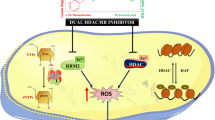
Abstract
Kaempferol illustrates an example of attempting to discover new treatments for neurodegeneration by investigating the potential efficacy of natural products. Despite the identification of several molecular targets for this bio-active compound, the precise underlying pathways are not well elucidated yet. Recently, it has been shown through pulldown assay that kaemferol directly interacts with p47phox, the organizer subunit of NADPH oxidase-2 (NOX2) complex. Hence, in this study, we used homology modelling, computational docking, mutation analysis, molecular dynamics simulations and free energy calculations to determine how kaempferol interacts with p47phox. Firstly, 3D structure of p47phox was generated using x-ray structures of its domains. Then, it was docked with kaempferol, and finally 100-ns molecular dynamics (MD) simulations were performed and the global properties like root-mean square deviation (RMSD) and root-mean square fluctuations (RMSF) were calculated. Literature survey and computational analysis of key interacting amino acid residues of p47phox provided insights into a possible binding site for kaempferol, approximately around Trp193 and Cys196 located within the N–terminal SH3 domain of p47phox. Moreover, free energy calculations indicated that in silico substitution of Trp193 and Cys196 with arginine and alanine, respectively, results in less favorable interaction corroborating their importance in binding with kaempferol. Taken together, these findings suggest that kaempferol directly attaches to N–SH3 domain p47 phox, with a subsequent diminution of p47phox protein–protein interaction and possibly attenuation of NOX2 complex assembly, which reduces reactive oxygen species (ROS) generation. These observations will be beneficial for researchers exploring neuroprotection and for the development of p47phox inhibitors.








Similar content being viewed by others
References
Szrok-Jurga, S., Turyn, J., Hebanowska, A., Swierczynski, J., Czumaj, A. & Sledzinski, T. et al. (2023). The role of Acyl-CoA β-oxidation in brain metabolism and neurodegenerative diseases. International Journal of Molecular Sciences, 24(18), 13977
S. Hernandes, M., & Britto, L. R. (2012). NADPH oxidase and neurodegeneration. Current neuropharmacology, 10(4), 321–327
Eid, S. A., Savelieff, M. G., Eid, A. A., & Feldman, E. L. (2022). Nox, Nox, are you there? The role of NADPH oxidases in the peripheral nervous system. Antioxidants & Redox Signaling, 37(7–9), 613–630
Fang, J., Sheng, R. & Qin, Z. H. (2021). NADPH oxidases in the central nervous system: Regional and cellular localization and the possible link to brain diseases. Antioxidants & Redox Signaling, 35(12), 951–973
Decourt, B., Lahiri, D. K. & Sabbagh, M. N. (2017). Targeting tumor necrosis factor alpha for Alzheimer’s disease. Current Alzheimer research, 14(4), 412–425
Altenhofer, S., Radermacher, K. A., Kleikers, P. W., Wingler, K., & Schmidt, H. H. (2015). Evolution of NADPH oxidase inhibitors: Selectivity and mechanisms for target engagement. Antioxidants & redox signaling, 23(5), 406–427
Rastogi, R., Geng, X., Li, F., & Ding, Y. (2016). NOX activation by subunit interaction and underlying mechanisms in disease. Frontiers in cellular neuroscience, 10, 301
Karimi, G., Houee Levin, C., Dagher, M. C., Baciou, L., & Bizouarn, T. (2014). Assembly of phagocyte NADPH oxidase: A concerted binding process? Biochimica et biophysica acta, 1840(11), 3277–3283
Meijles, D. N., Fan, L. M., Howlin, B. J., & Li, J. M. (2014). Molecular insights of p47phox phosphorylation dynamics in the regulation of NADPH oxidase activation and superoxide production. The Journal of Biological Chemistry, 289, 22759–22770
Raad, H., Mouawia, H., Hassan, H., El-Seblani, M., Arabi-Derkawi, R., & Boussetta, T., et al. (2020). The protein kinase A negatively regulates reactive oxygen species production by phosphorylating gp91phox/NOX2 in human neutrophils. Free Radical Biology and Medicine, 160, 19–27
Dang, P. M., Stensballe, A., Boussetta, T., Raad, H., Dewas, C., Kroviarski, Y., Hayem, G., Jensen, O. N., Gougerot-Pocidalo, M. A., & El-Benna, J. (2006). A specific p47phox -serine phosphorylated by convergent MAPKs mediates neutrophil NADPH oxidase priming at inflammatory sites. The Journal of Clinical Investigation, 116(7), 2033–2043
El-Benna, J., Dang, P. M., Gougerot-Pocidalo, M. A., Marie, J. C. & Braut-Boucher, F. (2009). p47phox, the phagocyte NADPH oxidase/NOX2 organizer: structure, phosphorylation and implication in diseases. Experimental & Molecular Medicine, 41(4), 217–225
Vermot, A., Petit-Härtlein, I., Smith, S. M. E., & Fieschi, F. (2021). NADPH oxidases (NOX): An overview from discovery, molecular mechanisms to physiology and pathology. Antioxidants, 10(6), 890
Cifuentes-Pagano, E., Saha, J., Csanyi, G., Ghouleh, I. A., Sahoo, S., Rodriguez, A., Wipf, P., Pagano, P. J., & Skoda, E. M. (2013). Bridged tetrahydroisoquinolines as selective NADPH oxidase 2 (Nox2) inhibitors. MedChemComm, 4, 1085–1092
Dang, D. K., Shin, E. J., Nam, Y., Ryoo, S., Jeong, J. H., Jang, C. G., Nabeshima, T., Hong, J. S., & Kim, H. C. (2016). Apocynin prevents mitochondrial burdens, microglial activation, and pro-apoptosis induced by a toxic dose of methamphetamine in the striatum of mice via inhibition of p47phox activation by ERK. Journal of Neuroinflammation, 13, 12
Mora-Pale, M., Kwon, S. J., Linhardt, R. J., & Dordick, J. S. (2012). Trimer hydroxylated quinone derived from apocynin targets cysteine residues of p47phox preventing the activation of human vascular NADPH oxidase. Free Radical Biology & Medicine, 52(5), 962–969
Macias Perez M. E., Hernandez Rodriguez M., Cabrera Perez L. C., Fragoso-Vazquez M. J., Correa-Basurto J., Padilla M., II, Mendez Luna D., Mera Jimenez E., Flores Sandoval C., Tamay Cach F., Rosales-Hernandez M. C. (2017) Aromatic regions govern the recognition of NADPH oxidase inhibitors as diapocynin and its analogues, Archiv der Pharmazie, 350(10), 1700041
Rezazadeh-Shojaee, F.-S., Ramazani, E., Kasaian, J., & Tayarani-Najaran, Z. (2022). Protective effects of 6-gingerol on 6-hydroxydopamine-induced apoptosis in PC12 cells through modulation of SAPK/JNK and survivin activation. Journal of Biochemical and Molecular Toxicology, 36(2), e22956
Gao, H. M., Zhou, H., & Hong, J. S. (2012). NADPH oxidases: Novel therapeutic targets for neurodegenerative diseases. Trends in Pharmacological Sciences, 33(6), 295–303
Hurkacz, M., Dobrek, L., & Wiela-Hojeńska, A. (2021). Antibiotics and the nervous system—which face of antibiotic therapy is real, Dr. Jekyll (Neurotoxicity) or Mr. Hyde (Neuroprotection)? Molecules, 26(24), 7456
Neha, & Parvez, S. (2023). Emerging therapeutics agents and recent advances in drug repurposing for Alzheimer’s disease. Ageing Research Reviews, 85, 101815
Garrido-Mesa, N., Zarzuelo, A., & Galvez, J. (2013). Minocycline: Far beyond an antibiotic. British Journal of Pharmacology, 169(2), 337–352
Garrido-Mesa, N., Zarzuelo, A., & Galvez, J. (2013). What is behind the non-antibiotic properties of minocycline? Pharmacological Research, 67(1), 18–30
Li, C., Yuan, K., & Schluesener, H. (2013). Impact of minocycline on neurodegenerative diseases in rodents: a meta-analysis. Reviews in the Neurosciences, 24(5), 553–562
Daulatzai, M. A. (2016). Pharmacotherpy and Alzheimer’s disease: The M-drugs (melatonin, minocycline, modafinil, and memantine) approach. Current Pharmaceutical Design, 22(16), 2411–2430
Karachitos, A., Garcia Del Pozo, J. S., de Groot, P. W., Kmita, H., & Jordan, J. (2013). Minocycline mediated mitochondrial cytoprotection: premises for therapy of cerebrovascular and neurodegenerative diseases. Current Drug Targets, 14(1), 47–55
Garcez, M. L., Mina, F., Bellettini-Santos, T., Carneiro, F. G., Luz, A. P., Schiavo, G. L., Andrighetti, M. S., Scheid, M. G., Bolfe, R. P., & Budni, J. (2017). Minocycline reduces inflammatory parameters in the brain structures and serum and reverses memory impairment caused by the administration of amyloid beta (1-42) in mice. Progress in neuro-psychopharmacology & biological psychiatry, 77, 23–31
Shultz, R. B., & Zhong, Y. (2017). Minocycline targets multiple secondary injury mechanisms in traumatic spinal cord injury. Neural Regeneration Research, 12(5), 702–713
Zhu, S., Stavrovskaya, I. G., Drozda, M., Kim, B. Y., Ona, V., Li, M., Sarang, S., Liu, A. S., Hartley, D. M., Wu, D. C., Gullans, S., Ferrante, R. J., Przedborski, S., Kristal, B. S., & Friedlander, R. M. (2002). Minocycline inhibits cytochrome c release and delays progression of amyotrophic lateral sclerosis in mice. Nature, 417(6884), 74–78
Chen, M., Ona, V. O., Li, M., Ferrante, R. J., Fink, K. B., Zhu, S., Bian, J., Guo, L., Farrell, L. A., Hersch, S. M., Hobbs, W., Vonsattel, J. P., Cha, J. H., & Friedlander, R. M. (2000). Minocycline inhibits caspase-1 and caspase-3 expression and delays mortality in a transgenic mouse model of Huntington disease. Nature Medicine, 6(7), 797–801
Xia, Z., & Friedlander, R. M. (2017). Minocycline in multiple sclerosis—compelling results but too early to tell. The New England Journal of Medicine, 376(22), 2191–2193
Sharifi, A. M., Eslami, H., Larijani, B., & Davoodi, J. (2009). Involvement of caspase-8, -9, and -3 in high glucose-induced apoptosis in PC12 cells. Neuroscience Letters, 459(2), 47–51.
Mansour, R. M., El Sayed, N. S., Ahmed, M. A. E., & El-Sahar, A. E. (2022). Addressing peroxisome proliferator-activated receptor-gamma in 3-nitropropionic acid-induced striatal neurotoxicity in rats. Molecular Neurobiology, 59(7), 4368–4383
Webb, B., & Sali, A. (2016). Comparative protein structure modeling using MODELLER. Current Protocols in Bioinformatics, 54, 5 6 1–5 6 37
Goodsell, D. S., Morris, G. M., & Olson, A. J. (1996). Automated docking of flexible ligands: Applications of AutoDock. Journal of Molecular Recognition, 9(1), 1–5
Van Der Spoel, D., Lindahl, E., Hess, B., Groenhof, G., Mark, A. E., & Berendsen, H. J. (2005). GROMACS: fast, flexible, and free. Journal of Computational Chemistry, 26(16), 1701–1718
Haji-Allahverdipoor, K., Jalali Javaran, M., Rashidi Monfared, S., Khadem-Erfan, M. B., Nikkhoo, B., & Bahrami Rad, Z., et al. (2023). Insights into the effects of amino acid substitutions on the stability of reteplase structure: A molecular dynamics simulation study. Iran J Biotechnol, 21(1), e3175
Pettersen, E. F., Goddard, T. D., Huang, C. C., Couch, G. S., Greenblatt, D. M., Meng, E. C., & Ferrin, T. E. (2004). UCSF Chimera–a visualization system for exploratory research and analysis. Journal of Computational Chemistry, 25(13), 1605–1612
Kumari, R., Kumar, R., C. Open Source Drug Discovery, & Lynn, A. (2014). g_mmpbsa–a GROMACS tool for high-throughput MM-PBSA calculations. Journal of Chemical Information and Modeling, 54(7), 1951–1962
Sato, Y., Fujimoto, S., Mukai, E., Sato, H., Tahara, Y., Ogura, K., Yamano, G., Ogura, M., Nagashima, K., & Inagaki, N. (2014). Palmitate induces reactive oxygen species production and beta-cell dysfunction by activating nicotinamide adenine dinucleotide phosphate oxidase through Src signaling. Journal of Diabetes Investigation, 5(1), 19–26
Amaral, M., Kokh, D. B., Bomke, J., Wegener, A., Buchstaller, H. P., Eggenweiler, H. M., Matias, P., Sirrenberg, C., Wade, R. C., & Frech, M. (2017). Protein conformational flexibility modulates kinetics and thermodynamics of drug binding. Nature Communications, 8(1), 2276
Giuliani, A. (2017). The application of principal component analysis to drug discovery and biomedical data. Drug Discovery Today, 22(7), 1069–1076
Wang L., Ran Zeng R., Qian Pang X., Gu Q., Tan W. (2015) The mechanisms of flavonoids inhibiting conformational transition of amyloid-β42 monomer: A comparative molecular dynamics simulation study. RSC Advances, 5(81), 66391–66402
El-Benna, J., Dang, P. M., & Gougerot-Pocidalo, M. A. (2008). Priming of the neutrophil NADPH oxidase activation: role of p47phox phosphorylation and NOX2 mobilization to the plasma membrane. Seminars in Immunopathology, 30(3), 279–289
Yamagishi, S., Yamada, M., Ishikawa, Y., Matsumoto, T., Ikeuchi, T., & Hatanaka, H. (2001). p38 mitogen-activated protein kinase regulates low potassium-induced c-Jun phosphorylation and apoptosis in cultured cerebellar granule neurons. The Journal of Biological Chemistry, 276(7), 5129–5133
Zhang, L., Xing, D., Liu, L., Gao, X., & Chen, M. (2007). TNFalpha induces apoptosis through JNK/Bax-dependent pathway in differentiated, but not naive PC12 cells. Cell Cycle, 6(12), 1479–1486
Brenner, D., Blaser, H., & Mak, T. W. (2015). Regulation of tumour necrosis factor signalling: Live or let die. Nature Reviews. Immunology, 15(6), 362–374
Cuadrado, A., & Nebreda, A. R. (2010). Mechanisms and functions of p38 MAPK signalling. The Biochemical Journal, 429(3), 403–417
Schrantz, N., Bourgeade, M. F., Mouhamad, S., Leca, G., Sharma, S., & Vazquez, A. (2001). p38-mediated regulation of an Fas-associated death domain protein-independent pathway leading to caspase-8 activation during TGFbeta-induced apoptosis in human Burkitt lymphoma B cells BL41. Molecular Biology of the Cell, 12(10), 3139–3151
Fernandez-Gomez, F. J., Gomez-Lazaro, M., Pastor, D., Calvo, S., Aguirre, N., Galindo, M. F., & Jordan, J. (2005). Minocycline fails to protect cerebellar granular cell cultures against malonate-induced cell death. Neurobiology of Disease, 20(2), 384–391
Yong, V. W., Wells, J., Giuliani, F., Casha, S., Power, C., & Metz, L. M. (2004). The promise of minocycline in neurology. The Lancet. Neurology, 3(12), 744–751
Diguet, E., Gross, C. E., Tison, F., & Bezard, E. (2004). Rise and fall of minocycline in neuroprotection: need to promote publication of negative results. Experimental Neurology, 189(1), 1–4
Cheng, S., Hou, J., Zhang, C., Xu, C., Wang, L., Zou, X., Yu, H., Shi, Y., Yin, Z., & Chen, G. (2015). Minocycline reduces neuroinflammation but does not ameliorate neuron loss in a mouse model of neurodegeneration. Scientific Reports, 5, 10535
Scott, G., Zetterberg, H., Jolly, A., Cole, J. H., De Simoni, S., Jenkins, P. O., Feeney, C., Owen, D. R., Lingford-Hughes, A., Howes, O., Patel, M. C., Goldstone, A. P., Gunn, R. N., Blennow, K., Matthews, P. M., & Sharp, D. J. (2018). Minocycline reduces chronic microglial activation after brain trauma but increases neurodegeneration. Brain : a Journal Of Neurology, 141(2), 459–471
Strahan, J. A., Walker, 2nd, W. H., Montgomery, T. R., & Forger, N. G. (2017). Minocycline causes widespread cell death and increases microglial labeling in the neonatal mouse brain. Developmental Neurobiology, 77(6), 753–766
Diguet, E., Fernagut, P. O., Wei, X., Du, Y., Rouland, R., Gross, C., Bezard, E., & Tison, F. (2004). Deleterious effects of minocycline in animal models of Parkinson’s disease and Huntington’s disease. The European Journal of Neuroscience, 19(12), 3266–3276
D.I. Huntington Study Group. (2010). A futility study of minocycline in Huntington’s disease, Movement disorders : official. Journal of the Movement Disorder Society, 25(13), 2219–2224
Arnoux, I., Hoshiko, M., Sanz Diez, A., & Audinat, E. (2014). Paradoxical effects of minocycline in the developing mouse somatosensory cortex. Glia, 62(3), 399–410
Wei, X., Zhao, L., Liu, J., Dodel, R. C., Farlow, M. R., & Du, Y. (2005). Minocycline prevents gentamicin-induced ototoxicity by inhibiting p38 MAP kinase phosphorylation and caspase 3 activation. Neuroscience, 131(2), 513–521
Guo, G., & Bhat, N. R. (2007). p38alpha MAP kinase mediates hypoxia-induced motor neuron cell death: A potential target of minocycline’s neuroprotective action. Neurochemical Research, 32(12), 2160–2166
El Benna, J., Han, J., Park, J. W., Schmid, E., Ulevitch, R. J., & Babior, B. M. (1996). Activation of p38 in stimulated human neutrophils: phosphorylation of the oxidase component p47phox by p38 and ERK but not by JNK. Archives of Biochemistry and Biophysics, 334(2), 395–400
Park, J. G., Yuk, Y., Rhim, H., Yi, S. Y., & Yoo, Y. S. (2002). Role of p38 MAPK in the regulation of apoptosis signaling induced by TNF-alpha in differentiated PC12 cells. Journal of Biochemistry and Molecular Biology, 35(3), 267–272
Shao, X., Yang, X., Shen, J., Chen, S., Jiang, X., & Wang, Q., et al. (2020). TNF-α–induced p53 activation induces apoptosis in neurological injury. Journal of Cellular and Molecular Medicine, 24, 6796–6803
El-Benna, J., Hurtado-Nedelec, M., Marzaioli, V., Marie, J. C., Gougerot-Pocidalo, M. A., & Dang, P. M. (2016). Priming of the neutrophil respiratory burst: Role in host defense and inflammation. Immunological Reviews, 273(1), 180–193
Sumimoto, H., Hata, K., Mizuki, K., Ito, T., Kage, Y., Sakaki, Y., Fukumaki, Y., Nakamura, M., & Takeshige, K. (1996). Assembly and activation of the phagocyte NADPH oxidase. Specific interaction of the N-terminal Src homology 3 domain of p47phox with p22phox is required to activate the NADPH oxidase. The Journal of Biological Chemistry, 271(36), 22152–22158
Acknowledgements
We would like to thank the Neuroscience Research Center and Neuropharmacology Institute for providing access to their research facilities. This work was supported by the research council of the Kerman University of Medical Sciences, Kerman, Iran. Also, we appreciate the Faculty of Pharmacy, Hormozgan University of Medicinal Sciences, for providing the supercomputing service.
Funding
Financial support for this study was provided by the research council of the Kerman University of Medical Sciences, Kerman, Iran. The funding facilitated various aspects of the research, including data collection, analysis, and interpretation.
Author information
Authors and Affiliations
Contributions
Habib Eslami and Kaveh Haji-Allahverdipoor led the conception, design, and coordination of the study, overseeing data collection and conducting experiments and simulation validation. Kaveh Haji-Allahverdipoor also served as the supervisor for the computational process and simulation validation. Koosha Rokhzadi operated in the simulation validation (computational process) and was involved in manuscript preparation and editing. Mohsen Basiri, Saeed Esmaeili-Mahani, and Zahra Mahmoodi contributed to data analysis, interpretation, and overall project management for manuscript preparation. Kaveh Haji-Allahverdipoor assisted in data collection, conducted the literature review, and played a significant role in manuscript revision. All authors reviewed and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interest.
Ethical Approval
Importantly, it is noteworthy that no human or animal subjects were used in this study, and the research strictly adhered to ethical guidelines and principles without involving any living organisms. Informed consent was obtained from all participants in adherence to committee guidelines.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Eslami, H., Rokhzadi, K., Basiri, M. et al. Direct Interaction of Minocycline to p47phox Contributes to its Attenuation of TNF-α-Mediated Neuronal PC12 Cell Death: Experimental and Simulation Validation. Cell Biochem Biophys (2024). https://doi.org/10.1007/s12013-024-01279-9
Accepted:
Published:
DOI: https://doi.org/10.1007/s12013-024-01279-9