more than 50%, disrupts the diversity of microbiota and leads to the growth of pathogenic bacteria C. difficile [361]. Thus, it is critical to carefully determine the extent and duration of CR.
Interventions for Brain Aging
Physical exercise is an excellent way of promoting brain health. Exercise counteracts cognitive impairment, reduces dementia risk, improves spatial memory, and enhances neuroplasticity [362]. Physical activity can attenuate the effects of risk alleles for memory impairment [363] and protect against the development of Alzheimer’s disease [364, 365]. A systematic review of 16 studies with a total of 163,797 participants reported that regular exercise led to 28% and 45% risk reduction in dementia and Alzheimer’s, respectively [366]. Of note, exercise-associated risk reduction was observed in most of the individual studies irrespective of the frequency and intensity of the exercise.
Studies suggest antioxidant and anti-inflammatory effects of exercise as potential mechanisms behind neuroprotection [367, 368]. Anti-inflammatory consequences of exercise include reduced circulating IL-6 but increased IL-10 and IL-1RA, lower numbers of Treg, and higher numbers of inflammatory monocytes in circulation, and inhibited monocyte function [369]. Besides these, physical exercise is associated with reduced senescent T cells, increased NK cell cytotoxicity and neutrophil phagocytosis, and longer telomeres in leukocytes [370]. Additionally, moderate cardiovascular exercise improved seroprotection after influenza vaccination in the elderly [371]. Slowing down immunosenescence would limit brain aging and cognitive decline through improved immunosurveillance and repair of the CNS.
Moreover, even a single exercise session increases BDNF levels which is further enhanced with regular exercise [372]. Interestingly, the exercise-related increase in BDNF is more pronounced in males compared to females. Ketone bodies are also shown to induce BDNF expression [373, 374], possibly contributing to the neuroprotective effect of ketogenic diets in neurological diseases [375].
CR is another intervention shown to prevent neuronal damage. It leads to increased BDNF expression and enhanced neurogenesis [376], causes an energetic shift from glycolysis to the use of ketone bodies, protects white matter integrity, and improves long-term memory in mice [377]. In rats, an alternate-day CR regimen promotes neuronal resistance to chemically induced damage [378]. One mechanism of CR-induced neuroprotection is likely due to the suppression of oxidative stress in the brain [379, 380]. However, severe CR with 50% reduction of calorie intake was reported to cause depressive behavior in rats [381]. In mouse models of Alzheimer’s disease, CR is able to limit amyloid plaque deposition [382, 383], possibly through a mechanism involving SIRT1 activation [384].
Despite all the positive results in rodents, neuroprotective effects of CR are not very clear in non-human primates, while large human studies are lacking [385]. Nevertheless, a small randomized controlled trial with humans resulted in no significant improvement in cognitive function [386]. Another clinical study on older adults showed improved memory scores upon 3 months of CR [387]. Improved memory, along with higher functional connectivity in the hippocampus, was reported in obese women that underwent a 3-month CR diet [388]. More extensive human studies with CR are necessary to understand the extent of the neuroprotective effects.
Interestingly, BCG vaccination was recently shown to reduce the risk of Alzheimer’s and Parkinson’s diseases in bladder cancer patients treated with BCG immunotherapy, compared to non-treated patients [389, 390]. In bladder cancer treatment, BCG is applied directly into the bladder, rather than the usual intradermal route of administration. Exciting future research projects would be assessing the effects of intradermal BCG on neurodegenerative diseases and investigating the underlying mechanisms to find out if trained immunity plays a role in the neuroprotective effects. Currently, a clinical trial is underway using intradermal BCG injections in late-onset Alzheimer’s patients (NCT04449926).
Concluding Remarks
Biological aging is a complex process involving all systems of the organism. The immune system is at the very center of it, interacting with all the others. The aging immune system is a culprit for the high susceptibility of the elderly to infections and age-related metabolic and neurodegenerative diseases, among others. Therefore, improving innate and adaptive immunological responses is immensely important to reduce infection-related morbidity and mortality and enhance vaccine responsiveness in older individuals. Here, we also presented a large body of research hinting towards new roles of immune memory in metabolic regulation and maintaining a healthy central nervous system. Approaching aging from all angles, with immunity as a central node, and designing anti-aging interventions targeting the common mechanisms ubiquitously affected by aging is a sensible way to further research. Behavioral interventions such as caloric restriction and physical exercise as well as pharmacological agents such as metformin and resveratrol are able to regulate many facets of aging and have yielded promising results in animal models and humans. A comprehensive strategy is essential for human beings striving to lead long lives with healthy guts, functional brains, and free of severe infections.
References
Nations U (2015) World population ageing. Available at https://www.unorg/en/development/desa/population/publications/pdf/ageing/WPA2015_Highlightspdf
Bulut O, Kilic G, Dominguez-Andres J, Netea MG (2020) Overcoming immune dysfunction in the elderly: trained immunity as a novel approach. Int Immunol 32(12):741–753. https://doi.org/10.1093/intimm/dxaa052
Pawelec G (2018) Age and immunity: what is immunosenescence? Exp Gerontol 105:4–9. https://doi.org/10.1016/j.exger.2017.10.024
Perrotta F, Corbi G, Mazzeo G, Boccia M, Aronne L, D’Agnano V et al (2020) COVID-19 and the elderly: insights into pathogenesis and clinical decision-making. Aging Clin Exp Res 32(8):1599–1608. https://doi.org/10.1007/s40520-020-01631-y
Organization WH (2020) Status of COVID-19 Vaccines within WHO EUL/PQ evaluation process. Available at https://www.extranetwhoint/pqweb/sites/default/files/documents/Status_of_COVID-19_Vaccines_within_WHO_EUL-PQ_evaluation_process-16June2021_Finalpdf
Spinelli R, Parrillo L, Longo M, Florese P, Desiderio A, Zatterale F et al (2020) Molecular basis of ageing in chronic metabolic diseases. J Endocrinol Invest 43(10):1373–1389. https://doi.org/10.1007/s40618-020-01255-z
Hou Y, Dan X, Babbar M, Wei Y, Hasselbalch SG, Croteau DL et al (2019) Ageing as a risk factor for neurodegenerative disease. Nat Rev Neurol 15(10):565–581. https://doi.org/10.1038/s41582-019-0244-7
Netea MG, Schlitzer A, Placek K, Joosten LAB, Schultze JL (2019) Innate and adaptive immune memory: an evolutionary continuum in the host’s response to pathogens. Cell Host Microbe 25(1):13–26. https://doi.org/10.1016/j.chom.2018.12.006
Germain RN (2002) T-cell development and the CD4-CD8 lineage decision. Nat Rev Immunol 2(5):309–322. https://doi.org/10.1038/nri798
den Braber I, Mugwagwa T, Vrisekoop N, Westera L, Mogling R, de Boer AB et al (2012) Maintenance of peripheral naive T cells is sustained by thymus output in mice but not humans. Immunity 36(2):288–297. https://doi.org/10.1016/j.immuni.2012.02.006
Gourley TS, Wherry EJ, Masopust D, Ahmed R (2004) Generation and maintenance of immunological memory. Semin Immunol 16(5):323–333. https://doi.org/10.1016/j.smim.2004.08.013
Thome JJ, Bickham KL, Ohmura Y, Kubota M, Matsuoka N, Gordon C et al (2016) Early-life compartmentalization of human T cell differentiation and regulatory function in mucosal and lymphoid tissues. Nat Med 22(1):72–77. https://doi.org/10.1038/nm.4008
Sakaguchi S, Yamaguchi T, Nomura T, Ono M (2008) Regulatory T cells and immune tolerance. Cell 133(5):775–787. https://doi.org/10.1016/j.cell.2008.05.009
Rosenblum MD, Way SS, Abbas AK (2016) Regulatory T cell memory. Nat Rev Immunol 16(2):90–101. https://doi.org/10.1038/nri.2015.1
Willinger T, Freeman T, Hasegawa H, McMichael AJ, Callan MF (2005) Molecular signatures distinguish human central memory from effector memory CD8 T cell subsets. J Immunol 175(9):5895–5903. https://doi.org/10.4049/jimmunol.175.9.5895
Gattinoni L, Lugli E, Ji Y, Pos Z, Paulos CM, Quigley MF et al (2011) A human memory T cell subset with stem cell-like properties. Nat Med 17(10):1290–1297. https://doi.org/10.1038/nm.2446
Henson SM, Lanna A, Riddell NE, Franzese O, Macaulay R, Griffiths SJ et al (2014) p38 signaling inhibits mTORC1-independent autophagy in senescent human CD8(+) T cells. J Clin Invest 124(9):4004–4016. https://doi.org/10.1172/JCI75051
Thome JJ, Yudanin N, Ohmura Y, Kubota M, Grinshpun B, Sathaliyawala T et al (2014) Spatial map of human T cell compartmentalization and maintenance over decades of life. Cell 159(4):814–828. https://doi.org/10.1016/j.cell.2014.10.026
Sathaliyawala T, Kubota M, Yudanin N, Turner D, Camp P, Thome JJ et al (2013) Distribution and compartmentalization of human circulating and tissue-resident memory T cell subsets. Immunity 38(1):187–197. https://doi.org/10.1016/j.immuni.2012.09.020
Kumar BV, Connors TJ, Farber DL (2018) Human T Cell Development, localization, and function throughout life. Immunity 48(2):202–213. https://doi.org/10.1016/j.immuni.2018.01.007
Zens KD, Chen JK, Farber DL (2016). Vaccine-generated lung tissue-resident memory T cells provide heterosubtypic protection to influenza infection. JCI Insight 1(10). https://doi.org/10.1172/jci.insight.85832
Shin HIwasaki A (2012) A vaccine strategy that protects against genital herpes by establishing local memory T cells. Nature 491(7424):463–467. https://doi.org/10.1038/nature11522
Liu W, Zeng Z, Luo S, Hu C, Xu N, Huang A et al (2019) Gastric subserous vaccination with Helicobacter pylori vaccine: an attempt to establish tissue-resident CD4+ memory T cells and induce prolonged protection. Front Immunol 10:1115. https://doi.org/10.3389/fimmu.2019.01115
Lapuente D, Storcksdieck Genannt Bonsmann M, Maaske A, Stab V, Heinecke V, Watzstedt K et al (2018) IL-1beta as mucosal vaccine adjuvant: the specific induction of tissue-resident memory T cells improves the heterosubtypic immunity against influenza A viruses. Mucosal Immunol 11(4):1265–1278. https://doi.org/10.1038/s41385-018-0017-4
Muller-Sieburg CE, Sieburg HB, Bernitz JM, Cattarossi G (2012) Stem cell heterogeneity: implications for aging and regenerative medicine. Blood 119(17):3900–3907. https://doi.org/10.1182/blood-2011-12-376749
Rube CE, Fricke A, Widmann TA, Furst T, Madry H, Pfreundschuh M et al (2011) Accumulation of DNA damage in hematopoietic stem and progenitor cells during human aging. PLoS ONE 6(3):e17487. https://doi.org/10.1371/journal.pone.0017487
Rajagopalan S, Moyle MW, Joosten I, Long EO (2010) DNA-PKcs controls an endosomal signaling pathway for a proinflammatory response by natural killer cells. Sci Signal 3(110):ra14. https://doi.org/10.1126/scisignal.2000467
Morales AJ, Carrero JA, Hung PJ, Tubbs AT, Andrews JM, Edelson BT et al (2017) A type I IFN-dependent DNA damage response regulates the genetic program and inflammasome activation in macrophages. Elife 6. https://doi.org/10.7554/eLife.24655
Palmer DB (2013) The effect of age on thymic function. Front Immunol 4:316. https://doi.org/10.3389/fimmu.2013.00316
Shanley DP, Aw D, Manley NR, Palmer DB (2009) An evolutionary perspective on the mechanisms of immunosenescence. Trends Immunol 30(7):374–381. https://doi.org/10.1016/j.it.2009.05.001
Jamieson BD, Douek DC, Killian S, Hultin LE, Scripture-Adams DD, Giorgi JV et al (1999) Generation of functional thymocytes in the human adult. Immunity 10(5):569–575. https://doi.org/10.1016/s1074-7613(00)80056-4
Thome JJ, Grinshpun B, Kumar BV, Kubota M, Ohmura Y, Lerner H et al (2016) Longterm maintenance of human naive T cells through in situ homeostasis in lymphoid tissue sites. Sci Immunol 1(6). https://doi.org/10.1126/sciimmunol.aah6506
Andrew D, Aspinall R (2002) Age-associated thymic atrophy is linked to a decline in IL-7 production. Exp Gerontol 37(2–3):455–463. https://doi.org/10.1016/s0531-5565(01)00213-3
Shitara S, Hara T, Liang B, Wagatsuma K, Zuklys S, Hollander GA et al (2013) IL-7 produced by thymic epithelial cells plays a major role in the development of thymocytes and TCRgammadelta+ intraepithelial lymphocytes. J Immunol 190(12):6173–6179. https://doi.org/10.4049/jimmunol.1202573
Schulz AR, Malzer JN, Domingo C, Jurchott K, Grutzkau A, Babel N et al (2015) Low thymic activity and dendritic cell numbers are associated with the immune response to primary viral infection in elderly humans. J Immunol 195(10):4699–4711. https://doi.org/10.4049/jimmunol.1500598
Goronzy JJ, Weyand CM (2017) Successful and maladaptive T cell aging. Immunity 46(3):364–378. https://doi.org/10.1016/j.immuni.2017.03.010
Di Benedetto S, Derhovanessian E, Steinhagen-Thiessen E, Goldeck D, Muller L, Pawelec G (2015) Impact of age, sex and CMV-infection on peripheral T cell phenotypes: results from the Berlin BASE-II Study. Biogerontology 16(5):631–643. https://doi.org/10.1007/s10522-015-9563-2
Zuhair M, Smit GSA, Wallis G, Jabbar F, Smith C, Devleesschauwer B et al (2019) Estimation of the worldwide seroprevalence of cytomegalovirus: a systematic review and meta-analysis. Rev Med Virol 29(3):e2034. https://doi.org/10.1002/rmv.2034
Gordon CL, Miron M, Thome JJ, Matsuoka N, Weiner J, Rak MA et al (2017) Tissue reservoirs of antiviral T cell immunity in persistent human CMV infection. J Exp Med 214(3):651–667. https://doi.org/10.1084/jem.20160758
Saurwein-Teissl M, Lung TL, Marx F, Gschosser C, Asch E, Blasko I et al (2002) Lack of antibody production following immunization in old age: association with CD8(+)CD28(-) T cell clonal expansions and an imbalance in the production of Th1 and Th2 cytokines. J Immunol 168(11):5893–5899. https://doi.org/10.4049/jimmunol.168.11.5893
Derhovanessian E, Maier AB, Hahnel K, McElhaney JE, Slagboom EP, Pawelec G (2014) Latent infection with cytomegalovirus is associated with poor memory CD4 responses to influenza A core proteins in the elderly. J Immunol 193(7):3624–3631. https://doi.org/10.4049/jimmunol.1303361
Simanek AM, Dowd JB, Pawelec G, Melzer D, Dutta A, Aiello AE (2011) Seropositivity to cytomegalovirus, inflammation, all-cause and cardiovascular disease-related mortality in the United States. PLoS ONE 6(2):e16103. https://doi.org/10.1371/journal.pone.0016103
Furman D, Jojic V, Sharma S, Shen-Orr SS, Angel CJ, Onengut-Gumuscu S et al (2015) Cytomegalovirus infection enhances the immune response to influenza. Sci Transl Med 7(281):281ra43. https://doi.org/10.1126/scitranslmed.aaa2293
Qi Q, Liu Y, Cheng Y, Glanville J, Zhang D, Lee JY et al (2014) Diversity and clonal selection in the human T-cell repertoire. Proc Natl Acad Sci USA 111(36):13139–13144. https://doi.org/10.1073/pnas.1409155111
Gil A, Yassai MB, Naumov YN, Selin LK (2015) Narrowing of human influenza A virus-specific T cell receptor alpha and beta repertoires with increasing age. J Virol 89(8):4102–4116. https://doi.org/10.1128/JVI.03020-14
Quinn KM, Fox A, Harland KL, Russ BE, Li J, Nguyen THO et al (2018) Age-related decline in primary CD8(+) T cell responses is associated with the development of senescence in virtual memory CD8(+) T cells. Cell Rep 23(12):3512–3524. https://doi.org/10.1016/j.celrep.2018.05.057
Briceno O, Lissina A, Wanke K, Afonso G, von Braun A, Ragon K et al (2016) Reduced naive CD8(+) T-cell priming efficacy in elderly adults. Aging Cell 15(1):14–21. https://doi.org/10.1111/acel.12384
Gomez I, Marx F, Gould EA, Grubeck-Loebenstein B (2004) T cells from elderly persons respond to neoantigenic stimulation with an unimpaired IL-2 production and an enhanced differentiation into effector cells. Exp Gerontol 39(4):597–605. https://doi.org/10.1016/j.exger.2003.11.018
Kverneland AH, Streitz M, Geissler E, Hutchinson J, Vogt K, Boes D et al (2016) Age and gender leucocytes variances and references values generated using the standardized ONE-Study protocol. Cytometry A 89(6):543–564. https://doi.org/10.1002/cyto.a.22855
Hoffman W, Lakkis FG, Chalasani G (2016) B cells, antibodies, and more. Clin J Am Soc Nephrol 11(1):137–154. https://doi.org/10.2215/CJN.09430915
Akkaya M, Kwak K, Pierce SK (2020) B cell memory: building two walls of protection against pathogens. Nat Rev Immunol 20(4):229–238. https://doi.org/10.1038/s41577-019-0244-2
Dunn-Walters DK, Stewart AT, Sinclair EL, Serangeli I (2020) Age-related changes in B cells relevant to vaccine responses. Interdiscip Top Gerontol Geriatr 43:56–72. https://doi.org/10.1159/000504479
Ademokun A, Wu YC, Dunn-Walters D (2010) The ageing B cell population: composition and function. Biogerontology 11(2):125–137. https://doi.org/10.1007/s10522-009-9256-9
Hagen M, Derudder E (2020) Inflammation and the alteration of B-cell physiology in aging. Gerontology 66(2):105–113. https://doi.org/10.1159/000501963
Marie-Cardine A, Divay F, Dutot I, Green A, Perdrix A, Boyer O et al (2008) Transitional B cells in humans: characterization and insight from B lymphocyte reconstitution after hematopoietic stem cell transplantation. Clin Immunol 127(1):14–25. https://doi.org/10.1016/j.clim.2007.11.013
Pillai S, Cariappa A (2009) The follicular versus marginal zone B lymphocyte cell fate decision. Nat Rev Immunol 9(11):767–777. https://doi.org/10.1038/nri2656
Wallace DJ, Hahn BH (2019) Dubois' lupus erythematosus and related syndromes. B Cells and Generation of Antibodies. 9th ed: Elsevier p. 101–15. https://doi.org/10.1016/C2015-0-06776-4
Cerutti A, Cols M, Puga I (2013) Marginal zone B cells: virtues of innate-like antibody-producing lymphocytes. Nat Rev Immunol 13(2):118–132. https://doi.org/10.1038/nri3383
Wang H, Shin DM, Abbasi S, Jain S, Kovalchuk AL, Beaty N et al (2012) Expression of plasma cell alloantigen 1 defines layered development of B-1a B-cell subsets with distinct innate-like functions. Proc Natl Acad Sci USA 109(49):20077–20082. https://doi.org/10.1073/pnas.1212428109
Wong JB, Hewitt SL, Heltemes-Harris LM, Mandal M, Johnson K, Rajewsky K et al (2019) B-1a cells acquire their unique characteristics by bypassing the pre-BCR selection stage. Nat Commun 10(1):4768. https://doi.org/10.1038/s41467-019-12824-z
Montecino-Rodriguez E, Leathers H, Dorshkind K (2006) Identification of a B-1 B cell-specified progenitor. Nat Immunol 7(3):293–301. https://doi.org/10.1038/ni1301
Tsay GJ, Zouali M (2018) The interplay between innate-like B cells and other cell types in autoimmunity. Front Immunol 9:1064. https://doi.org/10.3389/fimmu.2018.01064
Rodriguez-Zhurbenko N, Quach TD, Hopkins TJ, Rothstein TL, Hernandez AM (2019) Human B-1 cells and B-1 cell antibodies change with advancing age. Front Immunol 10:483. https://doi.org/10.3389/fimmu.2019.00483
Rossi DJ, Bryder D, Zahn JM, Ahlenius H, Sonu R, Wagers AJ et al (2005) Cell intrinsic alterations underlie hematopoietic stem cell aging. Proc Natl Acad Sci USA 102(26):9194–9199. https://doi.org/10.1073/pnas.0503280102
Miller JP, Allman D (2003) The decline in B lymphopoiesis in aged mice reflects loss of very early B-lineage precursors. J Immunol 171(5):2326–2330. https://doi.org/10.4049/jimmunol.171.5.2326
Frasca D, Nguyen D, Van der Put E, Riley RL, Blomberg BB (2003) The age-related decrease in E47 DNA-binding does not depend on increased Id inhibitory proteins in bone marrow-derived B cell precursors. Front Biosci 8:a110–a116. https://doi.org/10.2741/1059
Riley RL, Blomberg BB, Frasca D (2005) B cells, E2A, and aging. Immunol Rev 205:30–47. https://doi.org/10.1111/j.0105-2896.2005.00268.x
Frasca D, Van Der Put E, Riley RL, Blomberg BB (2004) Age-related differences in the E2A-encoded transcription factor E47 in bone marrow-derived B cell precursors and in splenic B cells. Exp Gerontol 39(4):481–489. https://doi.org/10.1016/j.exger.2003.09.027
Beck K, Peak MM, Ota T, Nemazee D, Murre C (2009) Distinct roles for E12 and E47 in B cell specification and the sequential rearrangement of immunoglobulin light chain loci. J Exp Med 206(10):2271–2284. https://doi.org/10.1084/jem.20090756
Nipper AJ, Smithey MJ, Shah RC, Canaday DH, Landay AL (2018) Diminished antibody response to influenza vaccination is characterized by expansion of an age-associated B-cell population with low PAX5. Clin Immunol 193:80–87. https://doi.org/10.1016/j.clim.2018.02.003
Tabibian-Keissar H, Hazanov L, Schiby G, Rosenthal N, Rakovsky A, Michaeli M et al (2016) Aging affects B-cell antigen receptor repertoire diversity in primary and secondary lymphoid tissues. Eur J Immunol 46(2):480–492. https://doi.org/10.1002/eji.201545586
Gibson KL, Wu YC, Barnett Y, Duggan O, Vaughan R, Kondeatis E et al (2009) B-cell diversity decreases in old age and is correlated with poor health status. Aging Cell 8(1):18–25. https://doi.org/10.1111/j.1474-9726.2008.00443.x
Muggen AF, de Jong M, Wolvers-Tettero ILM, Kallemeijn MJ, Teodosio C, Darzentas N et al (2019) The presence of CLL-associated stereotypic B cell receptors in the normal BCR repertoire from healthy individuals increases with age. Immun Ageing 16:22. https://doi.org/10.1186/s12979-019-0163-x
Hao Y, O’Neill P, Naradikian MS, Scholz JL, Cancro MP (2011) A B-cell subset uniquely responsive to innate stimuli accumulates in aged mice. Blood 118(5):1294–1304. https://doi.org/10.1182/blood-2011-01-330530
Rubtsov AV, Rubtsova K, Fischer A, Meehan RT, Gillis JZ, Kappler JW et al (2011) Toll-like receptor 7 (TLR7)-driven accumulation of a novel CD11c(+) B-cell population is important for the development of autoimmunity. Blood 118(5):1305–1315. https://doi.org/10.1182/blood-2011-01-331462
Cancro MP (2020) Age-Associated B Cells. Annu Rev Immunol 38:315–340. https://doi.org/10.1146/annurev-immunol-092419-031130
Rubtsova K, Rubtsov AV, Cancro MP, Marrack P (2015) Age-associated B cells: a T-bet-dependent effector with roles in protective and pathogenic immunity. J Immunol 195(5):1933–1937. https://doi.org/10.4049/jimmunol.1501209
Ratliff M, Alter S, Frasca D, Blomberg BB, Riley RL (2013) In senescence, age-associated B cells secrete TNFalpha and inhibit survival of B-cell precursors. Aging Cell 12(2):303–311. https://doi.org/10.1111/acel.12055
Naradikian MS, Hao Y, Cancro MP (2016) Age-associated B cells: key mediators of both protective and autoreactive humoral responses. Immunol Rev 269(1):118–129. https://doi.org/10.1111/imr.12380
Jenks SA, Cashman KS, Zumaquero E, Marigorta UM, Patel AV, Wang X et al (2018) Distinct effector B cells induced by unregulated Toll-like receptor 7 contribute to pathogenic responses in systemic lupus erythematosus. Immunity 49(4):25–39. https://doi.org/10.1016/j.immuni.2018.08.015
Rubtsov AV, Rubtsova K, Kappler JW, Jacobelli J, Friedman RS, Marrack P (2015) CD11c-Expressing B cells are located at the T cell/B cell border in spleen and are potent APCs. J Immunol 195(1):71–79. https://doi.org/10.4049/jimmunol.1500055
Swain SL, Kugler-Umana O, Kuang Y, Zhang W (2017) The properties of the unique age-associated B cell subset reveal a shift in strategy of immune response with age. Cell Immunol 321:52–60. https://doi.org/10.1016/j.cellimm.2017.05.009
Blomberg BB, Frasca D (2013) Age effects on mouse and human B cells. Immunol Res 57(1–3):354–360. https://doi.org/10.1007/s12026-013-8440-9
Frasca D, Diaz A, Romero M, Landin AM, Blomberg BB (2011) Age effects on B cells and humoral immunity in humans. Ageing Res Rev 10(3):330–335. https://doi.org/10.1016/j.arr.2010.08.004
Pritz T, Lair J, Ban M, Keller M, Weinberger B, Krismer M et al (2015) Plasma cell numbers decrease in bone marrow of old patients. Eur J Immunol 45(3):738–746. https://doi.org/10.1002/eji.201444878
Frasca D, Diaz A, Romero M, Blomberg BB (2017) Human peripheral late/exhausted memory B cells express a senescent-associated secretory phenotype and preferentially utilize metabolic signaling pathways. Exp Gerontol 87(Pt A):113–120. https://doi.org/10.1016/j.exger.2016.12.001
Enani S, Przemska-Kosicka A, Childs CE, Maidens C, Dong H, Conterno L et al (2018) Impact of ageing and a synbiotic on the immune response to seasonal influenza vaccination; a randomised controlled trial. Clin Nutr 37(2):443–451. https://doi.org/10.1016/j.clnu.2017.01.011
Frasca D, Blomberg BB (2020) Aging induces B cell defects and decreased antibody responses to influenza infection and vaccination. Immun Ageing 17(1):37. https://doi.org/10.1186/s12979-020-00210-z
Howard WA, Gibson KL, Dunn-Walters DK (2006) Antibody quality in old age. Rejuvenation Res 9(1):117–125. https://doi.org/10.1089/rej.2006.9.117
Kogut I, Scholz JL, Cancro MP, Cambier JC (2012) B cell maintenance and function in aging. Semin Immunol 24(5):342–349. https://doi.org/10.1016/j.smim.2012.04.004
Quintin J, Saeed S, Martens JHA, Giamarellos-Bourboulis EJ, Ifrim DC, Logie C et al (2012) Candida albicans infection affords protection against reinfection via functional reprogramming of monocytes. Cell Host Microbe 12(2):223–232. https://doi.org/10.1016/j.chom.2012.06.006
Gourbal B, Pinaud S, Beckers GJM, Van Der Meer JWM, Conrath U, Netea MG (2018) Innate immune memory: an evolutionary perspective. Immunol Rev 283(1):21–40. https://doi.org/10.1111/imr.12647
Milutinovic B, Kurtz J (2016) Immune memory in invertebrates. Semin Immunol 28(4):328–342. https://doi.org/10.1016/j.smim.2016.05.004
Netea MG, Joosten LA, Latz E, Mills KH, Natoli G, Stunnenberg HG et al (2016) Trained immunity: A program of innate immune memory in health and disease. Science 352(6284)1098. https://doi.org/10.1126/science.aaf1098
Bekkering S, Blok BA, Joosten LA, Riksen NP, van Crevel R, Netea MG (2016) In vitro experimental model of trained innate immunity in human primary monocytes. Clin Vaccine Immunol 23(12):926–933. https://doi.org/10.1128/CVI.00349-16
Dou Y, Fu B, Sun R, Li W, Hu W, Tian Z et al (2015) Influenza vaccine induces intracellular immune memory of human NK cells. PLoS ONE 10(3):e0121258. https://doi.org/10.1371/journal.pone.0121258
Placek K, Schultze JL, Netea MG (2019) Immune memory characteristics of innate lymphoid cells. Curr Opin Infect Dis 32(3):196–203. https://doi.org/10.1097/QCO.0000000000000540
Hole CR, Wager CML, Castro-Lopez N, Campuzano A, Cai H, Wozniak KL et al (2019) Induction of memory-like dendritic cell responses in vivo. Nat Commun 10(1):2955. https://doi.org/10.1038/s41467-019-10486-5
Moorlag S, Rodriguez-Rosales YA, Gillard J, Fanucchi S, Theunissen K, Novakovic B et al (2020) BCG vaccination induces long-term functional reprogramming of human neutrophils. Cell Rep 33(7):108387. https://doi.org/10.1016/j.celrep.2020.108387
Kleinnijenhuis J, Quintin J, Preijers F, Joosten LA, Ifrim DC, Saeed S et al (2012) Bacille Calmette-Guerin induces NOD2-dependent nonspecific protection from reinfection via epigenetic reprogramming of monocytes. Proc Natl Acad Sci USA 109(43):17537–17542. https://doi.org/10.1073/pnas.1202870109
Aaby P, Martins CL, Garly ML, Bale C, Andersen A, Rodrigues A et al (2010) Non-specific effects of standard measles vaccine at 4.5 and 9 months of age on childhood mortality: randomised controlled trial. BMJ 341:c6495. https://doi.org/10.1136/bmj.c6495
Nielsen S, Khalek MA, Benn CS, Aaby P, Hanifi SMA (2021) National immunisation campaigns with oral polio vaccine may reduce all-cause mortality: analysis of 2004–2019 demographic surveillance data in rural Bangladesh. EClinicalMedicine 36:100886. https://doi.org/10.1016/j.eclinm.2021.100886
Keating ST, Groh L, Thiem K, Bekkering S, Li Y, Matzaraki V et al (2020) Rewiring of glucose metabolism defines trained immunity induced by oxidized low-density lipoprotein. J Mol Med (Berl) 98(6):819–831. https://doi.org/10.1007/s00109-020-01915-w
Arts RJ, Novakovic B, Ter Horst R, Carvalho A, Bekkering S, Lachmandas E et al (2016) Glutaminolysis and fumarate accumulation integrate immunometabolic and epigenetic programs in trained immunity. Cell Metab 24(6):807–819. https://doi.org/10.1016/j.cmet.2016.10.008
Netea MG, Quintin J, van der Meer JW (2011) Trained immunity: a memory for innate host defense. Cell Host Microbe 9(5):355–361. https://doi.org/10.1016/j.chom.2011.04.006
Netea MG, Dominguez-Andres J, Barreiro LB, Chavakis T, Divangahi M, Fuchs E et al (2020) Defining trained immunity and its role in health and disease. Nat Rev Immunol 20(6):375–388. https://doi.org/10.1038/s41577-020-0285-6
Patel AA, Zhang Y, Fullerton JN, Boelen L, Rongvaux A, Maini AA et al (2017) The fate and lifespan of human monocyte subsets in steady state and systemic inflammation. J Exp Med 214(7):1913–1923. https://doi.org/10.1084/jem.20170355
Mitroulis I, Ruppova K, Wang B, Chen LS, Grzybek M, Grinenko T et al (2018) Modulation of myelopoiesis progenitors is an integral component of trained immunity. Cell 172(1–2):147–61. https://doi.org/10.1016/j.cell.2017.11.034
Kaufmann E, Sanz J, Dunn JL, Khan N, Mendonca LE, Pacis A et al (2018) BCG educates hematopoietic stem cells to generate protective innate immunity against tuberculosis. Cell, 172(1–2):176–90. https://doi.org/10.1016/j.cell.2017.12.031
Cirovic B, de Bree LCJ, Groh L, Blok BA, Chan J, van der Velden W et al (2020) BCG vaccination in humans elicits trained immunity via the hematopoietic progenitor compartment. Cell Host Microbe 28(2):322–34. https://doi.org/10.1016/j.chom.2020.05.014
Pereira B, Xu XN, Akbar AN (2020) Targeting inflammation and immunosenescence to improve vaccine responses in the elderly. Front Immunol 11:583019. https://doi.org/10.3389/fimmu.2020.583019
Koeken VA, de Bree LCJ, Mourits VP, Moorlag SJ, Walk J, Cirovic B et al (2020) BCG vaccination in humans inhibits systemic inflammation in a sex-dependent manner. J Clin Invest 130(10):5591–5602. https://doi.org/10.1172/JCI133935
Giamarellos-Bourboulis EJ, Tsilika M, Moorlag S, Antonakos N, Kotsaki A, Dominguez-Andres J et al (2020) Activate: randomized clinical trial of BCG vaccination against infection in the elderly. Cell 183(2):315–23. https://doi.org/10.1016/j.cell.2020.08.051
Wardhana DEA, Sultana A, Mandang VV, Jim E (2011) The efficacy of Bacillus Calmette-Guerin vaccinations for the prevention of acute upper respiratory tract infection in the elderly. Acta Med Indones 43(3):185–190
Ohrui T, Nakayama K, Fukushima T, Chiba H, Sasaki H (2005) Prevention of elderly pneumonia by pneumococcal, influenza and BCG vaccinations. Nihon Ronen Igakkai Zasshi 42(1):34–36. https://doi.org/10.3143/geriatrics.42.34
Lange C, Aaby P, Behr MA, Donald PR, Kaufmann SHE, Netea MG et al (2021) 100 years of Mycobacterium bovis bacille Calmette-Guerin. Lancet Infect Dis. https://doi.org/10.1016/S1473-3099(21)00403-5
Tsilika M, Taks E, Dolianitis K, Kotsaki A, Leventogiannis K, Damoulari C et al (2021) Activate-2: a double-blind randomized trial of BCG vaccination against COVID19 in individuals at risk
Rivas MN, Ebinger JE, Wu M, Sun N, Braun J, Sobhani K et al (2021) BCG vaccination history associates with decreased SARS-CoV-2 seroprevalence across a diverse cohort of health care workers. J Clin Invest 131(2). https://doi.org/10.1172/JCI145157
Ursell LK, Metcalf JL, Parfrey LW, Knight R (2012) Defining the human microbiome. Nutr Rev 70(Suppl 1):S38–44. https://doi.org/10.1111/j.1753-4887.2012.00493.x
Franceschi C, Garagnani P, Parini P, Giuliani C, Santoro A (2018) Inflammaging: a new immune-metabolic viewpoint for age-related diseases. Nat Rev Endocrinol 14(10):576–590. https://doi.org/10.1038/s41574-018-0059-4
Myers DR, Wheeler B, Roose JP (2019) mTOR and other effector kinase signals that impact T cell function and activity. Immunol Rev 291(1):134–153. https://doi.org/10.1111/imr.12796
Donnelly RP, Finlay DK (2015) Glucose, glycolysis and lymphocyte responses. Mol Immunol 68(2 Pt C):513–9. https://doi.org/10.1016/j.molimm.2015.07.034
M CML ND (2017) Lehninger principles of biochemistry. 7th ed
Lochner M, Berod L, Sparwasser T (2015) Fatty acid metabolism in the regulation of T cell function. Trends Immunol 36(2):81–91. https://doi.org/10.1016/j.it.2014.12.005
Martinez-Reyes I, Chandel NS (2020) Mitochondrial TCA cycle metabolites control physiology and disease. Nat Commun 11(1):102. https://doi.org/10.1038/s41467-019-13668-3
Peng M, Yin N, Chhangawala S, Xu K, Leslie CS, Li MO (2016) Aerobic glycolysis promotes T helper 1 cell differentiation through an epigenetic mechanism. Science 354(6311):481–484. https://doi.org/10.1126/science.aaf6284
Pougovkina O, te Brinke H, Ofman R, van Cruchten AG, Kulik W, Wanders RJ et al (2014) Mitochondrial protein acetylation is driven by acetyl-CoA from fatty acid oxidation. Hum Mol Genet 23(13):3513–3522. https://doi.org/10.1093/hmg/ddu059
Weinberg SE, Sena LA, Chandel NS (2015) Mitochondria in the regulation of innate and adaptive immunity. Immunity 42(3):406–417. https://doi.org/10.1016/j.immuni.2015.02.002
Balyan R, Gautam N, Gascoigne NRJ (2020) The ups and downs of metabolism during the lifespan of a T cell. Int J Mol Sci 21(21). https://doi.org/10.3390/ijms21217972
Rathmell JC, Farkash EA, Gao W, Thompson CB (2001) IL-7 enhances the survival and maintains the size of naive T cells. J Immunol 167(12):6869–6876. https://doi.org/10.4049/jimmunol.167.12.6869
Rathmell JC, Vander Heiden MG, Harris MH, Frauwirth KA, Thompson CB (2000) In the absence of extrinsic signals, nutrient utilization by lymphocytes is insufficient to maintain either cell size or viability. Mol Cell 6(3):683–692. https://doi.org/10.1016/s1097-2765(00)00066-6
Maciolek JA, Pasternak JA, Wilson HL (2014) Metabolism of activated T lymphocytes. Curr Opin Immunol 27:60–74. https://doi.org/10.1016/j.coi.2014.01.006
Chang CH, Curtis JD, Maggi LB Jr, Faubert B, Villarino AV, O’Sullivan D et al (2013) Posttranscriptional control of T cell effector function by aerobic glycolysis. Cell 153(6):1239–1251. https://doi.org/10.1016/j.cell.2013.05.016
Gubser PM, Bantug GR, Razik L, Fischer M, Dimeloe S, Hoenger G et al (2013) Rapid effector function of memory CD8+ T cells requires an immediate-early glycolytic switch. Nat Immunol 14(10):1064–1072. https://doi.org/10.1038/ni.2687
van der Windt GJ, O’Sullivan D, Everts B, Huang SC, Buck MD, Curtis JD et al (2013) CD8 memory T cells have a bioenergetic advantage that underlies their rapid recall ability. Proc Natl Acad Sci USA 110(35):14336–14341. https://doi.org/10.1073/pnas.1221740110
van der Windt GJ, Everts B, Chang CH, Curtis JD, Freitas TC, Amiel E et al (2012) Mitochondrial respiratory capacity is a critical regulator of CD8+ T cell memory development. Immunity 36(1):68–78. https://doi.org/10.1016/j.immuni.2011.12.007
Buck MD, O’Sullivan D, Klein Geltink RI, Curtis JD, Chang CH, Sanin DE et al (2016) Mitochondrial dynamics controls T cell fate through metabolic programming. Cell 166(1):63–76. https://doi.org/10.1016/j.cell.2016.05.035
Lanna A, Gomes DC, Muller-Durovic B, McDonnell T, Escors D, Gilroy DW et al (2017) A sestrin-dependent Erk-Jnk-p38 MAPK activation complex inhibits immunity during aging. Nat Immunol 18(3):354–363. https://doi.org/10.1038/ni.3665
Lucas CL, Kuehn HS, Zhao F, Niemela JE, Deenick EK, Palendira U et al (2014) Dominant-activating germline mutations in the gene encoding the PI(3)K catalytic subunit p110delta result in T cell senescence and human immunodeficiency. Nat Immunol 15(1):88–97. https://doi.org/10.1038/ni.2771
Moskowitz DM, Zhang DW, Hu B, Le Saux S, Yanes RE, Ye Z et al (2017) Epigenomics of human CD8 T cell differentiation and aging. Sci Immunol 2(8). https://doi.org/10.1126/sciimmunol.aag0192
Ron-Harel N, Notarangelo G, Ghergurovich JM, Paulo JA, Sage PT, Santos D et al (2018) Defective respiration and one-carbon metabolism contribute to impaired naive T cell activation in aged mice. Proc Natl Acad Sci USA 115(52):13347–13352. https://doi.org/10.1073/pnas.1804149115
Puleston DJ, Zhang H, Powell TJ, Lipina E, Sims S, Panse I et al (2014) Autophagy is a critical regulator of memory CD8(+) T cell formation. Elife 3. https://doi.org/10.7554/eLife.03706
Yanes RE, Zhang H, Shen Y, Weyand CM, Goronzy JJ (2019) Metabolic reprogramming in memory CD4 T cell responses of old adults. Clin Immunol 207:58–67. https://doi.org/10.1016/j.clim.2019.07.003
Ruderman NB, Xu XJ, Nelson L, Cacicedo JM, Saha AK, Lan F et al (2010) AMPK and SIRT1: a long-standing partnership? Am J Physiol Endocrinol Metab 298(4):E751–E760. https://doi.org/10.1152/ajpendo.00745.2009
Jeng MY, Hull PA, Fei M, Kwon HS, Tsou CL, Kasler H et al (2018) Metabolic reprogramming of human CD8(+) memory T cells through loss of SIRT1. J Exp Med 215(1):51–62. https://doi.org/10.1084/jem.20161066
Tilly G, Doan-Ngoc TM, Yap M, Caristan A, Jacquemont L, Danger R et al (2017) IL-15 harnesses pro-inflammatory function of TEMRA CD8 in kidney-transplant recipients. Front Immunol 8:778. https://doi.org/10.3389/fimmu.2017.00778
Yu Y, Clippinger AJ, Alwine JC (2011) Viral effects on metabolism: changes in glucose and glutamine utilization during human cytomegalovirus infection. Trends Microbiol 19(7):360–367. https://doi.org/10.1016/j.tim.2011.04.002
Almanzar G, Schwaiger S, Jenewein B, Keller M, Herndler-Brandstetter D, Wurzner R et al (2005) Long-term cytomegalovirus infection leads to significant changes in the composition of the CD8+ T-cell repertoire, which may be the basis for an imbalance in the cytokine production profile in elderly persons. J Virol 79(6):3675–3683. https://doi.org/10.1128/JVI.79.6.3675-3683.2005
Chen S, de Craen AJ, Raz Y, Derhovanessian E, Vossen AC, Westendorp RG et al (2012) Cytomegalovirus seropositivity is associated with glucose regulation in the oldest old. Results from the Leiden 85-plus Study. Immun Ageing 9(1):18. https://doi.org/10.1186/1742-4933-9-18
Jellusova J, Rickert RC (2016) The PI3K pathway in B cell metabolism. Crit Rev Biochem Mol Biol 51(5):359–378. https://doi.org/10.1080/10409238.2016.1215288
Boothby M, Rickert RC (2017) Metabolic regulation of the immune humoral response. Immunity 46(5):743–755. https://doi.org/10.1016/j.immuni.2017.04.009
Waters LR, Ahsan FM, Wolf DM, Shirihai O and Teitell MA (2018) Initial B cell activation induces metabolic reprogramming and mitochondrial remodeling. iScience 5:99–109. https://doi.org/10.1016/j.isci.2018.07.005
Jang KJ, Mano H, Aoki K, Hayashi T, Muto A, Nambu Y et al (2015) Mitochondrial function provides instructive signals for activation-induced B-cell fates. Nat Commun 6:6750. https://doi.org/10.1038/ncomms7750
Choi SC, Morel L (2020) Immune metabolism regulation of the germinal center response. Exp Mol Med 52(3):348–355. https://doi.org/10.1038/s12276-020-0392-2
Ersching J, Efeyan A, Mesin L, Jacobsen JT, Pasqual G, Grabiner BC et al (2017) Germinal center selection and affinity maturation require dynamic regulation of mTORC1 kinase. Immunity 46(6):1045–58. https://doi.org/10.1016/j.immuni.2017.06.005
Luo W, Weisel F, Shlomchik MJ (2018) B cell receptor and CD40 signaling are rewired for synergistic induction of the c-Myc transcription factor in germinal center B cells. Immunity 48(2):313–26. https://doi.org/10.1016/j.immuni.2018.01.008
Torigoe M, Iwata S, Nakayamada S, Sakata K, Zhang M, Hajime M et al (2017) Metabolic reprogramming commits differentiation of human CD27(+)IgD(+) B cells to plasmablasts or CD27(-)IgD(-) cells. J Immunol 199(2):425–434. https://doi.org/10.4049/jimmunol.1601908
Chen M, Hong MJ, Sun H, Wang L, Shi X, Gilbert BE et al (2014) Essential role for autophagy in the maintenance of immunological memory against influenza infection. Nat Med 20(5):503–510. https://doi.org/10.1038/nm.3521
Chen M, Kodali S, Jang A, Kuai L, Wang J (2015) Requirement for autophagy in the long-term persistence but not initial formation of memory B cells. J Immunol 194(6):2607–2615. https://doi.org/10.4049/jimmunol.1403001
Jones DD, Gaudette BT, Wilmore JR, Chernova I, Bortnick A, Weiss BM et al (2016) mTOR has distinct functions in generating versus sustaining humoral immunity. J Clin Invest 126(11):4250–4261. https://doi.org/10.1172/JCI86504
Lam WY, Becker AM, Kennerly KM, Wong R, Curtis JD, Llufrio EM et al (2016) Mitochondrial pyruvate import promotes long-term survival of antibody-secreting plasma cells. Immunity 45(1):60–73. https://doi.org/10.1016/j.immuni.2016.06.011
Caro-Maldonado A, Wang R, Nichols AG, Kuraoka M, Milasta S, Sun LD et al (2014) Metabolic reprogramming is required for antibody production that is suppressed in anergic but exaggerated in chronically BAFF-exposed B cells. J Immunol 192(8):3626–3636. https://doi.org/10.4049/jimmunol.1302062
Clarke AJ, Riffelmacher T, Braas D, Cornall RJ, Simon AK (2018) B1a B cells require autophagy for metabolic homeostasis and self-renewal. J Exp Med 215(2):399–413. https://doi.org/10.1084/jem.20170771
Kurupati RK, Haut LH, Schmader KE, Ertl HC (2019) Age-related changes in B cell metabolism. Aging (Albany NY) 11(13):4367–4381. https://doi.org/10.18632/aging.102058
Kannan S, Dawany N, Kurupati R, Showe LC, Ertl HC (2016) Age-related changes in the transcriptome of antibody-secreting cells. Oncotarget 7(12):13340–13353. https://doi.org/10.18632/oncotarget.7958
Sainz N, Barrenetxe J, Moreno-Aliaga MJ, Martinez JA (2015) Leptin resistance and diet-induced obesity: central and peripheral actions of leptin. Metabolism 64(1):35–46. https://doi.org/10.1016/j.metabol.2014.10.015
Frasca D, Diaz A, Romero M, Blomberg BB (2020) Leptin induces immunosenescence in human B cells. Cell Immunol 348:103994. https://doi.org/10.1016/j.cellimm.2019.103994
Lana A, Valdes-Becares A, Buno A, Rodriguez-Artalejo F, Lopez-Garcia E (2017) Serum leptin concentration is associated with incident frailty in older adults. Aging Dis 8(2):240–249. https://doi.org/10.14336/AD.2016.0819
Frasca D, Ferracci F, Diaz A, Romero M, Lechner S, Blomberg BB (2016) Obesity decreases B cell responses in young and elderly individuals. Obesity (Silver Spring) 24(3):615–625. https://doi.org/10.1002/oby.21383
Alter G, Ottenhoff THM, Joosten SA (2018) Antibody glycosylation in inflammation, disease and vaccination. Semin Immunol 39:102–110. https://doi.org/10.1016/j.smim.2018.05.003
Kristic J, Vuckovic F, Menni C, Klaric L, Keser T, Beceheli I et al (2014) Glycans are a novel biomarker of chronological and biological ages. J Gerontol A Biol Sci Med Sci 69(7):779–789. https://doi.org/10.1093/gerona/glt190
Catera M, Borelli V, Malagolini N, Chiricolo M, Venturi G, Reis CA et al (2016) Identification of novel plasma glycosylation-associated markers of aging. Oncotarget 7(7):7455–7468. https://doi.org/10.18632/oncotarget.7059
Donohoe DR, Bultman SJ (2012) Metaboloepigenetics: interrelationships between energy metabolism and epigenetic control of gene expression. J Cell Physiol 227(9):3169–3177. https://doi.org/10.1002/jcp.24054
Cheng SC, Quintin J, Cramer RA, Shepardson KM, Saeed S, Kumar V et al (2014) mTOR- and HIF-1alpha-mediated aerobic glycolysis as metabolic basis for trained immunity. Science 345(6204):1250684. https://doi.org/10.1126/science.1250684
Arts RJW, Carvalho A, La Rocca C, Palma C, Rodrigues F, Silvestre R et al (2016) Immunometabolic pathways in BCG-induced trained immunity. Cell Rep 17(10):2562–2571. https://doi.org/10.1016/j.celrep.2016.11.011
Bekkering S, Arts RJW, Novakovic B, Kourtzelis I, van der Heijden C, Li Y et al (2018) Metabolic induction of trained immunity through the mevalonate pathway. Cell 172(1–2):135–46. https://doi.org/10.1016/j.cell.2017.11.025
van der Heijden C, Keating ST, Groh L, Joosten LAB, Netea MG, Riksen NP (2020) Aldosterone induces trained immunity: the role of fatty acid synthesis. Cardiovasc Res 116(2):317–328. https://doi.org/10.1093/cvr/cvz137
Moore LD, Le T, Fan G (2013) DNA methylation and its basic function. Neuropsychopharmacology 38(1):23–38. https://doi.org/10.1038/npp.2012.112
Maurano MT, Wang H, John S, Shafer A, Canfield T, Lee K et al (2015) Role of DNA methylation in modulating transcription factor occupancy. Cell Rep 12(7):1184–1195. https://doi.org/10.1016/j.celrep.2015.07.024
Hannum G, Guinney J, Zhao L, Zhang L, Hughes G, Sadda S et al (2013) Genome-wide methylation profiles reveal quantitative views of human aging rates. Mol Cell 49(2):359–367. https://doi.org/10.1016/j.molcel.2012.10.016
Horvath S (2013) DNA methylation age of human tissues and cell types. Genome Biol 14(10):R115. https://doi.org/10.1186/gb-2013-14-10-r115
Bocklandt S, Lin W, Sehl ME, Sanchez FJ, Sinsheimer JS, Horvath S et al (2011) Epigenetic predictor of age. PLoS ONE 6(6):e14821. https://doi.org/10.1371/journal.pone.0014821
Bollati V, Schwartz J, Wright R, Litonjua A, Tarantini L, Suh H et al (2009) Decline in genomic DNA methylation through aging in a cohort of elderly subjects. Mech Ageing Dev 130(4):234–239. https://doi.org/10.1016/j.mad.2008.12.003
Oakes CC, Smiraglia DJ, Plass C, Trasler JM, Robaire B (2003) Aging results in hypermethylation of ribosomal DNA in sperm and liver of male rats. Proc Natl Acad Sci USA 100(4):1775–1780. https://doi.org/10.1073/pnas.0437971100
Wei L, Liu B, Tuo J, Shen D, Chen P, Li Z et al (2012) Hypomethylation of the IL17RC promoter associates with age-related macular degeneration. Cell Rep 2(5):1151–1158. https://doi.org/10.1016/j.celrep.2012.10.013
Sun D, Luo M, Jeong M, Rodriguez B, Xia Z, Hannah R et al (2014) Epigenomic profiling of young and aged HSCs reveals concerted changes during aging that reinforce self-renewal. Cell Stem Cell 14(5):673–688. https://doi.org/10.1016/j.stem.2014.03.002
Taiwo O, Wilson GA, Emmett W, Morris T, Bonnet D, Schuster E et al (2013) DNA methylation analysis of murine hematopoietic side population cells during aging. Epigenetics 8(10):1114–1122. https://doi.org/10.4161/epi.26017
Broske AM, Vockentanz L, Kharazi S, Huska MR, Mancini E, Scheller M et al (2009) DNA methylation protects hematopoietic stem cell multipotency from myeloerythroid restriction. Nat Genet 41(11):1207–1215. https://doi.org/10.1038/ng.463
Tserel L, Kolde R, Limbach M, Tretyakov K, Kasela S, Kisand K et al (2015) Age-related profiling of DNA methylation in CD8+ T cells reveals changes in immune response and transcriptional regulator genes. Sci Rep 5:13107. https://doi.org/10.1038/srep13107
Dozmorov MG, Coit P, Maksimowicz-McKinnon K, Sawalha AH (2017) Age-associated DNA methylation changes in naive CD4(+) T cells suggest an evolving autoimmune epigenotype in aging T cells. Epigenomics 9(4):429–445. https://doi.org/10.2217/epi-2016-0143
Dozmorov MG (2015) Polycomb repressive complex 2 epigenomic signature defines age-associated hypermethylation and gene expression changes. Epigenetics 10(6):484–495. https://doi.org/10.1080/15592294.2015.1040619
Margueron R, Reinberg D (2011) The Polycomb complex PRC2 and its mark in life. Nature 469(7330):343–349. https://doi.org/10.1038/nature09784
Gorelik G, Fang JY, Wu A, Sawalha AH, Richardson B (2007) Impaired T cell protein kinase C delta activation decreases ERK pathway signaling in idiopathic and hydralazine-induced lupus. J Immunol 179(8):5553–5563. https://doi.org/10.4049/jimmunol.179.8.5553
Suarez-Alvarez B, Rodriguez RM, Schlangen K, Raneros AB, Marquez-Kisinousky L, Fernandez AF et al (2017) Phenotypic characteristics of aged CD4(+) CD28(null) T lymphocytes are determined by changes in the whole-genome DNA methylation pattern. Aging Cell 16(2):293–303. https://doi.org/10.1111/acel.12552
Zhao M, Qin J, Yin H, Tan Y, Liao W, Liu Q et al (2016) Distinct epigenomes in CD4(+) T cells of newborns, middle-ages and centenarians. Sci Rep 6:38411. https://doi.org/10.1038/srep38411
Roychoudhuri R, Hirahara K, Mousavi K, Clever D, Klebanoff CA, Bonelli M et al (2013) BACH2 represses effector programs to stabilize T(reg)-mediated immune homeostasis. Nature 498(7455):506–510. https://doi.org/10.1038/nature12199
Maltby VE, Lea RA, Graves MC, Sanders KA, Benton MC, Tajouri L et al (2018) Genome-wide DNA methylation changes in CD19(+) B cells from relapsing-remitting multiple sclerosis patients. Sci Rep 8(1):17418. https://doi.org/10.1038/s41598-018-35603-0
Lai AY, Mav D, Shah R, Grimm SA, Phadke D, Hatzi K et al (2013) DNA methylation profiling in human B cells reveals immune regulatory elements and epigenetic plasticity at Alu elements during B-cell activation. Genome Res 23(12):2030–2041. https://doi.org/10.1101/gr.155473.113
Duran-Ferrer M, Clot G, Nadeu F, Beekman R, Baumann T, Nordlund J et al (2020) The proliferative history shapes the DNA methylome of B-cell tumors and predicts clinical outcome. Nat Cancer 1(11):1066–1081. https://doi.org/10.1038/s43018-020-00131-2
Barwick BG, Scharer CD, Martinez RJ, Price MJ, Wein AN, Haines RR et al (2018) B cell activation and plasma cell differentiation are inhibited by de novo DNA methylation. Nat Commun 9(1):1900. https://doi.org/10.1038/s41467-018-04234-4
van der Heijden C, Noz MP, Joosten LAB, Netea MG, Riksen NP, Keating ST (2018) Epigenetics and trained immunity. Antioxid Redox Signal 29(11):1023–1040. https://doi.org/10.1089/ars.2017.7310
Strahl BD, Allis CD (2000) The language of covalent histone modifications. Nature 403(6765):41–45. https://doi.org/10.1038/47412
Li B, Carey M, Workman JL (2007) The role of chromatin during transcription. Cell 128(4):707–719. https://doi.org/10.1016/j.cell.2007.01.015
Graff J, Tsai LH (2013) Histone acetylation: molecular mnemonics on the chromatin. Nat Rev Neurosci 14(2):97–111. https://doi.org/10.1038/nrn3427
Yi SJ, Kim K (2020) New insights into the role of histone changes in aging. Int J Mol Sci 21(21). https://doi.org/10.3390/ijms21218241
Jasiulionis MG (2018) Abnormal epigenetic regulation of immune system during aging. Front Immunol 9:197. https://doi.org/10.3389/fimmu.2018.00197
Cheung P, Vallania F, Warsinske HC, Donato M, Schaffert S, Chang SE et al (2018) Single-cell chromatin modification profiling reveals increased epigenetic variations with aging. Cell 173(6):1385–97 e14. https://doi.org/10.1016/j.cell.2018.03.079
Ucar D, Marquez EJ, Chung CH, Marches R, Rossi RJ, Uyar A et al (2017) The chromatin accessibility signature of human immune aging stems from CD8(+) T cells. J Exp Med 214(10):3123–3144. https://doi.org/10.1084/jem.20170416
Palmer MJ, Mahajan VS, Trajman LC, Irvine DJ, Lauffenburger DA, Chen J (2008) Interleukin-7 receptor signaling network: an integrated systems perspective. Cell Mol Immunol 5(2):79–89. https://doi.org/10.1038/cmi.2008.10
Gleyzer N, Vercauteren K, Scarpulla RC (2005) Control of mitochondrial transcription specificity factors (TFB1M and TFB2M) by nuclear respiratory factors (NRF-1 and NRF-2) and PGC-1 family coactivators. Mol Cell Biol 25(4):1354–1366. https://doi.org/10.1128/MCB.25.4.1354-1366.2005
Kuwahara M, Suzuki J, Tofukuji S, Yamada T, Kanoh M, Matsumoto A et al (2014) The Menin-Bach2 axis is critical for regulating CD4 T-cell senescence and cytokine homeostasis. Nat Commun 5:3555. https://doi.org/10.1038/ncomms4555
Koohy H, Bolland DJ, Matheson LS, Schoenfelder S, Stellato C, Dimond A et al (2018) Genome organization and chromatin analysis identify transcriptional downregulation of insulin-like growth factor signaling as a hallmark of aging in developing B cells. Genome Biol 19(1):126. https://doi.org/10.1186/s13059-018-1489-y
Landreth KS, Narayanan R, Dorshkind K (1992) Insulin-like growth factor-I regulates pro-B cell differentiation. Blood 80(5):1207–1212
Saeed S, Quintin J, Kerstens HH, Rao NA, Aghajanirefah A, Matarese F et al (2014) Epigenetic programming of monocyte-to-macrophage differentiation and trained innate immunity. Science 345(6204):1251086. https://doi.org/10.1126/science.1251086
Pacis A, Tailleux L, Morin AM, Lambourne J, MacIsaac JL, Yotova V et al (2015) Bacterial infection remodels the DNA methylation landscape of human dendritic cells. Genome Res 25(12):1801–1811. https://doi.org/10.1101/gr.192005.115
Verma D, Parasa VR, Raffetseder J, Martis M, Mehta RB, Netea M et al (2017) Anti-mycobacterial activity correlates with altered DNA methylation pattern in immune cells from BCG-vaccinated subjects. Sci Rep 7(1):12305. https://doi.org/10.1038/s41598-017-12110-2
Das J, Verma D, Gustafsson M, Lerm M (2019) Identification of DNA methylation patterns predisposing for an efficient response to BCG vaccination in healthy BCG-naive subjects. Epigenetics 14(6):589–601. https://doi.org/10.1080/15592294.2019.1603963
Badal VD, Vaccariello ED, Murray ER, Yu KE, Knight R, Jeste DV et al (2020) The gut microbiome, aging, and longevity: a systematic review. Nutrients 12(12). https://doi.org/10.3390/nu12123759
Mazmanian SK, Liu CH, Tzianabos AO, Kasper DL (2005) An immunomodulatory molecule of symbiotic bacteria directs maturation of the host immune system. Cell 122(1):107–118. https://doi.org/10.1016/j.cell.2005.05.007
Cong Y, Feng T, Fujihashi K, Schoeb TR, Elson CO (2009) A dominant, coordinated T regulatory cell-IgA response to the intestinal microbiota. Proc Natl Acad Sci USA 106(46):19256–19261. https://doi.org/10.1073/pnas.0812681106
Zheng D, Liwinski T, Elinav E (2020) Interaction between microbiota and immunity in health and disease. Cell Res 30(6):492–506. https://doi.org/10.1038/s41422-020-0332-7
Zhao Q, Elson CO (2018) Adaptive immune education by gut microbiota antigens. Immunology 154(1):28–37. https://doi.org/10.1111/imm.12896
Peterson DA, McNulty NP, Guruge JL, Gordon JI (2007) IgA response to symbiotic bacteria as a mediator of gut homeostasis. Cell Host Microbe 2(5):328–339. https://doi.org/10.1016/j.chom.2007.09.013
Tanoue T, Atarashi K, Honda K (2016) Development and maintenance of intestinal regulatory T cells. Nat Rev Immunol 16(5):295–309. https://doi.org/10.1038/nri.2016.36
Zhang Z, Li J, Zheng W, Zhao G, Zhang H, Wang X et al (2016) Peripheral lymphoid volume expansion and maintenance are controlled by gut microbiota via RALDH+ dendritic cells. Immunity 44(2):330–342. https://doi.org/10.1016/j.immuni.2016.01.004
Hapfelmeier S, Lawson MA, Slack E, Kirundi JK, Stoel M, Heikenwalder M et al (2010) Reversible microbial colonization of germ-free mice reveals the dynamics of IgA immune responses. Science 328(5986):1705–1709. https://doi.org/10.1126/science.1188454
Round JL, Mazmanian SK (2009) The gut microbiota shapes intestinal immune responses during health and disease. Nat Rev Immunol 9(5):313–323. https://doi.org/10.1038/nri2515
Ragonnaud E, Biragyn A (2021) Gut microbiota as the key controllers of “healthy” aging of elderly people. Immun Ageing 18(1):2. https://doi.org/10.1186/s12979-020-00213-w
Rojas OL, Probstel AK, Porfilio EA, Wang AA, Charabati M, Sun T et al (2019) Recirculating intestinal IgA-producing cells regulate neuroinflammation via IL-10. Cell 176(3):610–24. https://doi.org/10.1016/j.cell.2018.11.035
Brown RL, Sequeira RP, Clarke TB (2017) The microbiota protects against respiratory infection via GM-CSF signaling. Nat Commun 8(1):1512. https://doi.org/10.1038/s41467-017-01803-x
Wilkins LJ, Monga M, Miller AW (2019) Defining dysbiosis for a cluster of chronic diseases. Sci Rep 9(1):12918. https://doi.org/10.1038/s41598-019-49452-y
Xu C, Zhu H, Qiu P (2019) Aging progression of human gut microbiota. BMC Microbiol 19(1):236. https://doi.org/10.1186/s12866-019-1616-2
Claesson MJ, Cusack S, O’Sullivan O, Greene-Diniz R, de Weerd H, Flannery E et al (2011) Composition, variability, and temporal stability of the intestinal microbiota of the elderly. Proc Natl Acad Sci USA 108(Suppl 1):4586–4591. https://doi.org/10.1073/pnas.1000097107
Musso G, Gambino R, Cassader M (2010) Obesity, diabetes, and gut microbiota: the hygiene hypothesis expanded? Diabetes Care 33(10):2277–2284. https://doi.org/10.2337/dc10-0556
Qin J, Li Y, Cai Z, Li S, Zhu J, Zhang F et al (2012) A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature 490(7418):55–60. https://doi.org/10.1038/nature11450
Jiang C, Li G, Huang P, Liu Z, Zhao B (2017) The gut microbiota and Alzheimer’s disease. J Alzheimers Dis 58(1):1–15. https://doi.org/10.3233/JAD-161141
Alam MS, Gangiredla J, Hasan NA, Barnaba T, Tartera C (2021) Aging-induced dysbiosis of gut microbiota as a risk factor for increased Listeria monocytogenes infection. Front Immunol 12:672353. https://doi.org/10.3389/fimmu.2021.672353
Wiertsema SP, van Bergenhenegouwen J, Garssen J, Knippels LMJ (2021) The interplay between the gut microbiome and the immune system in the context of infectious diseases throughout life and the role of nutrition in optimizing treatment strategies. Nutrients 13(3). https://doi.org/10.3390/nu13030886
Fuentes S, den Hartog G, Nanlohy NM, Wijnands L, Ferreira JA, Nicolaie MA et al (2021) Associations of faecal microbiota with influenza-like illness in participants aged 60 years or older: an observational study. The Lancet Healthy Longevity 2(1):e13–e23. https://doi.org/10.1016/S2666-7568(20)30034-9
Biragyn A, Ferrucci L (2018) Gut dysbiosis: a potential link between increased cancer risk in ageing and inflammaging. Lancet Oncol 19(6):e295–e304. https://doi.org/10.1016/S1470-2045(18)30095-0
Claesson MJ, Jeffery IB, Conde S, Power SE, O’Connor EM, Cusack S et al (2012) Gut microbiota composition correlates with diet and health in the elderly. Nature 488(7410):178–184. https://doi.org/10.1038/nature11319
Wilmanski T, Diener C, Rappaport N, Patwardhan S, Wiedrick J, Lapidus J et al (2021) Gut microbiome pattern reflects healthy ageing and predicts survival in humans. Nat Metab 3(2):274–286. https://doi.org/10.1038/s42255-021-00348-0
Salosensaari A, Laitinen V, Havulinna AS, Meric G, Cheng S, Perola M et al (2021) Taxonomic signatures of cause-specific mortality risk in human gut microbiome. Nat Commun 12(1):2671. https://doi.org/10.1038/s41467-021-22962-y
Popkes M, Valenzano DR (2020) Microbiota-host interactions shape ageing dynamics. Philos Trans R Soc Lond B Biol Sci 375(1808):20190596. https://doi.org/10.1098/rstb.2019.0596
Manfredo Vieira S, Hiltensperger M, Kumar V, Zegarra-Ruiz D, Dehner C, Khan N et al (2018) Translocation of a gut pathobiont drives autoimmunity in mice and humans. Science 359(6380):1156–1161. https://doi.org/10.1126/science.aar7201
Ansaldo E, Slayden LC, Ching KL, Koch MA, Wolf NK, Plichta DR et al (2019) Akkermansia muciniphila induces intestinal adaptive immune responses during homeostasis. Science 364(6446):1179–1184. https://doi.org/10.1126/science.aaw7479
Bodogai M, O'Connell J, Kim K, Kim Y, Moritoh K, Chen C et al (2018) Commensal bacteria contribute to insulin resistance in aging by activating innate B1a cells. Sci Transl Med 10(467). https://doi.org/10.1126/scitranslmed.aat4271
Shen X, Miao J, Wan Q, Wang S, Li M, Pu F et al (2018) Possible correlation between gut microbiota and immunity among healthy middle-aged and elderly people in southwest China. Gut Pathog 10:4. https://doi.org/10.1186/s13099-018-0231-3
Rhoades N, Mendoza N, Jankeel A, Sureshchandra S, Alvarez AD, Doratt B et al (2019) Altered immunity and microbial dysbiosis in aged individuals with long-term controlled HIV infection. Front Immunol 10:463. https://doi.org/10.3389/fimmu.2019.00463
Koeth RA, Wang Z, Levison BS, Buffa JA, Org E, Sheehy BT et al (2013) Intestinal microbiota metabolism of L-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat Med 19(5):576–585. https://doi.org/10.1038/nm.3145
Cianci R, Franza L, Massaro MG, Borriello R, De Vito F, Gambassi G (2020) The interplay between immunosenescence and microbiota in the efficacy of vaccines. Vaccines (Basel) 8(4). https://doi.org/10.3390/vaccines8040636
Akatsu H, Iwabuchi N, Xiao JZ, Matsuyama Z, Kurihara R, Okuda K et al (2013) Clinical effects of probiotic Bifidobacterium longum BB536 on immune function and intestinal microbiota in elderly patients receiving enteral tube feeding. JPEN J Parenter Enteral Nutr 37(5):631–640. https://doi.org/10.1177/0148607112467819
Boge T, Remigy M, Vaudaine S, Tanguy J, Bourdet-Sicard R, van der Werf S (2009) A probiotic fermented dairy drink improves antibody response to influenza vaccination in the elderly in two randomised controlled trials. Vaccine 27(41):5677–5684. https://doi.org/10.1016/j.vaccine.2009.06.094
Bosch M, Mendez M, Perez M, Farran A, Fuentes MC, Cune J (2012) Lactobacillus plantarum CECT7315 and CECT7316 stimulate immunoglobulin production after influenza vaccination in elderly. Nutr Hosp 27(2):504–509. https://doi.org/10.1590/S0212-16112012000200023
Langkamp-Henken B, Bender BS, Gardner EM, Herrlinger-Garcia KA, Kelley MJ, Murasko DM et al (2004) Nutritional formula enhanced immune function and reduced days of symptoms of upper respiratory tract infection in seniors. J Am Geriatr Soc 52(1):3–12. https://doi.org/10.1111/j.1532-5415.2004.52003.x
Bunout D, Hirsch S, Pia de la Maza M, Munoz C, Haschke F, Steenhout P et al (2002) Effects of prebiotics on the immune response to vaccination in the elderly. JPEN J Parenter Enteral Nutr 26(6):372–376. https://doi.org/10.1177/0148607102026006372
Maruyama M, Abe R, Shimono T, Iwabuchi N, Abe F, Xiao JZ (2016) The effects of non-viable Lactobacillus on immune function in the elderly: a randomised, double-blind, placebo-controlled study. Int J Food Sci Nutr 67(1):67–73. https://doi.org/10.3109/09637486.2015.1126564
Clarke TB, Davis KM, Lysenko ES, Zhou AY, Yu Y, Weiser JN (2010) Recognition of peptidoglycan from the microbiota by Nod1 enhances systemic innate immunity. Nat Med 16(2):228–231. https://doi.org/10.1038/nm.2087
Khosravi A, Yanez A, Price JG, Chow A, Merad M, Goodridge HS et al (2014) Gut microbiota promote hematopoiesis to control bacterial infection. Cell Host Microbe 15(3):374–381. https://doi.org/10.1016/j.chom.2014.02.006
Negi S, Das DK, Pahari S, Nadeem S, Agrewala JN (2019) Potential role of gut microbiota in induction and regulation of innate immune memory. Front Immunol 10:2441. https://doi.org/10.3389/fimmu.2019.02441
Levy M, Thaiss CA, Elinav E (2016) Metabolites: messengers between the microbiota and the immune system. Genes Dev 30(14):1589–1597. https://doi.org/10.1101/gad.284091.116
Schutless J, Pandey S, Capitani M, Rue-Albrecht KC, Arnold I, Franchini F et al (2019) The short chain fatty acid butyrate imprints an antimicrobial program in macrophages. Immunity 50(2):432–445. https://doi.org/10.1016/j.immuni.2018.12.018
Ospelt C, Reedquist KA, Gay S, Tak PP (2011) Inflammatory memories: is epigenetics the missing link to persistent stromal cell activation in rheumatoid arthritis? Autoimmun Rev 10(9):519–524. https://doi.org/10.1016/j.autrev.2011.04.001
Naik S, Larsen SB, Gomez NC, Alaverdyan K, Sendoel A, Yuan S et al (2017) Inflammatory memory sensitizes skin epithelial stem cells to tissue damage. Nature 550(7677):475–480. https://doi.org/10.1038/nature24271
Hamada A, Torre C, Drancourt M, Ghigo E (2018) Trained immunity carried by non-immune cells. Front Microbiol 9:3225. https://doi.org/10.3389/fmicb.2018.03225
Owens BM (2015) Inflammation, innate immunity, and the intestinal stromal cell niche: Opportunities and Challenges. Front Immunol 6:319. https://doi.org/10.3389/fimmu.2015.00319
Mattson MPMagnus T. (2006) Ageing and neuronal vulnerability. Nat Rev Neurosci 7(4):278–294. https://doi.org/10.1038/nrn1886
Ziv Y, Ron N, Butovsky O, Landa G, Sudai E, Greenberg N et al (2006) Immune cells contribute to the maintenance of neurogenesis and spatial learning abilities in adulthood. Nat Neurosci 9(2):268–275. https://doi.org/10.1038/nn1629
Ginhoux F, Greter M, Leboeuf M, Nandi S, See P, Gokhan S et al (2010) Fate mapping analysis reveals that adult microglia derive from primitive macrophages. Science 330(6005):841–845. https://doi.org/10.1126/science.1194637
Prinz M, Jung S, Priller J (2019) Microglia biology: one century of evolving concepts. Cell 179(2):292–311. https://doi.org/10.1016/j.cell.2019.08.053
Olah M, Patrick E, Villani AC, Xu J, White CC, Ryan KJ et al (2018) A transcriptomic atlas of aged human microglia. Nat Commun 9(1):539. https://doi.org/10.1038/s41467-018-02926-5
Deczkowska A, Schwartz M (2018) Targeting neuro-immune communication in neurodegeneration: Challenges and opportunities. J Exp Med 215(11):2702–2704. https://doi.org/10.1084/jem.20181737
Kunis G, Baruch K, Rosenzweig N, Kertser A, Miller O, Berkutzki T et al (2013) IFN-gamma-dependent activation of the brain’s choroid plexus for CNS immune surveillance and repair. Brain 136(Pt 11):3427–3440. https://doi.org/10.1093/brain/awt259
Yoles E, Hauben E, Palgi O, Agranov E, Gothilf A, Cohen A et al (2001) Protective autoimmunity is a physiological response to CNS trauma. J Neurosci 21(11):3740–3748
Ron-Harel N, Schwartz M (2009) Immune senescence and brain aging: can rejuvenation of immunity reverse memory loss? Trends Neurosci 32(7):367–375. https://doi.org/10.1016/j.tins.2009.03.003
Baruch K, Schwartz M (2013) CNS-specific T cells shape brain function via the choroid plexus. Brain Behav Immun 34:11–16. https://doi.org/10.1016/j.bbi.2013.04.002
Garg SK, Banerjee R, Kipnis J (2008) Neuroprotective immunity: T cell-derived glutamate endows astrocytes with a neuroprotective phenotype. J Immunol 180(6):3866–3873. https://doi.org/10.4049/jimmunol.180.6.3866
Butovsky O, Hauben E, Schwartz M (2001) Morphological aspects of spinal cord autoimmune neuroprotection: colocalization of T cells with B7–2 (CD86) and prevention of cyst formation. FASEB J 15(6):1065–1067. https://doi.org/10.1096/fj.00-0550fje
Baron R, Nemirovsky A, Harpaz I, Cohen H, Owens T, Monsonego A (2008) IFN-gamma enhances neurogenesis in wild-type mice and in a mouse model of Alzheimer’s disease. FASEB J 22(8):2843–2852. https://doi.org/10.1096/fj.08-105866
Ron-Harel N, Segev Y, Lewitus GM, Cardon M, Ziv Y, Netanely D et al (2008) Age-dependent spatial memory loss can be partially restored by immune activation. Rejuvenation Res 11(5):903–913. https://doi.org/10.1089/rej.2008.0755
Banerjee R, Mosley RL, Reynolds AD, Dhar A, Jackson-Lewis V, Gordon PH et al (2008) Adaptive immune neuroprotection in G93A-SOD1 amyotrophic lateral sclerosis mice. PLoS ONE 3(7):e2740. https://doi.org/10.1371/journal.pone.0002740
Beers DR, Henkel JS, Zhao W, Wang J, Appel SH (2008) CD4+ T cells support glial neuroprotection, slow disease progression, and modify glial morphology in an animal model of inherited ALS. Proc Natl Acad Sci USA 105(40):15558–15563. https://doi.org/10.1073/pnas.0807419105
Chiu IM, Chen A, Zheng Y, Kosaras B, Tsiftsoglou SA, Vartanian TK et al (2008) T lymphocytes potentiate endogenous neuroprotective inflammation in a mouse model of ALS. Proc Natl Acad Sci USA 105(46):17913–17918. https://doi.org/10.1073/pnas.0804610105
Brochard V, Combadiere B, Prigent A, Laouar Y, Perrin A, Beray-Berthat V et al (2009) Infiltration of CD4+ lymphocytes into the brain contributes to neurodegeneration in a mouse model of Parkinson disease. J Clin Invest 119(1):182–192. https://doi.org/10.1172/JCI36470
Li Y, Luikart BW, Birnbaum S, Chen J, Kwon CH, Kernie SG et al (2008) TrkB regulates hippocampal neurogenesis and governs sensitivity to antidepressive treatment. Neuron 59(3):399–412. https://doi.org/10.1016/j.neuron.2008.06.023
Heldt SA, Stanek L, Chhatwal JP, Ressler KJ (2007) Hippocampus-specific deletion of BDNF in adult mice impairs spatial memory and extinction of aversive memories. Mol Psychiatry 12(7):656–670. https://doi.org/10.1038/sj.mp.4001957
Barnes P, Thomas KL (2008) Proteolysis of proBDNF is a key regulator in the formation of memory. PLoS ONE 3(9):e3248. https://doi.org/10.1371/journal.pone.0003248
Kozisek ME, Middlemas D, Bylund DB (2008) Brain-derived neurotrophic factor and its receptor tropomyosin-related kinase B in the mechanism of action of antidepressant therapies. Pharmacol Ther 117(1):30–51. https://doi.org/10.1016/j.pharmthera.2007.07.001
Lewitus GM, Wilf-Yarkoni A, Ziv Y, Shabat-Simon M, Gersner R, Zangen A et al (2009) Vaccination as a novel approach for treating depressive behavior. Biol Psychiatry 65(4):283–288. https://doi.org/10.1016/j.biopsych.2008.07.014
Lewitus GM, Cohen H, Schwartz M (2008) Reducing post-traumatic anxiety by immunization. Brain Behav Immun 22(7):1108–1114. https://doi.org/10.1016/j.bbi.2008.05.002
Hohlfeld R, Kerschensteiner M, Stadelmann C, Lassmann H, Wekerle H (2006) The neuroprotective effect of inflammation: implications for the therapy of multiple sclerosis. Neurol Sci 27(Suppl 1):S1-7. https://doi.org/10.1007/s10072-006-0537-7
Beers DR, Henkel JS, Zhao W, Wang J, Huang A, Wen S et al (2011) Endogenous regulatory T lymphocytes ameliorate amyotrophic lateral sclerosis in mice and correlate with disease progression in patients with amyotrophic lateral sclerosis. Brain 134(Pt 5):1293–1314. https://doi.org/10.1093/brain/awr074
Baek H, Ye M, Kang GH, Lee C, Lee G, Choi DB et al (2016) Neuroprotective effects of CD4+CD25+Foxp3+ regulatory T cells in a 3xTg-AD Alzheimer’s disease model. Oncotarget 7(43):69347–69357. https://doi.org/10.18632/oncotarget.12469
Jamshidian A, Shaygannejad V, Pourazar A, Zarkesh-Esfahani SH, Gharagozloo M (2013) Biased Treg/Th17 balance away from regulatory toward inflammatory phenotype in relapsed multiple sclerosis and its correlation with severity of symptoms. J Neuroimmunol 262(1–2):106–112. https://doi.org/10.1016/j.jneuroim.2013.06.007
Wendeln AC, Degenhardt K, Kaurani L, Gertig M, Ulas T, Jain G et al (2018) Innate immune memory in the brain shapes neurological disease hallmarks. Nature 556(7701):332–338. https://doi.org/10.1038/s41586-018-0023-4
Heng Y, Zhang X, Borggrewe M, van Weering HRJ, Brummer ML, Nijboer TW et al (2021) Systemic administration of beta-glucan induces immune training in microglia. J Neuroinflammation 18(1):57. https://doi.org/10.1186/s12974-021-02103-4
Caserta MT, Bannon Y, Fernandez F, Giunta B, Schoenberg MR, Tan J (2009) Normal brain aging clinical, immunological, neuropsychological, and neuroimaging features. Int Rev Neurobiol 84:1–19. https://doi.org/10.1016/S0074-7742(09)00401-2
Norden DM, Muccigrosso MM, Godbout JP (2015) Microglial priming and enhanced reactivity to secondary insult in aging, and traumatic CNS injury, and neurodegenerative disease. Neuropharmacology 96(Pt A):29–41. https://doi.org/10.1016/j.neuropharm.2014.10.028
Barrientos RM, Kitt MM, Watkins LR, Maier SF (2015) Neuroinflammation in the normal aging hippocampus. Neuroscience 309:84–99. https://doi.org/10.1016/j.neuroscience.2015.03.007
Ekdahl CT, Claasen JH, Bonde S, Kokaia Z, Lindvall O (2003) Inflammation is detrimental for neurogenesis in adult brain. Proc Natl Acad Sci USA 100(23):13632–13637. https://doi.org/10.1073/pnas.2234031100
Monje ML, Toda H, Palmer TD (2003) Inflammatory blockade restores adult hippocampal neurogenesis. Science 302(5651):1760–1765. https://doi.org/10.1126/science.1088417
Miranda M, Morici JF, Zanoni MB, Bekinschtein P (2019) Brain-derived neurotrophic factor: a key molecule for memory in the healthy and the pathological brain. Front Cell Neurosci 13:363. https://doi.org/10.3389/fncel.2019.00363
Ritzel RM, Crapser J, Patel AR, Verma R, Grenier JM, Chauhan A et al (2016) Age-associated resident memory CD8 T cells in the central nervous system are primed to potentiate inflammation after ischemic brain injury. J Immunol 196(8):3318–3330. https://doi.org/10.4049/jimmunol.1502021
Fritzsching B, Haas J, Konig F, Kunz P, Fritzsching E, Poschl J et al (2011) Intracerebral human regulatory T cells: analysis of CD4+ CD25+ FOXP3+ T cells in brain lesions and cerebrospinal fluid of multiple sclerosis patients. PLoS ONE 6(3):e17988. https://doi.org/10.1371/journal.pone.0017988
Puntener U, Booth SG, Perry VH, Teeling JL (2012) Long-term impact of systemic bacterial infection on the cerebral vasculature and microglia. J Neuroinflammation 9:146. https://doi.org/10.1186/1742-2094-9-146
Rubio-Ruiz ME, Peredo-Escarcega AE, Cano-Martinez A, Guarner-Lans V (2015) An evolutionary perspective of nutrition and inflammation as mechanisms of cardiovascular disease. Int J Evol Biol 2015:179791. https://doi.org/10.1155/2015/179791
Hwangbo DS, Lee HY, Abozaid LS, Min KJ (2020) Mechanisms of lifespan regulation by calorie restriction and intermittent fasting in model organisms. Nutrients 12(4). https://doi.org/10.3390/nu12041194
Colman RJ, Beasley TM, Kemnitz JW, Johnson SC, Weindruch R, Anderson RM (2014) Caloric restriction reduces age-related and all-cause mortality in rhesus monkeys. Nat Commun 5:3557. https://doi.org/10.1038/ncomms4557
Colman RJ, Anderson RM, Johnson SC, Kastman EK, Kosmatka KJ, Beasley TM et al (2009) Caloric restriction delays disease onset and mortality in rhesus monkeys. Science 325(5937):201–204. https://doi.org/10.1126/science.1173635
Mattison JA, Roth GS, Beasley TM, Tilmont EM, Handy AM, Herbert RL et al (2012) Impact of caloric restriction on health and survival in rhesus monkeys from the NIA study. Nature 489(7415):318–321. https://doi.org/10.1038/nature11432
Ravussin E, Redman LM, Rochon J, Das SK, Fontana L, Kraus WE et al (2015) A 2-year randomized controlled trial of human caloric restriction: feasibility and effects on predictors of health span and longevity. J Gerontol A Biol Sci Med Sci 70(9):1097–1104. https://doi.org/10.1093/gerona/glv057
Lee C, Longo V (2016) Dietary restriction with and without caloric restriction for healthy aging. F1000Res 5. https://doi.org/10.12688/f1000research.7136.1
Messaoudi I, Warner J, Fischer M, Park B, Hill B, Mattison J et al (2006) Delay of T cell senescence by caloric restriction in aged long-lived nonhuman primates. Proc Natl Acad Sci USA 103(51):19448–19453. https://doi.org/10.1073/pnas.0606661103
Gardner EM (2005) Caloric restriction decreases survival of aged mice in response to primary influenza infection. J Gerontol A Biol Sci Med Sci 60(6):688–694. https://doi.org/10.1093/gerona/60.6.688
Goldberg EL, Romero-Aleshire MJ, Renkema KR, Ventevogel MS, Chew WM, Uhrlaub JL et al (2015) Lifespan-extending caloric restriction or mTOR inhibition impair adaptive immunity of old mice by distinct mechanisms. Aging Cell 14(1):130–138. https://doi.org/10.1111/acel.12280
Palma C, La Rocca C, Gigantino V, Aquino G, Piccaro G, Di Silvestre D et al (2021) Caloric restriction promotes immunometabolic reprogramming leading to protection from tuberculosis. Cell Metab 33(2):300–18. https://doi.org/10.1016/j.cmet.2020.12.016
Nieman DC, Henson DA, Gusewitch G, Warren BJ, Dotson RC, Butterworth DE et al (1993) Physical activity and immune function in elderly women. Med Sci Sports Exerc 25(7):823–831. https://doi.org/10.1249/00005768-199307000-00011
Duggal NA, Pollock RD, Lazarus NR, Harridge S, Lord JM (2018) Major features of immunesenescence, including reduced thymic output, are ameliorated by high levels of physical activity in adulthood. Aging Cell 17(2). https://doi.org/10.1111/acel.12750
Mejias-Pena Y, Estebanez B, Rodriguez-Miguelez P, Fernandez-Gonzalo R, Almar M, de Paz JA et al (2017) Impact of resistance training on the autophagy-inflammation-apoptosis crosstalk in elderly subjects. Aging (Albany NY) 9(2):408–418. https://doi.org/10.18632/aging.101167
Moreira OC, Estebanez B, Martinez-Florez S, de Paz JA, Cuevas MJ, Gonzalez-Gallego J (2017) Mitochondrial function and mitophagy in the elderly: effects of exercise. Oxid Med Cell Longev 2017:2012798. https://doi.org/10.1155/2017/2012798
Kulkarni AS, Gubbi S, Barzilai N (2020) Benefits of metformin in attenuating the hallmarks of aging. Cell Metab 32(1):15–30. https://doi.org/10.1016/j.cmet.2020.04.001
Mannick JB, Del Giudice G, Lattanzi M, Valiante NM, Praestgaard J, Huang B et al (2014) mTOR inhibition improves immune function in the elderly. Sci Transl Med 6(268):268ra179. https://doi.org/10.1126/scitranslmed.3009892
Mannick JB, Morris M, Hockey HP, Roma G, Beibel M, Kulmatycki K et al (2018) TORC1 inhibition enhances immune function and reduces infections in the elderly. Sci Transl Med 10(449). https://doi.org/10.1126/scitranslmed.aaq1564
Wang Q, Yan C, Xin M, Han L, Zhang Y, Sun M (2017) Sirtuin 1 (Sirt1) Overexpression in BaF3 cells contributes to cell proliferation promotion, apoptosis resistance and pro-inflammatory cytokine production. Med Sci Monit 23:1477–1482. https://doi.org/10.12659/msm.900754
Houtkooper RH, Pirinen E, Auwerx J (2012) Sirtuins as regulators of metabolism and healthspan. Nat Rev Mol Cell Biol 13(4):225–238. https://doi.org/10.1038/nrm3293
Mourits VP, Helder LS, Matzaraki V, Koeken V, Groh L, de Bree LCJ et al (2021) The role of sirtuin 1 on the induction of trained immunity. Cell Immunol 366:104393. https://doi.org/10.1016/j.cellimm.2021.104393
Pearson KJ, Baur JA, Lewis KN, Peshkin L, Price NL, Labinskyy N et al (2008) Resveratrol delays age-related deterioration and mimics transcriptional aspects of dietary restriction without extending life span. Cell Metab 8(2):157–168. https://doi.org/10.1016/j.cmet.2008.06.011
Minor RK, Baur JA, Gomes AP, Ward TM, Csiszar A, Mercken EM et al (2011) SRT1720 improves survival and healthspan of obese mice. Sci Rep 1:70. https://doi.org/10.1038/srep00070
Flachsbart F, Croucher PJ, Nikolaus S, Hampe J, Cordes C, Schreiber S et al (2006) Sirtuin 1 (SIRT1) sequence variation is not associated with exceptional human longevity. Exp Gerontol 41(1):98–102. https://doi.org/10.1016/j.exger.2005.09.008
Howitz KT, Bitterman KJ, Cohen HY, Lamming DW, Lavu S, Wood JG et al (2003) Small molecule activators of sirtuins extend Saccharomyces cerevisiae lifespan. Nature 425(6954):191–196. https://doi.org/10.1038/nature01960
McCubrey JA, Lertpiriyapong K, Steelman LS, Abrams SL, Yang LV, Murata RM et al (2017) Effects of resveratrol, curcumin, berberine and other nutraceuticals on aging, cancer development, cancer stem cells and microRNAs. Aging (Albany NY) 9(6):1477–1536. https://doi.org/10.18632/aging.101250
Baur JA, Sinclair DA (2006) Therapeutic potential of resveratrol: the in vivo evidence. Nat Rev Drug Discov 5(6):493–506. https://doi.org/10.1038/nrd2060
Docherty JJ, Smith JS, Fu MM, Stoner T, Booth T (2004) Effect of topically applied resveratrol on cutaneous herpes simplex virus infections in hairless mice. Antiviral Res 61(1):19–26. https://doi.org/10.1016/j.antiviral.2003.07.001
Docherty JJ, Fu MM, Hah JM, Sweet TJ, Faith SA, Booth T (2005) Effect of resveratrol on herpes simplex virus vaginal infection in the mouse. Antiviral Res 67(3):155–162. https://doi.org/10.1016/j.antiviral.2005.06.008
Strong R, Miller RA, Astle CM, Baur JA, de Cabo R, Fernandez E et al (2013) Evaluation of resveratrol, green tea extract, curcumin, oxaloacetic acid, and medium-chain triglyceride oil on life span of genetically heterogeneous mice. J Gerontol A Biol Sci Med Sci 68(1):6–16. https://doi.org/10.1093/gerona/gls070
Baur JA, Pearson KJ, Price NL, Jamieson HA, Lerin C, Kalra A et al (2006) Resveratrol improves health and survival of mice on a high-calorie diet. Nature 444(7117):337–342. https://doi.org/10.1038/nature05354
Jimenez-Gomez Y, Mattison JA, Pearson KJ, Martin-Montalvo A, Palacios HH, Sossong AM et al (2013) Resveratrol improves adipose insulin signaling and reduces the inflammatory response in adipose tissue of rhesus monkeys on high-fat, high-sugar diet. Cell Metab 18(4):533–545. https://doi.org/10.1016/j.cmet.2013.09.004
Timmers S, Konings E, Bilet L, Houtkooper RH, van de Weijer T, Goossens GH et al (2011) Calorie restriction-like effects of 30 days of resveratrol supplementation on energy metabolism and metabolic profile in obese humans. Cell Metab 14(5):612–622. https://doi.org/10.1016/j.cmet.2011.10.002
Poulsen MM, Vestergaard PF, Clasen BF, Radko Y, Christensen LP, Stodkilde-Jorgensen H et al (2013) High-dose resveratrol supplementation in obese men: an investigator-initiated, randomized, placebo-controlled clinical trial of substrate metabolism, insulin sensitivity, and body composition. Diabetes 62(4):1186–1195. https://doi.org/10.2337/db12-0975
Li Y, Daniel M, Tollefsbol TO (2011) Epigenetic regulation of caloric restriction in aging. BMC Med 9:98. https://doi.org/10.1186/1741-7015-9-98
Fahy GM, Brooke RT, Watson JP, Good Z, Vasanawala SS, Maecker H et al (2019) Reversal of epigenetic aging and immunosenescent trends in humans. Aging Cell 18(6):e13028. https://doi.org/10.1111/acel.13028
Maegawa S, Lu Y, Tahara T, Lee JT, Madzo J, Liang S et al (2017) Caloric restriction delays age-related methylation drift. Nat Commun 8(1):539. https://doi.org/10.1038/s41467-017-00607-3
Wang T, Tsui B, Kreisberg JF, Robertson NA, Gross AM, Yu MK et al (2017) Epigenetic aging signatures in mice livers are slowed by dwarfism, calorie restriction and rapamycin treatment. Genome Biol 18(1):57. https://doi.org/10.1186/s13059-017-1186-2
Hadad N, Unnikrishnan A, Jackson JA, Masser DR, Otalora L, Stanford DR et al (2018) Caloric restriction mitigates age-associated hippocampal differential CG and non-CG methylation. Neurobiol Aging 67:53–66. https://doi.org/10.1016/j.neurobiolaging.2018.03.009
Bridgeman SC, Ellison GC, Melton PE, Newsholme P, Mamotte CDS (2018) Epigenetic effects of metformin: From molecular mechanisms to clinical implications. Diabetes Obes Metab 20(7):1553–1562. https://doi.org/10.1111/dom.13262
Lee H, Lee Y, Kim J, An J, Lee S, Kong H et al (2018) Modulation of the gut microbiota by metformin improves metabolic profiles in aged obese mice. Gut Microbes 9(2):155–165. https://doi.org/10.1080/19490976.2017.1405209
de la Cuesta-Zuluaga J, Mueller NT, Corrales-Agudelo V, Velasquez-Mejia EP, Carmona JA, Abad JM et al (2017) Metformin is associated with higher relative abundance of mucin-degrading Akkermansia muciniphila and several short-chain fatty acid-producing microbiota in the gut. Diabetes Care 40(1):54–62. https://doi.org/10.2337/dc16-1324
Ouyang J, Lin J, Isnard S, Fombuena B, Peng X, Marette A et al (2020) The bacterium Akkermansia muciniphila: a sentinel for gut permeability and its relevance to HIV-related inflammation. Front Immunol 11:645. https://doi.org/10.3389/fimmu.2020.00645
Ahmadi S, Razazan A, Nagpal R, Jain S, Wang B, Mishra SP et al (2020) Metformin reduces aging-related leaky gut and improves cognitive function by beneficially modulating gut microbiome/goblet cell/mucin axis. J Gerontol A Biol Sci Med Sci 75(7):e9–e21. https://doi.org/10.1093/gerona/glaa056
Ale EC, Binetti AG (2021) Role of probiotics, prebiotics, and synbiotics in the elderly: insights into their applications. Front Microbiol 12:631254. https://doi.org/10.3389/fmicb.2021.631254
Alfa MJ, Strang D, Tappia PS, Graham M, Van Domselaar G, Forbes JD et al (2018) A randomized trial to determine the impact of a digestion resistant starch composition on the gut microbiome in older and mid-age adults. Clin Nutr 37(3):797–807. https://doi.org/10.1016/j.clnu.2017.03.025
Roy Sarkar S, Mitra Mazumder P, Banerjee S (2020) Probiotics protect against gut dysbiosis associated decline in learning and memory. J Neuroimmunol 348:577390. https://doi.org/10.1016/j.jneuroim.2020.577390
Finamore A, Roselli M, Donini L, Brasili DE, Rami R, Carnevali P et al (2019) Supplementation with Bifidobacterium longum Bar33 and Lactobacillus helveticus Bar13 mixture improves immunity in elderly humans (over 75 years) and aged mice. Nutrition 63–64:184–192. https://doi.org/10.1016/j.nut.2019.02.005
Yamamoto Y, Saruta J, Takahashi T, To M, Shimizu T, Hayashi T et al (2019) Effect of ingesting yogurt fermented with Lactobacillus delbrueckii ssp. bulgaricus OLL1073R-1 on influenza virus-bound salivary IgA in elderly residents of nursing homes: a randomized controlled trial. Acta Odontol Scand 77(7):517–24. https://doi.org/10.1080/00016357.2019.1609697
Rampelli S, Candela M, Severgnini M, Biagi E, Turroni S, Roselli M et al (2013) A probiotics-containing biscuit modulates the intestinal microbiota in the elderly. J Nutr Health Aging 17(2):166–172. https://doi.org/10.1007/s12603-012-0372-x
Qu H, Zhang Y, Chai H, Gao ZY, Shi DZ (2019) Effects of microbiota-driven therapy on inflammatory responses in elderly individuals: a systematic review and meta-analysis. PLoS ONE 14(2):e0211233. https://doi.org/10.1371/journal.pone.0211233
Cicero AFG, Fogacci F, Bove M, Giovannini M, Borghi C (2021) Impact of a short-term synbiotic supplementation on metabolic syndrome and systemic inflammation in elderly patients: a randomized placebo-controlled clinical trial. Eur J Nutr 60(2):655–663. https://doi.org/10.1007/s00394-020-02271-8
Macfarlane S, Cleary S, Bahrami B, Reynolds N, Macfarlane GT (2013) Synbiotic consumption changes the metabolism and composition of the gut microbiota in older people and modifies inflammatory processes: a randomised, double-blind, placebo-controlled crossover study. Aliment Pharmacol Ther 38(7):804–816. https://doi.org/10.1111/apt.12453
Zeng T, Cui H, Tang D, Garside GB, Wang Y, Wu J et al (2019) Short-term dietary restriction in old mice rejuvenates the aging-induced structural imbalance of gut microbiota. Biogerontology 20(6):837–848. https://doi.org/10.1007/s10522-019-09830-5
Zhang C, Li S, Yang L, Huang P, Li W, Wang S et al (2013) Structural modulation of gut microbiota in life-long calorie-restricted mice. Nat Commun 4:2163. https://doi.org/10.1038/ncomms3163
von Schwartzenberg RJ, Bisanz JE, Lyalina S, Spanogiannopoulos P, Ang QY, Cai J et al (2021) Caloric restriction disrupts the microbiota and colonization resistance. Nature. https://doi.org/10.1038/s41586-021-03663-4
Ahlskog JE, Geda YE, Graff-Radford NR, Petersen RC (2011) Physical exercise as a preventive or disease-modifying treatment of dementia and brain aging. Mayo Clin Proc 86(9):876–884. https://doi.org/10.4065/mcp.2011.0252
Ferencz B, Laukka EJ, Welmer AK, Kalpouzos G, Angleman S, Keller L et al (2014) The benefits of staying active in old age: physical activity counteracts the negative influence of PICALM, BIN1, and CLU risk alleles on episodic memory functioning. Psychol Aging 29(2):440–449. https://doi.org/10.1037/a0035465
Wirth M, Haase CM, Villeneuve S, Vogel J, Jagust WJ (2014) Neuroprotective pathways: lifestyle activity, brain pathology, and cognition in cognitively normal older adults. Neurobiol Aging 35(8):1873–1882. https://doi.org/10.1016/j.neurobiolaging.2014.02.015
Tolppanen AM, Solomon A, Kulmala J, Kareholt I, Ngandu T, Rusanen M et al (2015) Leisure-time physical activity from mid- to late life, body mass index, and risk of dementia. Alzheimers Dement 11(4):434–43. https://doi.org/10.1016/j.jalz.2014.01.008
Hamer M, Chida Y (2009) Physical activity and risk of neurodegenerative disease: a systematic review of prospective evidence. Psychol Med 39(1):3–11. https://doi.org/10.1017/S0033291708003681
de Sousa CV, Sales MM, Rosa TS, Lewis JE, de Andrade RV, Simoes HG (2017) The antioxidant effect of exercise: a systematic review and meta-analysis. Sports Med 47(2):277–293. https://doi.org/10.1007/s40279-016-0566-1
Valenzuela PL, Castillo-Garcia A, Morales JS, de la Villa P, Hampel H, Emanuele E et al (2020) Exercise benefits on Alzheimer’s disease: state-of-the-science. Ageing Res Rev 62:101108. https://doi.org/10.1016/j.arr.2020.101108
Gleeson M, Bishop NC, Stensel DJ, Lindley MR, Mastana SS, Nimmo MA (2011) The anti-inflammatory effects of exercise: mechanisms and implications for the prevention and treatment of disease. Nat Rev Immunol 11(9):607–615. https://doi.org/10.1038/nri3041
Simpson RJ, Lowder TW, Spielmann G, Bigley AB, LaVoy EC, Kunz H (2012) Exercise and the aging immune system. Ageing Res Rev 11(3):404–420. https://doi.org/10.1016/j.arr.2012.03.003
Woods JA, Keylock KT, Lowder T, Vieira VJ, Zelkovich W, Dumich S et al (2009) Cardiovascular exercise training extends influenza vaccine seroprotection in sedentary older adults: the immune function intervention trial. J Am Geriatr Soc 57(12):2183–2191. https://doi.org/10.1111/j.1532-5415.2009.02563.x
Szuhany KL, Bugatti M, Otto MW (2015) A meta-analytic review of the effects of exercise on brain-derived neurotrophic factor. J Psychiatr Res 60:56–64. https://doi.org/10.1016/j.jpsychires.2014.10.003
Marosi K, Kim SW, Moehl K, Scheibye-Knudsen M, Cheng A, Cutler R et al (2016) 3-Hydroxybutyrate regulates energy metabolism and induces BDNF expression in cerebral cortical neurons. J Neurochem 139(5):769–781. https://doi.org/10.1111/jnc.13868
Sleiman SF, Henry J, Al-Haddad R, El Hayek L, Abou Haidar E, Stringer T, et al (2016) Exercise promotes the expression of brain derived neurotrophic factor (BDNF) through the action of the ketone body beta-hydroxybutyrate. Elife 5. https://doi.org/10.7554/eLife.15092
Yang H, Shan W, Zhu F, Wu J, Wang Q (2019) Ketone bodies in neurological diseases: focus on neuroprotection and underlying mechanisms. Front Neurol 10:585. https://doi.org/10.3389/fneur.2019.00585
Lee J, Seroogy KB, Mattson MP (2002) Dietary restriction enhances neurotrophin expression and neurogenesis in the hippocampus of adult mice. J Neurochem 80(3):539–547. https://doi.org/10.1046/j.0022-3042.2001.00747.x
Guo J, Bakshi V, Lin AL (2015) Early shifts of brain metabolism by caloric restriction preserve white matter integrity and long-term memory in aging mice. Front Aging Neurosci 7:213. https://doi.org/10.3389/fnagi.2015.00213
Bruce-Keller AJ, Umberger G, McFall R, Mattson MP (1999) Food restriction reduces brain damage and improves behavioral outcome following excitotoxic and metabolic insults. Ann Neurol 45(1):8–15
Hyun DH, Emerson SS, Jo DG, Mattson MP, de Cabo R (2006) Calorie restriction up-regulates the plasma membrane redox system in brain cells and suppresses oxidative stress during aging. Proc Natl Acad Sci USA 103(52):19908–19912. https://doi.org/10.1073/pnas.0608008103
Poon HF, Shepherd HM, Reed TT, Calabrese V, Stella AM, Pennisi G et al (2006) Proteomics analysis provides insight into caloric restriction mediated oxidation and expression of brain proteins associated with age-related impaired cellular processes: mitochondrial dysfunction, glutamate dysregulation and impaired protein synthesis. Neurobiol Aging 27(7):1020–1034. https://doi.org/10.1016/j.neurobiolaging.2005.05.014
Jahng JW, Kim JG, Kim HJ, Kim BT, Kang DW, Lee JH (2007) Chronic food restriction in young rats results in depression- and anxiety-like behaviors with decreased expression of serotonin reuptake transporter. Brain Res 1150:100–107. https://doi.org/10.1016/j.brainres.2007.02.080
Patel NV, Gordon MN, Connor KE, Good RA, Engelman RW, Mason J et al (2005) Caloric restriction attenuates Abeta-deposition in Alzheimer transgenic models. Neurobiol Aging 26(7):995–1000. https://doi.org/10.1016/j.neurobiolaging.2004.09.014
Wang J, Ho L, Qin W, Rocher AB, Seror I, Humala N et al (2005) Caloric restriction attenuates beta-amyloid neuropathology in a mouse model of Alzheimer’s disease. FASEB J 19(6):659–661. https://doi.org/10.1096/fj.04-3182fje
Qin W, Yang T, Ho L, Zhao Z, Wang J, Chen L et al (2006) Neuronal SIRT1 activation as a novel mechanism underlying the prevention of Alzheimer disease amyloid neuropathology by calorie restriction. J Biol Chem 281(31):21745–21754. https://doi.org/10.1074/jbc.M602909200
Pifferi F, Aujard F (2019) Caloric restriction, longevity and aging: recent contributions from human and non-human primate studies. Prog Neuropsychopharmacol Biol Psychiatry 95:109702. https://doi.org/10.1016/j.pnpbp.2019.109702
Martin CK, Anton SD, Han H, York-Crowe E, Redman LM, Ravussin E et al (2007) Examination of cognitive function during six months of calorie restriction: results of a randomized controlled trial. Rejuvenation Res 10(2):179–190. https://doi.org/10.1089/rej.2006.0502
Witte AV, Fobker M, Gellner R, Knecht S, Floel A (2009) Caloric restriction improves memory in elderly humans. Proc Natl Acad Sci USA 106(4):1255–1260. https://doi.org/10.1073/pnas.0808587106
Prehn K, Jumpertz von Schwartzenberg R, Mai K, Zeitz U, Witte AV, Hampel D et al (2017) Caloric restriction in older adults-differential effects of weight loss and reduced weight on brain structure and function. Cereb Cortex 27(3):1765–1778. https://doi.org/10.1093/cercor/bhw008
Gofrit ON, Klein BY, Cohen IR, Ben-Hur T, Greenblatt CL, Bercovier H (2019) Bacillus Calmette-Guerin (BCG) therapy lowers the incidence of Alzheimer’s disease in bladder cancer patients. PLoS ONE 14(11):e0224433. https://doi.org/10.1371/journal.pone.0224433
Klinger D, Hill BL, Barda N, Halperin E, Gofrit ON, Greenblatt CL et al (2021) Bladder cancer immunotherapy by BCG is associated with a significantly reduced risk of Alzheimer's disease and Parkinson's disease. Vaccines (Basel) 9(5). https://doi.org/10.3390/vaccines9050491
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bulut, O., Kilic, G. & Domínguez-Andrés, J. Immune Memory in Aging: a Wide Perspective Covering Microbiota, Brain, Metabolism, and Epigenetics. Clinic Rev Allerg Immunol 63, 499–529 (2022). https://doi.org/10.1007/s12016-021-08905-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12016-021-08905-x