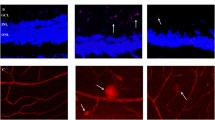
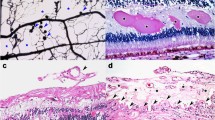
Abstract
Accumulated evidences support diabetic retinopathy is caused by functional and structural alterations in both the microvascular and neuroglial compartments. Here, we explored any beneficial effects of combination of Bevacizumab and NGF to prevent diabetic retinopathy. STZ-induced diabetic rats were randomly grouped into three groups: sham, Bevacizumab and combination group (Bevacizumab + NGF). End of the treatment, vascular abnormality was evaluated by FITC-dextran. Inflammation and angiogenesis factors were also investigated by real-time PCR and western blot. Our results showed both Bevacizumab and the combination could dramatically reduce abnormal vascular permeability and limit the inflammatory and angiogenesis in 6-week diabetic rats. However, the combination indicated better preventive effects in 12-week diabetic rats compared with Bevacizumab alone. Our findings suggest that the combination of Bevacizumab with NGF may be a new method for the preventive intervention of diabetic retinopathy.






Similar content being viewed by others
References
Pascolini, D., & Mariotti, S. P. (2012). Global estimates of visual impairment: 2010. British Journal of Ophthalmology, 96, 614–618.
Sivaprasad, S., Gupta, B., Crosby-Nwaobi, R., & Evans, J. (2012). Prevalence of diabetic retinopathy in various ethnic groups: A worldwide perspective. Survey of Ophthalmology, 57, 347–370.
Yau, J. W., Rogers, S. L., Kawasaki, R., Lamoureux, E. L., Kowalski, J. W., Bek, T., et al. (2012). Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care, 35, 556–564.
Lock, J. H., & Fong, K. C. (2011). An update on retinal laser therapy. Clinical and Experimental Optometry, 94, 43–51.
Giuliari, G. P. (2012). Diabetic retinopathy: Current and new treatment options. Current Diabetes Review, 8, 32–41.
Stewart, M. W. (2012). Corticosteroid use for diabetic macular edema: Old fad or new trend? Current Diabetes Reports, 12, 364–375.
Adamis, A. P., Miller, J. W., Bernal, M. T., Folkman, J., Yeo, T. K., et al. (1994). Increased vascular endothelial growth factor levels in the vitreous of eyes with proliferative diabetic retinopathy. American Journal of Ophthalmology, 118, 445–450.
Aiello, L. P., Avery, R. L., Arrigg, P. G., Keyt, B. A., Jampel, H. D., Shah, S. T., et al. (1994). Vascular endothelial growth factor in ocular fluid of patients with diabetic retinopathy and other retinal disorders. New England Journal of Medicine, 331, 1480–1487.
Zechmeister-Koss, I., & Huic, M. (2012). Vascular endothelial growth factor inhibitors (anti-VEGF) in the management of diabetic macular oedema: a systematic review. British Journal of Ophthalmology, 96, 167–178.
Barber, A. J., Lieth, E., Khin, S. A., Antonetti, D. A., Buchanan, A. G., & Gardner, T. W. (1998). Neural apoptosis in the retina during experimental and human diabetes. Early onset and effect of insulin. Journal of Clinical Investigation, 102, 783–791.
Mizutani, M., Gerhardinger, C., & Lorenzi, M. (1998). Muller cell changesin human diabetic retinopathy. Diabetes, 47, 445–449.
Colafrancesco, Valeria, Coassin, Marco, Rossi, Simona, & Aloe, Luigi. (2011). Effect of eye NGF administration on two animal models of retinal ganglion cells degeneration. Annali dell Superiore di Sanità, 47(3), 284–289.
Colafrancesco, V., Coassin, M., Rossi, S., & Aloe, L. (2011). Effect of eye NGF administration on two animal models of retinal ganglion cells degeneration. Annali dell Istituto Superiore di Sanita, 47(3), 284–289.
Ma, J., Zhu, T., Tang, X., Ye, P., & Zhang, Z. (2010). Effect of an intravitreal injection of bevacizumab on the expression of VEGF and CD34 in the retina of diabetic rats. Clinical and Experimental Ophthalmology, 38(9), 875–884.
Yoshimura, T., Sonoda, K. H., Sugahara, M., Mochizuki, Y., Enaida, H., et al. (2009). Comprehensive analysis of inflammatory immune mediators in vitreoretinal diseases. PLoS One, 4, e8158.
Elner, S. G., Elner, V. M., Jaffe, G. J., Stuart, A., Kunkel, S. L., et al. (1995). Cytokines in proliferative diabetic retinopathy and proliferative vitreoretinopathy. Current Eye Research, 14, 1045–1053.
Fonollosa, A., Garcia-Arumi, J., Santos, E., Macia, C., Fernandez, P., et al. (2010). Vitreous levels of interleukine-8 and monocyte chemoattractant protein-1 in macular oedema with branch retinal vein occlusion. Eye (Lond), 24, 1284–1290.
Schoenberger, S. D., Kim, S. J., Sheng, J., Rezaei, K. A., Lalezary, M., et al. (2012). Increased prostaglandin E2 (PGE2) levels in proliferative diabetic retinopathy, and correlation with VEGF and inflammatory cytokines. Investigative Ophthalmology and Visual Science, 53, 5906–5911.
Wakabayashi, Y., Usui, Y., Okunuki, Y., Kezuka, T., Takeuchi, M., et al. (2010). Correlation of vascular endothelial growth factor with chemokines in the vitreous in diabetic retinopathy. Retina, 30, 339–344.
Watanabe, D., Suzuma, K., Suzuma, I., Ohashi, H., Ojima, T., et al. (2005). Vitreous levels of angiopoietin 2 and vascular endothelial growth factor in patients with proliferative diabetic retinopathy. American Journal of Ophthalmology, 139, 476–481.
Freyberger, H., Bröcker, M., Yakut, H., Hammer, J., Effert, R., et al. (2000). Increased levels of platelet-derived growth factor in vitreous fluid of patients with proliferative diabetic retinopathy. Experimental and Clinical Endocrinology and Diabetes, 108, 106–109.
Butler, J. M., Guthrie, S. M., Koc, M., Afzal, A., Caballero, S., et al. (2005). SDF-1 is both necessary and sufficient to promote proliferative retinopathy. Journal of Clinical Investigation, 115, 86–93.
Manzano, R. P., Peyman, G. A., Khan, P., et al. (2007). Inhibition of experimental corneal neovascularization by bevacizumab (Avastin). British Journal of Ophthalmology, 91, 804–807.
Klettner, A., & Roider, J. (2008). Comparison of bevacizumab, ranibizumab, and pegaptanib in vitro: Efficiency and possible additional pathways. Investigative Ophthalmology and Visual Science, 49, 4523–4527.
Ameri, H., Chader, G. J., Kim, J., et al. (2007). The effects of intravitreous bevacizumab on retinal neovascular membrane and normal capillaries in rabbits. Investigative Ophthalmology and Visual Science, 48, 5708–5715.
Lassota, N., Prause, J. U., Scherfig, E., et al. (2010). Clinical and histological findings after intravitreal injection of bevacizumab (Avastin(R) in a porcine model of choroidal neovascularization. Acta Ophthalmologica, 88, 300–308.
Heiduschka, P., Fietz, H., Hofmeister, S., et al. (2007). Penetration of bevacizumab through the retina after intravitreal injection in the monkey. Investigative Ophthalmology and Visual Science, 48, 2814–2823.
Bloodworth, J. M, Jr. (1962). Diabetic retinopathy. Diabetes, 11, 1–22.
Wolter, J. R. (1961). Diabetic retinopathy. American Journal of Ophthalmology, 51, 1123–1141.
Hammes, H. P., Federoff, H. J., & Brownlee, M. (1995). Nerve growth factor prevents both neuroretinal programmed cell death and capillary pathology in experimental diabetes. Molecular Medicine, 1, 527–534.
Martin, P. M., Roon, P., Van Ells, T. K., Ganapathy, V., & Smith, S. B. (2004). Death of retinal neurons in streptozotocin-induced diabetic mice. Investigative Ophthalmology and Visual Science, 45, 3330–3336.
Ali, T. K., Matragoon, S., Pillai, B. A., Liou, G. I., & El-Remessy, A. B. (2008). Peroxynitrite mediates retinal neurodegeneration by inhibiting nerve growth factor survival signaling in experimental and human diabetes. Diabetes, 57, 889–898.
El-Remessy, A. B., Khalifa, Y., Ola, S., & Liou, G. (2010). Diabetes-induced tyrosine nitration impairs glutamine synthetase activity: Protective effects of cannabidiol. Molecular Vision, 16, 1487–1495.
Connor, B., & Dragunow, M. (1998). The role of neuronal growth factors in neurodegenerative disorders of the human brain. Brain Research, 27, 1–39.
Yune, T. Y., Lee, J. Y., Jung, G. Y., Kim, S. J., Jiang, M. H., Kim, Y. C., et al. (2007). Minocycline alleviates death of oligodendrocytes by inhibiting pro-nerve growth factor production in microglia after spinal cord injury. Journal of Neuroscience, 27, 7751–7761.
Lee, R., Kermani, P., Teng, K. K., & Hempstead, B. L. (2001). Regulation of cell survival by secreted proneurotrophins. Science, 294, 1945–1948.
Von Bartheld, C. S. (1998). Neurotrophins in the developing and regenerating visual system. Histology and Histopathology, 13, 437–459.
Ali, T. K., Al-Gayyar, M. M. H., Matragoon, S., Pillai, B. A., Abdelsaid, M. A., Nussbaum, J. J., & El-Remessy, A. B. (2010). Diabetes-induced peroxynitrite impairs the balance of ProNGF/NGF and causes neurovascular injury. Diabetologia, 54(3), 657–668.
Jee, D., & Lee, W. K. (2012). Inhibitory effect of intravitreal injection of bevacizumab on nerve growth factor. Current Eye Research, 37(5), 408–415.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, P., Zhou, Z. Combination of Bevacizumab and NGF Reduces the Risk of Diabetic Retinopathy. Cell Biochem Biophys 73, 79–85 (2015). https://doi.org/10.1007/s12013-015-0564-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-015-0564-1