Abstract
Magnesium (Mg) is the second most frequent intracellular cation, having an important role in normal enzyme function and insulin secretion. Polycystic ovarian syndrome (PCOS) is the most prevalent endocrinopathy in women of reproductive age and often associated with insulin resistance. Two systematic reviews and meta-analyses have been conducted to compare mean serum Mg levels between PCOS and control groups. Both studies detected unexplained heterogeneity among input studies and the two conclusions contradict each other, while approximately 1% of total body Mg is present in extracellular fluid (ECF) and serum Mg level does not represent Mg status well. For the first time, we investigated magnesium renal fraction excretion (FEMg) and compared mean values between PCOS and non-PCOS control women. This study is a cross-sectional analysis conducted at an academic medical center. Forty-four women were included in the PCOS group based on the Rotterdam criteria and 50 non-PCOS women were included in the control group. Statistical analysis of the relationship between 24-h urinary Mg content and FEMg, and also physical and metabolic variables, was performed. Main outcome measurements are 24-h urinary Mg content and FEMg. Mean values of 24-h urinary Mg content and FEMg did not significantly differ between PCOS and control groups (P = 0.22 and P = 0.24, respectively). Also, serum Mg levels and Ca/Mg ratio were similar between the groups (P = 0.17 and P = 0.26, respectively). Our data suggested Mg status in the PCOS group was similar to the non-PCOS control group and both were not magnesium deficient. For further investigation, we recommend using FEMg for evaluating Mg status rather than serum Mg levels. Considering collection of background diet is helpful and desired for future studies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Polycystic ovarian syndrome (PCOS) is the most prevalent cause of chronic anovulation with androgen excess. It affects 5–10% of women in reproductive age [1]. There is evidence that dietary magnesium (Mg) is inversely associated with obesity, type 2 diabetes mellitus, metabolic syndrome, and chronic hypertension [2,3,4,5,6,7]. So we were encouraged to investigate the role of magnesium status in PCOS as a metabolic disorder.
Mg is the major intracellular divalent cation. Intracellular magnesium forms a key complex with adenosine triphosphate (ATP) and is an important cofactor for a wide range of enzymes, transporters, and nucleic acids required for normal cellular function, replication, and energy metabolism [8]. Several studies have shown significant differences in serum Mg levels in PCOS women in comparison with the control group [9, 10], while there are several studies that found no significant difference [11,12,13,14]. Two systematic reviews and meta-analyses have been conducted in 2020 that investigated serum magnesium levels in PCOS and non-PCOS control groups. They proved unexplained heterogeneity among input studies and their conclusions contradict each other [15, 16]. While Mg is the second most frequent intracellular cation and serum Mg level does not reflect total body Mg status [8], for the first time, this study compared Mg status in PCOS women vs non-PCOS control using 24-h urinary Mg content and magnesium renal fraction excretion (FEMg) rather than serum Mg levels.
Material and Methods
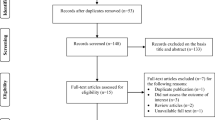
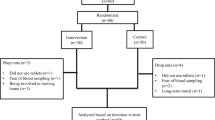
This study was a cross-sectional study conducted at the Milad Infertility Clinic at Imam Reza Hospital, Mashhad University of Medical Sciences, Iran, during 2021. Forty-four PCOS and 50 control non-PCOS women were recruited from outpatients who visited the Milad Infertility Clinic. The study design was approved by local and national ethics committee. Ethical code is IR.MUMS.MEDICAL.REC.1398.541, registered on https://ethics.research.ac.ir/. The study has followed the Declaration of Helsinki guideline, and informed consent was given by all participants.
When the study was proposed, we decided to have participants fulfill the Food Frequency Questionnaire (FFQ), but when faced with the COVID-19 pandemic, we were expected to minimize the exposure, so FFQ was omitted. To conform dietary magnesium intake between PCOS and control groups, a 3-day wash out period was considered. Participants were asked not to consume magnesium-rich food such as cocoa derivatives, nuts, seafood, and green vegetables in this period then go to laboratory. We provided “Guide to 24-h urine collection” in papers and distributed to participants. We also instructed them verbally and answered any question they had about the urine collection process.
The Rotterdam criteria were the basis of PCOS diagnosis in the PCOS group. Each woman with PCOS met at least two of the following criteria:
-
1.
hirsutism or hyperandrogenemia in the absence of alternative known cause
-
2.
oligomenorrhea or dysfunctional uterine bleeding
-
3.
polycystic ovaries on ultrasonography
Infertile women who did not have any of the exclusion criteria were included in the non-PCOS control group. Exclusion criteria were as follows:
-
1.
Known causes of hyperandrogenism and chronic anovulation
-
2.
Previous known endocrinopathies including diabetes mellitus, untreated thyroid abnormalities, or parathyroid abnormalities
-
3.
Chronic hypertension or previous known cardiovascular disease
-
4.
Chronic kidney disease or electrolyte abnormalities including hypokalemia or hypercalcemia
-
5.
Pregnant or breastfeeding women
-
6.
Consumption of drugs and toxins including corticosteroid, Mg-based antacids/cathartics, diuretics (loop, thiazide, osmotic), and ethanol within the past 3 months
Measurements
Variables and measuring methods are listed in Table 1. Anthropometric and clinical variables were asked and measured at the clinic, and then participants were referred to laboratory, received as a routine patient. The main outcomes are 24-h urine Mg content and FEMg. Body mass index (BMI) and age were potential confounders.
Statistical Analysis
The Kolmogorov–Smirnov test was used to define whether the sample drawn complies normal distribution or not. To compare continuous data, the independent t-test was used for normally distributed variables and the Mann–Whitney test for non-normally distributed ones. We used Pearson’s correlation test for parametric and Spearman’s rho test for non-parametric variables to find out if there is correlation between these parameters and magnesium measures. P value < 0.05 was regarded statistically significant for all calculations. The data are reported in accordance with STROBE guidelines (https://www.equator-network.org/reporting-guidelines/strobe/). SPSS 26 statistical software was used to perform all statistical analyses. The statistical variable used to compare the main measurable outcome and calculating sample size was the mean Mg content of 24-h urine sample for both PCOS and control groups.
Sample Size
There was no study investigating 24-h urinary Mg content in PCOS women, so we calculated sample size due to studies conducted on normal Chinese men and type 2 diabetes mellitus patients [17, 18]. Sample size was calculated by Eq. 1 considering type 1 error (α) of 0.05 and type 2 error (β) of 0.20 (power = 80%). We used 31.0 mg/24-h as the effect size (d) of 24-h urine Mg content and 51.7 mg/24 h as standard deviation (SD). With the assumptions, we calculated 44 cases that were required in each group.
Results
3All participants were infertile. All were received at the laboratory and provided a 24-h urine and one-time blood sample. PCOS and control groups were age-matched but BMI was significantly higher in the PCOS group (P = 0.002). Profile of anthropometric and laboratory characteristics of PCOS and control groups is presented in Table 2. Frequency of important clinical features is listed in Table 3. Serum Mg levels were similar between the groups (P value = 0.17). There was no significant difference in 24-h urinary Mg content (P value = 0.22) and FEMg (P value = 0.24) between the PCOS and control groups.
In medical practice, many clinicians request 24-h urine creatinine to check if urine collection is acceptable. The acceptable range is 500–2000 mg. We did not exclude any participants because of unacceptable urine collection. There were 2 participants with 24-h urine creatinine less than 500 mg, one 440 mg and the other 335 mg, both in the PCOS group. Urine volumes were respectively 1100 mL and 650 mL. No participant had urine creatinine more than 2000 mg.
Discussion
The results of this study demonstrate similar magnesium status between PCOS and non-PCOS control groups. No significant difference was seen in 24-h urinary Mg content or FEMg between PCOS and control groups (P = 0.22 and P = 0.24, respectively). Also, serum magnesium level was similar between the groups (P = 0.17).
Ca/Mg ratio was calculated by dividing the corrected Ca by the Mg: corrected Ca (mg/dL) = measured Ca (mg/dL) + [4 − albumin (g/dL)]. There was no significant difference among the groups (P value = 0.26).
PCOS is considered a heterogeneous disorder with multifactorial causes. Pathophysiology of PCOS is complex and incompletely understood. During the reproductive years, PCOS is associated with important reproductive morbidity, including infertility, irregular uterine bleeding, and increased pregnancy loss [1].
PCOS is a metabolic disorder and in many cases will be associated with diabetes mellitus, chronic hypertension, and metabolic syndrome. It is evident that dietary Mg is inversely associated with obesity, type 2 diabetes mellitus, metabolic syndrome, and chronic hypertension [2,3,4,5,6,7]. Intracellular magnesium is crucial for normal energy metabolism, as a cofactor for ATP and numerous enzymes and transporters, which is reflected in the rather global clinical effects that accompany disorders of magnesium homeostasis [19].
In a systematic review and meta-analysis, Babapour et al. focused on 8 studies and proved serum Mg level is lower in PCOS women than the control group (WMD, 95% CI; − 0.09 (− 0.17, − 0.02) mmol/L; P = 0.01). Also, they detected significant heterogeneity among the studies (I2 = 98.0%, P < 0.001). They classified studies based on BMI classes but they did not detect the source of heterogeneity. They observed significant differences in the pooled effect sizes of Mg concentration between PCOS and control groups in the studies conducted on overweight or obese women (− 0.07 (− 0.14, − 0.01) mmol/L; P = 0.02). They observed no difference in Mg concentration between the groups in the subgroup of normal BMI (− 0.11 (− 0.25, 0.04) mmol/L; P = 0.14) [16].
In another systematic review and meta-analysis, Yin et al. focused on 7 studies. They detected no significant difference in serum Mg levels between PCOS and control groups (SMD = − 0.40, 95% CI: − 1.04 to 0.23). Again, significant levels of heterogeneity were detected (I2 = 97%). They observed serum Mg was not significantly different between PCOS patients and healthy controls, irrespective of whether or not PCOS women were obese (overweight/obese: SMD = − 1.05, 95% CI: − 2.47 to 0.37; normal weight: SMD = − 1.11, 95% CI: − 3.24 to 1.02) [15].
Mg is an intracellular cation and serum levels may not reflect body Mg status well. According to Al-Ghamdi et al., a decrease of 24-h Mg excretion less than 12 mg is convincing evidence of magnesium deficiency for individuals. Also stated, FEMg > 0.5% or more than 24 mg in 24-h urine rules out magnesium deficiency [20]. For calculating fractional excretion of magnesium, both serum magnesium and 24-h urinary magnesium are considered. Serum magnesium concentration reflects intestinal magnesium absorption; fractional excretion of magnesium can be useful even if dietary magnesium is not measured. It can be an appropriate stand-alone indicator for magnesium status according to Al-Ghamdi et al.
Hypothetically, the heterogeneity observed in both systematic reviews and meta-analyses conducted on serum Mg levels may reveal the fact that serum Mg does not reflect magnesium status. For further investigation, we recommend using FEMg for evaluating Mg status rather than serum Mg levels.
Mg absorption was found to be normal or minimally reduced in vitamin D–deficient patients and vitamin D repletion resulted in a small, although significant increase in magnesium absorption, while overall balance was not affected due to increased urinary magnesium excretion. In our study [20], vitamin D mean values were similar between the PCOS and control groups (P value = 0.41).
Serum creatinine was significantly different between PCOS and control groups (P value = 0.02), and also serum potassium and albumin (respectively P = 0.007 and P = 0.033). One of the important reasons we measured these three parameters was to check similarity between PCOS and control groups, to find out if the groups are comparable or not. We used Pearson’s correlation test for parametric and Spearman’s rho test for non-parametric variables to find out if there is correlation between these parameters and magnesium measures. The results are listed in Table 4.
It is known that serum calcium is correlated with albumin. We observed it in our study. But there was no correlation between these three statistically significant parameters and magnesium measures. Ideally, we desire a control group with similar mean levels of albumin, serum creatinine, and serum potassium. Results of this study and variables we measured can be material of a bigger systematic review and meta-analysis.
It is known that hypomagnesemia will affect PTH receptor sensitivity and cause resistant hypokalemia and hypocalcemia. This effect happens at low levels of magnesium status when it is symptomatic. Although differences in serum creatinine, serum potassium, and albumin are statistically significant between PCOS and control groups, it does not mean that it has clinical importance.
In the present study, BMI was significantly higher in the PCOS group (P = 0.002). Sample size was not large enough for linear regression to assess BMI effect as a confounder. All participants were infertile; the fact should be considered while evaluating external validity of the research.
Conclusion
Our study suggested Mg status in the PCOS group was similar to the non-PCOS control group, and both were not Mg deficient. There was no clue of magnesium deficiency so the hypothesis comes to mind which use of Mg supplements would not be helpful in PCOS especially infertile PCOS women as well as infertile non-PCOS women.
For further investigation, we recommend using FEMg for evaluating Mg status rather than serum Mg levels. Also, considering collection of background diet is helpful and desired for future studies.
Data Availability
Related data and the analyzed file have been attached and sent.
References
Bulun SE (2016) Reproduction. In: Melmed S, Polonsky KS, Larsen PR, Kronenberg H (eds) Williams textbook of endocrinology, 13th edn. Elsevier, Philadelphia, pp 627–637
Shay CM, Van Horn L, Stamler J, Dyer AR, Brown IJ, Chan Q, Miura K, Zhao L, Okuda N, Daviglus ML (2012) Food and nutrient intakes and their associations with lower BMI in middle-aged US adults: the International Study of Macro-/Micronutrients and Blood Pressure (INTERMAP). Am J Clin Nutr 96:483–491. https://doi.org/10.3945/ajcn.111.025056
De Lourdes LM, Cruz T, Rodrigues LE, Bomfim O, Melo J, Correia R, Porto M, Cedro A, Vicente E (2009) Serum and intracellular magnesium deficiency in patients with metabolic syndrome—evidences for its relation to insulin resistance. Diabetes Res Clin Pract 83:257–262. https://doi.org/10.1016/j.diabres.2008.11.019
Barbagallo M, Dominguez LJ, Galioto A, Ferlisi A, Cani C, Malfa L, Pineo A, Paolisso G (2003) Role of magnesium in insulin action, diabetes and cardio-metabolic syndrome X. Mol Aspects Med 24:39–52. https://doi.org/10.1016/S0098-2997(02)00090-0
Dibaba DT, Xun P, Song Y, Rosanoff A, Shechter M, He K (2017) The effect of magnesium supplementation on blood pressure in individuals with insulin resistance, prediabetes, or noncommunicable chronic diseases: a meta-analysis of randomized controlled trials. Am J Clin Nutr 106:921–929. https://doi.org/10.3945/ajcn.117.155291
Song Y, Manson JE, Buring JE, Liu S (2004) Dietary magnesium intake in relation to plasma insulin levels and risk of type 2 diabetes in women. Diabetes Care 27:59–65. https://doi.org/10.2337/diacare.27.1.59
Hruby A, Guasch-Ferré M, Bhupathiraju SN, Manson JE, Willett WC, Mckeown NM, Hu FB (2017) Magnesium intake, quality of carbohydrates, and risk of type 2 diabetes: results from three US cohorts. Diabetes Care 40:1695–1702. https://doi.org/10.2337/dc17-1143
Bringhurst FR, Demay MB, Kronenberg H (2018) Bone and mineral metabolism in health and disease. In: Jameson JL, Fauci AS, Kasper DL, Hauser SL, Longo DL, Loscalzo J (eds) Harrison’s principles of internal medicine, 2, 20th ed. Mc Graw Hill Education, p 2909–2921
Rajeswari G, Gopal PS, Veerabhadrudu B, Suresh E (2016) Study of magnesium levels in polycystic ovarian syndrome. Int J Appl Res 2:610–613. https://doi.org/10.7439/ijasr
Chakraborty P, Ghosh S, Goswami S, Kabir SN, Chakravarty B, Jana K (2013) Altered trace mineral milieu might play an aetiological role in the pathogenesis of polycystic ovary syndrome. Biol Trace Elem Res 152:9–15. https://doi.org/10.1007/s12011-012-9592-5
Kauffman RP, Tullar PE, Nipp RD, Castracane VD (2011) Serum magnesium concentrations and metabolic variables in polycystic ovary syndrome. Acta Obstet Gynecol Scand 90:452–458. https://doi.org/10.1111/j.1600-0412.2010.01067.x
Sharifi F, Mazloomi S, Hajihosseini R, Mazloomzadeh S (2012) Serum magnesium concentrations in polycystic ovary syndrome and its association with insulin resistance. Gynecol Endocrinol 28:7–11. https://doi.org/10.3109/09513590.2011.579663
Li M, Tang Y, Lin C, Huang Q, Lei D, Hu Y (2017) Serum macroelement and microelement concentrations in patients with polycystic ovary syndrome: a cross-sectional study. Biol Trace Elem Res 176:73–80. https://doi.org/10.1007/s12011-016-0782-4
Kurdoglu Z, Kurdoglu M, Demir H, Sahin H (2012) Serum trace elements and heavy metals in polycystic ovary syndrome. Human Exp Toxicol 31:452–456. https://doi.org/10.1177/0960327111424299
Yin J, Hong X, Ma J, Bu Y, Liu RJFIE (2020) Serum trace elements in patients with polycystic ovary syndrome: a systematic review and meta-analysis. Front Endocrinol 11:572384. https://doi.org/10.3389/fendo.2020.572384
Babapour M, Mohammadi H, Kazemi M, Hadi A, Rezazadegan M, Askari GJBTER (2021) Associations between serum magnesium concentrations and polycystic ovary syndrome status: a systematic review and meta-analysis. Biol Trace Elem Res 199:1297–1305. https://doi.org/10.1007/s12011-020-02275-9
He J, Klag MJ, Whelton PK, Chen J-Y, Mo J-P, Qian M-C, Coresh J, Mo P-S, He GQ (1993) Agreement between overnight and 24-hour urinary cation excretions in southern Chinese men. Am J Epidemiol 137:1212–1220. https://doi.org/10.1093/oxfordjournals.aje.a116623
Zahra H, Berriche O, Mizouri R, Boukhayatia F, Khiari M, Gamoudi A, Lahmar I, Ben Amor N, Mahjoub F, Zayet S, Jamoussi H (2021) Plasmatic magnesium deficiency in 101 outpatients living with type 2 diabetes mellitus. Clin Pract 11:791–800. https://doi.org/10.3390/clinpract11040095
Bringhurst FR, Demay MB, Kronenberg H (2016) Mineral metabolism. In: Melmed S, Polonsky KS, Larsen PR, Kronenberg H (eds) Williams textbook of endocrinology, 13th edn. Elsevier, Philadelphia, pp 1307–1310
Al-Ghamdi SM, Cameron EC, Sutton RA (1994) Magnesium deficiency: pathophysiologic and clinical overview. Am J Kidney Dis 24:737–752. https://doi.org/10.1016/s0272-6386(12)80667-6
Acknowledgements
We appreciate the Vice-chancellor for Research and Technology of Faculty of Medicine, Mashhad University of Medical Sciences, for support in every single step of this study. We also thank the personnel of the Milad Infertility Clinic for their kindness and support: Nayereh Khadem (Professor of Obstetrics and Gynecology, Department of Obstetrics and Gynecology, School of Medicine, Head of Milad Infertility Clinic, Mashhad University of Medical Sciences), Maliheh Mahmoudinia (Assistant Professor of Obstetrics and Gynecology, Department of Obstetrics and Gynecology, School of Medicine, Milad Infertility Clinic, Mashhad University of Medical Sciences).
Funding
This study was supported by a grant from the Vice-chancellor for Research and Technology of Faculty of Medicine, Mashhad University of Medical Sciences, Mashhad, Iran.
Author information
Authors and Affiliations
Contributions
SA, MM, MDM, and MN contributed to the conception and design. SA, MM, SHMV, MA, and NJ contributed to sampling and clinical part of the study. SA and MDM contributed to statistical analysis. SA drafted the manuscript. MM supervised the study.
Corresponding author
Ethics declarations
Ethics Approval
This was a cross-sectional study registered on https://research.mums.ac.ir/general/homePage.action website on 10/23/2019 with research code 980229. Ethical code is IR.MUMS.MEDICAL.REC.1398.541, registered on 8/13/2019 and accessible on https://ethics.research.ac.ir/ which is national ethics committee’s website. This study has followed the Declaration of Helsinki guideline, and informed consent was given by all participants.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Abbasi, S., Mohebbi, M., Mousavi Vahed, S.H. et al. Comparison of Magnesium Status Using 24-h Urine Magnesium Content and Magnesium Fraction Excretion in PCOS with Non-PCOS Control Women: a Cross-sectional Study. Biol Trace Elem Res 201, 5601–5606 (2023). https://doi.org/10.1007/s12011-023-03626-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-023-03626-y