Abstract
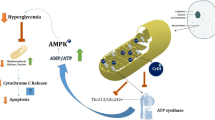
A recent report has shown the active site of the beta subunit of mitochondrial ATP synthase is probably the site of action of Cr(III) action, independent of the insulin signaling pathway. This works appears to answer an important question about the mode of action of Cr(III) at a molecular level when supplied in supra-nutritional levels to rodents. However, as with any good research, the research also raises several questions. The relationship between this study and the results of rodent studies of chromium supplementation and between this study and the current understanding the chromium(III) transport and detoxification system are put into perspective.


Similar content being viewed by others
Data Availability
No datasets were generated during the current study.
Code Availability
Not applicable.
References
Vincent JB (2013) The Bioinorganic Chemistry of Chromium. Wiley, Chichester
Wang H, Hu L, Li H, Lai Y-T, Wei X, Xu X, Cao Z, Cao H, Wan Q, Chang Y-Y, Xu A, Zhou Q, Jiang G, He M-L, Sun H (2023) Mitochondrial ATP synthase as a direct molecular target of chromium(III) to ameliorate hyperglycaemic stress. Nature Commun 14:1738
Donaldson DL, Lee DM, Smith CC, Rennert OM (1985) Glucose tolerance and plasma lipid distributions in rats fed a high-sucrose, high-cholesterol, low-chromium diet. Metabolism 34:1086–1093
Jain R, Verch RL, Wallach S, Peabody RA (1981) Tissue chromium exchange in the rat. Am J Clin Nutr 34:2199–2204
Flatt PR, Juntti-Berggren L, Beggren P-O, Gould BJ, Swanston-Flatt SK (1989) Effects of dietary inorganic trivalent chromium (Cr) on the development of glucose homeostasis in rats. Diabete Metab 15:93–97
National Research Council (2002) Dietary Reference Intakes for Vitamin A, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc. A report of the Panel of Micronutrients, Subcommittee on Upper Reference Levels of Nutrients and of Interpretations and Uses of Dietary Reference Intakes, and the Standing Committee on the Scientific Evaluation of Dietary Reference Intakes. National Academy of Sciences, Washington, DC
Striffler JS, Law JS, Polansky MM, Bhathena SJ, Anderson RA (1995) Chromium improves insulin response to glucose in rats. Metabolism 44:1314–1320
Striffler JS, Polansky MM, Anderson RA (1998) Dietary chromium decreases insulin resistance in rats fed a high-fat, mineral-imbalanced diet. Metabolism 47:396–400
Striffer JS, Polansky MM, Anderson RA (1999) Overproduction of insulin in the chromium-deficient rat. Metabolism 48:1063–1068
Di Bona KR, Love S, Rhodes NR, McAdory D, Sinha SH, Kern N, Kent J, Strickland J, Wilson A, Beaird J, Ramage J, Rasco JF, Vincent JB (2011) Chromium is not an essential trace element for mammals: effects of a “low-chromium” diet. J Biol Inorg Chem 16:381–390
Bertinato J, Griffin P (2023) A low chromium diet increases body fat, energy intake and circulating triglycerides and insulin in male and female rats fed a moderately high-fat, high-sucrose diet from puberty to adult young age. PLoS One 18:e0281019
Sun Y, Clodfelder BJ, Shute AA, Irvin T, Vincent JB (2002) The biomimetic [Cr3O(O2CCH2CH3)6(H2O)3]+ decreases plasma insulin, cholesterol and triglycerides in healthy and type II but not type I diabetic rats. J Biol Inorg Chem 7:852–862
Sun Y, Mallya K, Ramirez J, Vincent JB (1999) The biomimetic [Cr3O(O2CCH2CH3)6(H2O)3]+ decreases plasma cholesterol and triglycerides in rat: Towards chromium-containing therapeutics. J Biol Inorg Chem 4:838–845
Clodfelder BJ, Gullick BM, Lukaski HC, Neggers Y, Vincent JB (2005) Oral administration of the biomimetic [Cr3O(O2CCH2CH3)6(H2O)3]+ increases insulin sensitivity and improves blood plasma variables in healthy and type 2 diabetic rats. J Biol Inorg Chem 10:119–130
Bennett R, Adams B, French A, Neggers Y, Vincent JB (2006) High dose chromium(III) supplementation has no effects on body mass and composition while altering plasma hormone and cholesterol levels. Biol Trace Elem Res 113:53–66
Krol E, Krejpcio Z (2011) Evaluation of antidiabetic potential of chromium(III) propionate complex in streptozotocin-induced experimental diabetes. Food Chem Toxicol 49:3217–3223
Krol E, Krejpcio Z (2010) Chromium(III) propionate complex supplementation improves carbohydrate metabolism in insulin-resistance rat model. Food Chem Toxicol 48:2791–2796
Vincent JB (2014) Is chromium pharmacologically relevant? J Trace Elem Med Biol 28:397–405
Vincent JB (2019) Effects of chromium supplementation on body composition, human and animal health, and insulin and glucose metabolism. Curr Opin Clin Nutr Metab Care 22:483–489
Wang ZQ, Zhang XH, Russell JC, Hulver M, Cefalu WT (2005) Chromium picolinate enhances skeletal muscle cellular insulin signaling in vivo in obese, insulin-resistant JCR:LA-cp rats. J Nutr 136:415–420
Wang H, Kruszewski A, Brautigan DL (2005) Cellular chromium enhances activation of insulin receptor kinase. Biochemistry 44:8167–8175
Brautigan DL, Kruszewski A, Wang H (2006) Chromium and vanadate combination increases insulin-induced glucose uptake by 3T#-L1 adipocytes. Biochem Biophys Res Commun 347:769–773
Miranda ER, Dey CS (2004) Effect of chromium and zinc on insulin signaling in skeletal muscle cells. Biol Trace Elem Res 101:19–36
Yang X, Palanichamy K, Ontko AC, Rao MNA, Fang CX, Ren J, Sreejayan N (2005) A newly synthesized chromium complex – chromium(phenylalanine)3 improves insulin responsiveness and reduces whole body glucose tolerance. FEBS Lett 579:1458–1464
Dong F, Yang X, Sreejayan N, Run J (2007) Chromium (D-phenylalanine)3 improves obesity-induced cardiac contractile defect in ob/ob mice. Obesity 15:2699–2711
Pattar GR, Tackett L, Liu P, Elmendorf JS (2206) Chromium picolinate positively influences the glucose transporter system via affecting cholesterol homeostasis in adipocytes cultured under hyperglycemic diabetic conditions. Mutat Res 610:93–100
Hoffman NJ, Penque BA, Habegger KM, Sealls W, Tackett L, Elmendorf JS (2014) Chromium enhances insulin responsiveness via AMPK. J Nutr Biochem 25:565–572
Yamamoto A, Wada O, Ono T (1987) Isolation of a biologically active low-molecular-mass chromium compound from rabbit liver. Eur J Biochem 165:627–631
Yamamoto A, Wada O, Suzuki H (1988) Purification and properties of biologically active chromium complex from bovine colostrum. J Nutr 118:39–45
Vincent JB (1994) Relationship between glucose tolerance factor and low-molecular-weight chromium-binding substance. J Nutr 124:117–118
Davis CM, Vincent JB (1997) Chromium oligopeptide activates insulin receptor kinase activity. Biochemistry 36:4382–4385
Vincent JB (2000) The biochemistry of chromium. J Nutr 130:715–718
Vincent JB (2000) Elucidating a biological role for chromium at a molecular level. Acc Chem Res 33:503–510
Goldstein BJ, Zhu L, Hager R, Zilbering A, Sun Y, Vincent JB (2001) Enhancement of post-receptor insulin signaling by trivalent chromium in hepatoma cells is associated with differential inhibition of specific protein-tyrosine phosphatases. J Trace Elem Exp Med 14:93–404
Davis CM, Sumrall KH, Vincent JB (1996) A biologically active form of chromium may activate a membrane phosphotyrosine phosphatase (PTP). Biochemistry 35:12963–12969
Deng G, Wu K, Cruce A, Bowman MW, Vincent JB (2015) Binding of trivalent chromium to serum transferrin is sufficiently rapid to be physiologically relevant. J Inorg Biochem 143:48–55
Edwards KC, Kim H, Vincent JB (2020) Release of trivalent chromium from serum transferrin is sufficiently rapid to be physiologically relevant. J Inorg Biochem 202:110901
Edwards KC, Kim H, Ferguson R, Lockart VJB (2020) Significance of conformational changes during the binding and release of chromium(III) from human serum transferrin. J Inorg Biochem 206:111040
Edwards KC, Gannon MW, Frantom PA, Vincent JB (2021) Low-molecular-weight chromium-binding substance (LMWCr) may bind and carry Cr(III) from the endosome. J Inorg Biochem 223:111555
Sun Y, Ramirez J, Woski SA, Vincent JB (2000) the binding of trivalent chromium to low-molecular-weight chromium-binding substance (LMWCr) and the transfer of chromium from transferrin and Cr(pic)3 to LMWCr. J Biol Inorg Chem 5:129–136
Jacquamet L, Sun Y, Hatfield J, Gu W, Cramer SP, Crowder MW, Lorigan GA, Vincent JB, Latour J-M (2003) Characterization of chromodulin by X-ray absorption and electron paramagnetic resonance spectroscopies and magnetic susceptibility measurements. J Am Chem Soc 125:774–780
Costello RB, Dwyer JT, Merkel JM (2019) Chromium supplements in health and disease. In: Vincent JBJ (ed) The Nutritional Biochemistry of Chromium, 2nd edn. Elsevier, Amsterdam, pp 219–249
Gullick, BM (2005) Characterizing the potential therapeutic agent [Cr3O(O2CCH2CH3)6(H2O)3]+: Its uptake and subcellular distribution and the effects of its oral administration on blood variables. Ph.D. dissertation, The University of Alabama
Clodfelder BJ, Emamaullee J, Hepburn DDD, Chakov NE, Nettles HS, Vincent JB (2001) The trail of chromium(III) in vivo from the blood to the urine: The roles of transferrin and chromodulin. J Biol Inorg Chem 6:608–617
Clodfelder BJ, Upchurch RG, Vincent JB (2004) A comparison of the insulin-sensitive transport of chromium in healthy and model diabetic rats. J Inorg Biochem 98:522–533
Clodfelder BJ, Vincent JB (2005) The time-dependent transport of chromium in adult rats from the bloodstream to the urine. J Biol Inorg Chem 10:383–393
Danenberg KD, Cleland WW (1975) Use of chromium-adenosine triphosphate and lyxose to elucidate the kinetic mechanism and coordination state of the nucleotide substrate for yeast hexokinase. Biochemistry 14:28–39
Bossard MJ, Schuster SM (1981) Structural preferences for the binding of chromium nucleotides by beef heart mitochondrial ATPase. J Biol Chem 256:6617–6622
Schuster SM, Ebel RE, Lardy HA (1975) Kinetic studies on rat liver and beef heart mitochondrial adenosine triphosphate: The effects of the chromium complexes of adenosine triphosphate and adenosine diphosphate on the kinetic properties. Arch Biochem Biophys 171:656–661
Mulkani L, Levina A, Lay PA (2004) Biomimetic oxidation of chromium(III): Does the antidiabetic activity of chromium(III) involve carcinogenic chromium(VI)? Angew Chem Int Ed 43:4504–4507
Levina A, Lay PA (2005) mechanistic studies of relevance to the biological activities of chromium. Coord Chem Rev 249:281–298
Cocheme HM, Quin C, McQuaker SJ, Cabreiro F, Logan A, Prime TA, Abakumova I, Patel JV, Fearnley IM, James AM, Porteous CM, Smith RAJ, Saeed S, Carre JE, Singer M, Gems D, Hartley RC, Partridge L, Murphy MP (2011) Measurement of H2O2 within living Drosophila during aging using a ratiometric mass spectrometry probe targeted to the mitochondrial matrix. Cell Metab 13:340–350
Stein KT, Moon SJ, Nguyen AN, Sikes HD (2020) Kinetic modeling of H2O2 dynamics in the mitochondria of HeLa cells. PLoS Comput Biol 16:e1008202
de Cubas L, Pak VV, Belousov VV, Ayte J, Hidalgo E (2021) The mitochondria-to-cytosol H2O2 gradient is caused by peroxiredoxin-dependent cytosolic scavenging. Antioxidants 10:731
Funding
This work was supported by the National Institutes of Health, R15ES033800 (to J.B.V).
Author information
Authors and Affiliations
Contributions
J.B.V. is responsible for all aspects of this manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics Approval
Not applicable.
Conflict of Interest
The author declares no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Vincent, J.B. What Are the Implications of Cr(III) Serving as an Inhibitor of the Beta Subunit of Mitochondrial ATP Synthase?. Biol Trace Elem Res 202, 1335–1344 (2024). https://doi.org/10.1007/s12011-023-03809-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-023-03809-7