Abstract
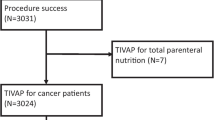
The purpose of this study was to investigate the incidence and risk factors of infections associated with totally implantable venous-access ports (TIVAPs) in our hospital. From April 2007 through November 2013, a total of 1,026 patients with TIVAP were included in this study. We retrospectively analyzed the incidence of port-related infection, patient characteristics, and history. Risk factors for IVAP-related infection were determined statistically. Overall, 97 (9.59 %) of 1,026 infectious cases were reported. By reviewing the medical record of the patients, we found that younger age, hematogenous malignancy, and palliative chemotherapy were associated with higher infection incidence rate in patients who had implanted TIVAP (P < 0.05) in the subgroup analysis. In contrast, gender and insertion site were irrelevant factors in predicting the infection risk. Overall, TIVAP were proved to be safe and effective. Younger age, hematogenous malignancy, and palliative chemotherapy were associated with higher infection in patients who had implanted TIVAP.
Similar content being viewed by others
References
Pittiruti, M., Hamilton, H., Biffi, R., MacFie, J., & Pertkiewicz, M. (2009). Espen, ESPEN guidelines on parenteral nutrition: Central venous catheters (access, care, diagnosis and therapy of complications). Clinical Nutrition, 28, 365–377.
Niederhuber, J. E., Ensminger, W., Gyves, J. W., Liepman, M., Doan, K., & Cozzi, E. (1982). Totally implanted venous and arterial access system to replace external catheters in cancer treatment. Surgery, 92, 706–712.
Vescia, S., Baumgartner, A. K., Jacobs, V. R., Kiechle-Bahat, M., Rody, A., Loibl, S., & Harbeck, N. (2008). Management of venous port systems in oncology: A review of current evidence. Annals of Oncology: Official Journal of The European Society for Medical Oncology/ESMO, 19, 9–15.
Nocito, A., Wildi, S., Rufibach, K., Clavien, P. A., & Weber, M. (2009). Randomized clinical trial comparing venous cutdown with the Seldinger technique for placement of implantable venous access ports. The British Journal of Surgery, 96, 1129–1134.
Barbetakis, N., Asteriou, C., Kleontas, A., & Tsilikas, C. (2011). Totally implantable central venous access ports. Analysis of 700 cases. Journal of Surgical Oncology, 104, 654–656.
Kurul, S., Saip, P., & Aydin, T. (2002). Totally implantable venous-access ports: local problems and extravasation injury. The lancet Oncology, 3, 684–692.
Schwarz, R. E., Groeger, J. S., & Coit, D. G. (1997). Subcutaneously implanted central venous access devices in cancer patients: A prospective analysis. Cancer, 79, 1635–1640.
Kock, H. J., Pietsch, M., Krause, U., Wilke, H., & Eigler, F. W. (1998). Implantable vascular access systems: experience in 1500 patients with totally implanted central venous port systems. World Journal of Surgery, 22, 12–16.
Chang, L., Tsai, J. S., Huang, S. J., & Shih, C. C. (2003). Evaluation of infectious complications of the implantable venous access system in a general oncologic population. American Journal of Infection Control, 31, 34–39.
Hsieh, C. C., Weng, H. H., Huang, W. S., Wang, W. K., Kao, C. L., Lu, M. S., & Wang, C. S. (2009). Analysis of risk factors for central venous port failure in cancer patients. World Journal of Gastroenterology: WJG, 15, 4709–4714.
Fischer, L., Knebel, P., Schroder, S., Bruckner, T., Diener, M. K., Hennes, R., et al. (2008). Reasons for explantation of totally implantable access ports: a multivariate analysis of 385 consecutive patients. Annals of Surgical Oncology, 15, 1124–1129.
Groeger, J. S., Lucas, A. B., Thaler, H. T., Friedlander-Klar, H., Brown, A. E., Kiehn, T. E., & Armstrong, D. (1993). Infectious morbidity associated with long-term use of venous access devices in patients with cancer. Annals of Internal Medicine, 119, 1168–1174.
Bassi, K. K., Giri, A. K., Pattanayak, M., Abraham, S. W., & Pandey, K. K. (2012). Totally implantable venous access ports: retrospective review of long-term complications in 81 patients. Indian Journal of Cancer, 49, 114–118.
Mermel, L.A., Farr, B.M., Sherertz, R.J., Raad, I.I., O’Grady, N., Harris, J.S., Craven, D.E. (2001). A. Infectious Diseases Society of, M. American College of Critical Care, A. Society for Healthcare Epidemiology of, Guidelines for the management of intravascular catheter-related infections. Clinical Infectious Diseases: An Official Publication of The Infectious Diseases Society of America 32, 1249–1272.
Mermel, L. A., Allon, M., Bouza, E., Craven, D. E., Flynn, P., O’Grady, N. P., et al. (2009). Clinical practice guidelines for the diagnosis and management of intravascular catheter-related infection: 2009 Update by the Infectious Diseases Society of America. Clinical Infectious Diseases: An Official Publication of The Infectious Diseases Society of America, 49, 1–45.
Shim, J., Seo, T. S., Song, M. G., Cha, I. H., Kim, J. S., Choi, C. W., et al. (2014). Incidence and risk factors of infectious complications related to implantable venous-access ports. Korean Journal of Radiology: Official Journal of the Korean Radiological Society, 15, 494–500.
Hickman, R. O., Buckner, C. D., Clift, R. A., Sanders, J. E., Stewart, P., & Thomas, E. D. (1979). A modified right atrial catheter for access to the venous system in marrow transplant recipients. Surgery, Gynecology & Obstetrics, 148, 871–875.
Broviac, J. W., Cole, J. J., & Scribner, B. H. (1973). A silicone rubber atrial catheter for prolonged parenteral alimentation. Surgery, Gynecology & Obstetrics, 136, 602–606.
Reed, W. P., Newman, K. A., de Jongh, C., Wade, J. C., Schimpff, S. C., Wiernik, P. H., & McLaughlin, J. S. (1983). Prolonged venous access for chemotherapy by means of the Hickman catheter. Cancer, 52, 185–192.
Carde, P., Cosset-Delaigue, M. F., Laplanche, A., & Chareau, I. (1989). Classical external indwelling central venous catheter versus totally implanted venous access systems for chemotherapy administration: A randomized trial in 100 patients with solid tumors. European Journal of Cancer & Clinical Oncology, 25, 939–944.
Stanislav, G. V., Fitzgibbons, R. J, Jr, Bailey, R. T, Jr, Mailliard, J. A., Johnson, P. S., & Feole, J. B. (1987). Reliability of implantable central venous access devices in patients with cancer. Archives of Surgery, 122, 1280–1283.
Biffi, R., Corrado, F., de Braud, F., de Lucia, F., Scarpa, D., Testori, A., et al. (1997). Long-term, totally implantable central venous access ports connected to a Groshong catheter for chemotherapy of solid tumours: experience from 178 cases using a single type of device. European Journal of Cancer, 33, 1190–1194.
Silberzweig, J. E., Sacks, D., Khorsandi, A. S., & Bakal, C. W. (2000). Reporting standards for central venous access. Technology Assessment Committee. Journal of Vascular and Interventional Radiology: JVIR, 11, 391–400.
Gebauer, B., El-Sheik, M., Vogt, M., & Wagner, H. J. (2009). Combined ultrasound and fluoroscopy guided port catheter implantation–high success and low complication rate. European Journal of Radiology, 69, 517–522.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Ji, L., Yang, J., Miao, J. et al. Infections Related to Totally Implantable Venous-Access Ports: Long-Term Experience in One Center. Cell Biochem Biophys 72, 235–240 (2015). https://doi.org/10.1007/s12013-014-0443-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-014-0443-1