Abstract
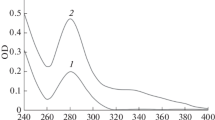
In diabetes, protein glycation mostly occurs at intrachain lysine residues resulting in the formation of early stage Amadori products which are finally converted to advance glycation end products (AGEs). Several studies have reported autoantibodies against AGEs in diabetes but not much data are found in respect of Amadori products. In this study, poly-l-lysine (PLL) was glycated with 50 mM glucose and the resultant Amadori products were estimated by fructosamine or nitroblue tetrazolium assay. We report high content of Amadori products in PLL upon glycation. Glycated PLL showed marked hyperchromicity in the UV spectrum, ellipticity changes in CD spectroscopy, and variations in ε-methylene protons shift in NMR. It was better recognized by autoantibodies in type 2 diabetics compared to the native PLL. Induced antibodies against glycated PLL were successfully used to probe early glycation in the IgG isolated from diabetes type 2 patients. Role of Amadori products of glycated proteins in the induction of autoantibodies in type 2 diabetes as well as in associated secondary complications has been discussed.





Similar content being viewed by others
Abbreviations
- PLL:
-
Poly-l-lysine
- NBT:
-
Nitroblue tetrazolium
- HMF:
-
5-Hydroxymethylfurfural
- NaBH4 :
-
Sodium borohydride
- AGEs:
-
Advanced glycation end products
- UV:
-
Ultraviolet
- CD spectroscopy:
-
Circular dichroism
- NMR:
-
Nuclear magnetic resonance
- ELISA:
-
Enzyme-linked immunosorbent assay
References
Ansari, N. A., & Moinuddin, (2013). Amadori glycated proteins: role in production of autoantibodies in diabetes mellitus and effect of inhibitors on non-enzymatic glycation. Aging and Disease, 4(1), 50–56.
Armbruster, D. A. (1987). Fructosamine: structure, analysis and clinical usefulness. Clinical Chemistry, 33, 2153–2163.
Schalkwijk, G. C., & Miyata, T. (2012). Early- and advanced non-enzymatic glycation in diabetic vascular complications: The search for therapeutics. Amino Acids, 42(4), 1193–1204.
Khan, M. W. A., Qadrie, Z. L., & Khan, W. A. (2010). Antibodies against gluco-oxidatively modified human serum albumin detected in diabetes-associated complications. International Archives of Allergy and Immunology, 153, 207–214.
Ansari, N. A., Moinuddin, Alam, K., & Ali, A. (2009). Preferential recognition of Amadori-rich lysine residues by serum antibodies in diabetes mellitus: Role of protein glycation in the disease process. Human Immunology, 70, 417–424.
Arif, B., Ashraf, J. M., Moinuddin, Ahmad, J., Arif, Z., & Alam, K. (2012). Structural and immunological characterization of Amadori-rich human serum albumin: role in diabetes mellitus. Archives of Biochemistry and Biophysics, 522(1), 17–25.
Fu, M. X., Wells-knecht, K. J., Blackledge, J. A., Lyons, T. J., Thorpe, S. R., & Baynes, J. W. (1994). Glycation, glycoxidation and cross-linking of collagen by glucose. Diabetes, 43, 676–683.
Ahmed, M. U., Thorpe, S. R., & Baynes, J. W. (1986). Identification of N epsilon-carboxymethyllysine as a degradation product of fructose lysine in glycated protein. Journal of Biological Chemistry, 261, 4889–4894.
Hunt, J. V., Bottoms, M. A., Clare, K., Skamarauskas, J. T., & Mitchinson, M. J. (1994). Glucose oxidation and low-density lipoprotein-induced macrophage ceroid accumulation. Biochemical Journal, 300, 243–249.
Johnson, R., Metcalf, P. A., & Baker, J. R. (1982). Fructosamine: a new approach to the estimation of serum glycosylprotein. Clinica Chimica Acta, 127, 87–95.
Fluckiger, R., & Winterhalter, K. H. (1976). In vitro synthesis of hemoglobin A1C. FEBS Letters, 71, 356–360.
Nagai, R., Ikeda, K., Higashi, T., et al. (1997). Hydroxyl radical mediates N ε-(carboxymethyl) lysine formation from Amadori product. Biochemical and Biophysical Research Communications, 234, 167–172.
Njoroge, F. G., Fernandes, A. A., & Monnier, V. M. (1988). Mechanism of formation of the putative advanced glycosylation end product and protein cross-link 2-(2-furoyl)-4(5)-(2-furanyl)-lH-imidazole. Journal of Biological Chemistry, 263, 10646–10652.
Ali, R., & Alam, K. (2002). Evaluation of antibodies against oxygen free radical-modified DNA by ELISA. In D. Armstrong (Ed.), Methods in molecular biology: Oxidative stress biomarkers and antioxidants protocols (pp. 171–181). New Jersey: Humana Press.
Habib, S., & Moinuddin, Ali R. (2005). Acquired antigenicity of DNA after modification with peroxynitrite. International Journal of Biological Macromolecules, 35, 221–225.
Dixit, K., Moinuddin, & Ali, A. (2005). Immunological studies on peroxynitrite modified human DNA. Life Sciences, 77, 2626–2642.
Towbin, H., Staehelin, T., & Gordon, J. (1979). Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets, procedure and some applications. Proceedings of the National Academy of Sciences of the United States of America, 76, 4350–4354.
Ansari, N. A., & Moinuddin, Ali R. (2011). Physico-chemical analysis of poly-l-lysine: An insight into the changes induced in lysine residues of proteins upon modification with glucose. IUBMB Life, 63, 26–29.
Graf, E., Empson, K. L., & Eaton, J. W. (1987). Phytic acid a natural antioxidant. Journal of Biological Chemistry, 262(11647), 11650.
Graf, E., Moheney, J. R., & Bryant, R. G. (1984). Iron catalysed hydroxyl radical formation. Journal of Biological Chemistry, 259, 3620–3624.
Kim, J., Hong, C., Koo, Y., Choi, H., & Lee, K. (2012). Anti-glycation effect of gold nanoparticles on collagen. Biological and Pharmaceutical Bulletin, 35, 260–264.
Ney, K. A., Colley, K. J., & Pizzo, S. V. (1981). The standardization of thiobarbituric acid assay for nonenzymatic glucosylation of human serum albumin. Analytical Biochemistry, 118, 294–298.
Tsuchiya, S., Sakurai, T., & Sekiguchi, S. (1984). Nonenzymatic glucosylation of human serum albumin and its influence on binding capacity of sulfonylureas. Biochemical Pharmacology, 33, 2967–2971.
Rosenheck, K., & Doty, P. (1961). The far ultraviolet absorption spectra of polypeptide and protein solutions and their dependence on conformation. Proceedings of the National Academy of Sciences of the United States of America, 47, 1775–1785.
Rahmanpour, R., & Bathai, S. Z. (2011). Histone H1 structural changes and its interaction with DNA in the presence of high glucose concentration in vivo and in vitro. Journal of Biomolecular Structure & Dynamics, 28(4), 575–586.
Oliveira, L. M. A., Lages, A., Gomes, R. A., Neves, H., Família, C., Coelho, A. V., et al. (2011). Insulin glycation by methylglyoxal results in native-like aggregation and inhibition of fibril formation. BMC Biochemistry, 12, 41.
Liang, J. N. (1990). Circular dichroism of the non-enzymatic browning products of poly-l-lysine and albumin. International Journal of Biological Macromolecules, 12, 273–277.
Tressi, R., Piechotta, C. T., Rewicki, D. T., & Krause, E. (2002). Modification of peptide lysine during Maillard reaction of d-glucose and d-lactose. International Congress Series, 1245, 203–209.
Kalia, K., Sharma, S., & Mistry, K. (2004). Non-enzymatic glycosylation of immunoglobulins in diabetic nephropathy. Clinica Chimica Acta, 347, 169–176.
Stig, B., & Hajdu, N. (2008). Endproducts and receptor of advanced glycation and lipoxidation (AGE, ALE, RAGE) and chronic diseases—a food perspective. Orvosi Hetilap, 149, 771–778.
Jaleel, A., Halvatsiotis, P., & Williamson, B. (2005). Identification of Amadori-modified plasma proteins in type 2 diabetes and the effect of short-term intensive insulin treatment. Diabetes Care, 28, 645–652.
Cohen, M. P., & Ziyadeh, F. N. (1994). Amadori glucose adducts modulate mesangial cell growth and collagen gene expression. Kidney International, 45, 475–484.
Sakai, H., Jinde, K., Suzuki, D., Yagame, M., & Nomoto, Y. (1996). Localization of glycated proteins in the glomeruli of patients with diabetic nephropathy. Nephrology Dialysis Transplantation, 11, 66–71.
Ibrahim, A. S., El-Remessy, A. B., Matragoon, S., Zhang, W., Patel, Y., Khan, S., et al. (2011). Retinal microglial activation and inflammation induced by Amadori-glycated albumin in a rat model of diabetes. Diabetes, 60(4), 1122–1133.
Monnier, V. M., Sell, D. R., Dai, Z., Nemet, I., Collard, F., & Zhang, J. (2008). The role of the Amadori product in complications of diabetes. Annals of the New York Academy of Sciences, 1126, 81–88.
Amore, A., Cirina, P., Mitola, S., Peruzzi, L., Gianoglio, B., Rabbone, I., et al. (1997). Nonenzymatically glycated albumin (Amadori adducts) enhances nitric oxide synthase activity and gene expression in endothelial cells. Kidney International, 51, 27–35.
Predescu, D., Simionescu, M., Simionescu, N., & Palade, G. (1988). Binding and transcytosis of glycoalbumin by the microvascular endothelium of the murine myocardium: Evidence that glycoalbumin behaves as a bifunctional ligand. Journal of Cell Biology, 107, 1729–1738.
Cohen, M. P., Hud, E., Wu, V. Y., & Shearman, C. W. (2008). Amelioration of diabetes-associated abnormalities in the vitreous fluid by an inhibitor of albumin glycation. Investigative Ophthalmology & Visual Science, 49, 5089–5093.
Schalkwijk, C. G., Ligtvoet, N., Twaalfhoven, H., Jager, A., Blaauwgeers, H. G., Schlingemann, R. O., et al. (1999). Amodori albumin in type 1 diabetic patients, correlation with markers of endothelial function associated with diabetic nephropathy and localization in retinal capillaries. Diabetes, 48, 2446–2453.
Kaneshige, H. (1987). Nonenzymatic glycosylation of serum IgG and its effect on antibody activity in patients with diabetes mellitus. Diabetes, 36, 822–828.
Ahmad, S., & Moinuddin, Ali A. (2012). Immunological studies on glycated human IgG. Life Sciences, 90, 980–987.
Lapolla, A., Molin, L., & Traldi, P. (2013). Protein glycation in diabetes as determined by mass spectrometry. International Journal of Endocrinology, 2013, 412103. doi:10.1155/2013/412103.
Acknowledgments
We are thankful to Sophisticated Analytical Instrument Facility of Central Drug Research Institute, Lucknow (India) for NMR experiments. Thanks are also due to Prof. Jamal Ahmad (Department of Medicine, J.N. Medical College, Aligarh Muslim University) for providing the type 2 diabetes mellitus samples. Financial assistance from UPCST, Lucknow and ICMR, New Delhi to Moinuddin is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ansari, N.A., Moinuddin, Mir, A.R. et al. Role of Early Glycation Amadori Products of Lysine-Rich Proteins in the Production of Autoantibodies in Diabetes Type 2 Patients. Cell Biochem Biophys 70, 857–865 (2014). https://doi.org/10.1007/s12013-014-9991-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-014-9991-7