Abstract
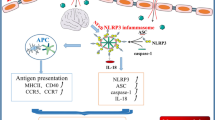
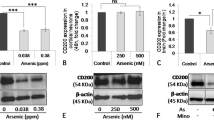
Arsenic exerts neurotoxicity and immunomodulatory effects. Studies have shown that the nervous system is not considered to be an immune-privileged site. However, the effect of arsenic-induced neuroimmune toxicity has rarely been reported. We aimed to investigate the toxic effects of arsenic on the NOD-like receptor family pyrin domain containing 3 (NLRP3) inflammasome and the Th1/Th2/Th17/Treg balance in the brain tissue of mice. Mice were exposed to NaAsO2 (0, 2.5, 5, and 10 mg/kg) for 24 h. Our results showed that 10 mg/kg arsenic exposure significantly decreased brain and hippocampal indices (p < 0.05). The mRNA and protein levels of the blood‒brain barrier (BBB) tight junction protein occludin were decreased in the 5 and 10 mg/kg arsenic-treated groups. Compared with those in the control group, NLRP3 protein levels in 10 mg/kg arsenic-treated mice, caspase-1 protein levels in 2.5, 5, and 10 mg/kg arsenic-treated mice, and IL-1β protein levels in 5 and 10 mg/kg arsenic-treated mice were increased in the hippocampus (p < 0.05). In addition, arsenic induced a hippocampal inflammatory response by upregulating the mRNA levels of the proinflammatory factors IL-6 and TNF-α and downregulating the mRNA level of the anti-inflammatory factor IL-10. Moreover, arsenic decreased the mRNA levels of the Th1 and Th2 transcription factors T-bet and GATA3 and the cytokines IFN-γ and IL-4 and increased the mRNA levels of the Th17 transcription factor RORγt and the cytokine IL-22 (p < 0.05). Collectively, our study demonstrated that arsenic could induce immune-inflammatory responses by regulating the NLRP3 inflammasome and CD4+ T lymphocyte differentiation. These results provide a novel strategy to block the arsenic-induced impairment of neuroimmune responses.




Similar content being viewed by others
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Wang X, Nie Y, Si B, Wang T, Hei TK, Du H, Zhao G, Chen S, Xu A, Liu Y (2021) Silver nanoparticles protect against arsenic induced genotoxicity via attenuating arsenic bioaccumulation and elevating antioxidation in mammalian cells. J Hazard Mater 413:125287. https://doi.org/10.1016/j.jhazmat.2021.125287
Wu S, Rao G, Wang R, Pang Q, Zhang X, Huang R, Li T, Tang Z, Hu L (2021) The neuroprotective effect of curcumin against ATO triggered neurotoxicity through Nrf2 and NF-κB signaling pathway in the brain of ducks. Ecotoxicol Environ Saf 228:112965. https://doi.org/10.1016/j.ecoenv.2021.112965
Wang B, Liu J, Liu B, Liu X, Yu X (2018) Prenatal exposure to arsenic and neurobehavioral development of newborns in China. Environ Int 121(Pt 1):421–427. https://doi.org/10.1016/j.envint.2018.09.031
Freire C, Amaya E, Gil F, Fernández MF, Murcia M, Llop S, Andiarena A, Aurrekoetxea J, Bustamante M, Guxens M, Ezama E, Fernández-Tardón G, Olea N, INMA Project (2018) Prenatal co-exposure to neurotoxic metals and neurodevelopment in preschool children: The Environment and Childhood (INMA) Project. Sci Total Environ 621:340–351. https://doi.org/10.1016/j.scitotenv.2017.11.273
Zhou H, Zhao W, Ye L, Chen Z, Cui Y (2018) Postnatal low-concentration arsenic exposure induces autism-like behavior and affects frontal cortex neurogenesis in rats. Environ Toxicol Pharmacol 62:188–198. https://doi.org/10.1016/j.etap.2018.07.012
Duan X, Xu G, Li J, Yan N, Li X, Liu X, Li B (2022) Arsenic induces continuous inflammation and regulates Th1/Th2/Th17/Treg balance in liver and kidney in vivO. Mediators Inflamm 2022:8414047. https://doi.org/10.1155/2022/8414047
Zhao L, Yang S, Guo Y, Sun G, Li B (2019) Chronic arsenic exposure in drinking water interferes with the balances of T lymphocyte subpopulations as well as stimulates the functions of dendritic cells in vivo. Int Immunopharmacol 71:115–131. https://doi.org/10.1016/j.intimp.2019.03.022
Ikegami A, Wake H (2021) Microglial regulation of blood brain barrier, the neuro-immunological interface. Brain Nerve 73(8):913–919. https://doi.org/10.11477/mf.1416201861
Firdaus F, Zafeer MF, Anis E, Ahmad M, Afzal M (2018) Ellagic acid attenuates arsenic induced neuro-inflammation and mitochondrial dysfunction associated apoptosis. Toxicol Rep 5:411–417. https://doi.org/10.1016/j.toxrep.2018.02.017
Cervellati C, Trentini A, Pecorelli A, Valacchi G (2020) Inflammation in neurological disorders: the thin boundary between brain and periphery. Antioxid Redox Signal 33(3):191–210. https://doi.org/10.1089/ars.2020.8076
He XF, Li LL, Xian WB, Li MY, Zhang LY, Xu JH, Pei Z, Zheng HQ, Hu XQ (2021) Chronic colitis exacerbates NLRP3-dependent neuroinflammation and cognitive impairment in middle-aged brain. J Neuroinflammation 18(1):153. https://doi.org/10.1186/s12974-021-02199-8
Madhu LN, Kodali M, Attaluri S, Shuai B, Melissari L, Rao X, Shetty AK (2021) Melatonin improves brain function in a model of chronic Gulf War Illness with modulation of oxidative stress, NLRP3 inflammasomes, and BDNF-ERK-CREB pathway in the hippocampus. Redox Biol 43:101973. https://doi.org/10.1016/j.redox.2021.101973
Zuniga EI, Macal M, Lewis GM, Harker JA (2015) Innate and adaptive immune regulation during chronic viral infections. Ann Rev Virol 2(1):573–597. https://doi.org/10.1146/annurev-virology-100114-055226
Yin X, Chen S, Eisenbarth SC (2021) Dendritic cell regulation of T helper cells. Annu Rev Immunol 39:759–790. https://doi.org/10.1146/annurev-immunol-101819-025146
Zhang Y, Liu M, Sun H, Zhang YKY, Liu M, Sun H, Yin K (2015) Matrine improves cognitive impairment and modulates the balance of Th17/Treg cytokines in a rat model of Aβ1-42-induced Alzheimer’s disease. Cent Eur J Immunol 40(4):411–419. https://doi.org/10.5114/ceji.2015.56961
Duan X, Gao S, Li J, Wu L, Zhang Y, Li W, Zhao L, Chen J, Yang S, Sun G, Li B (2017) Acute arsenic exposure induces inflammatory responses and CD4+ T cell subpopulations differentiation in spleen and thymus with the involvement of MAPK, NF-kB, and Nrf2. Mol Immunol 81:160–172. https://doi.org/10.1016/j.molimm.2016.12.005
Mondal P, Shaw P, DeyBhowmik A, Bandyopadhyay A, Sudarshan M, Chakraborty A, Chattopadhyay A (2021) Combined effect of arsenic and fluoride at environmentally relevant concentrations in zebrafish (Danio rerio) brain: alterations in stress marker and apoptotic gene expression. Chemosphere 269:128678. https://doi.org/10.1016/j.chemosphere.2020.128678
Firdaus F, Zafeer MF, Ahmad M, Afzal M (2018) Anxiolytic and anti-inflammatory role of thymoquinone in arsenic-induced hippocampal toxicity in Wistar rats. Heliyon 20 4(6):e00650. https://doi.org/10.1016/j.heliyon.2018.e00650
Escudero-Lourdes C, Uresti-Rivera EE, Oliva-González C, Torres-Ramos MA, Aguirre-Bañuelos P, Gandolfi AJ (2016) Cortical astrocytes acutely exposed to the monomethylarsonous acid (MMAIII) show increased pro-inflammatory cytokines gene expression that is consistent with APP and BACE-1: over-expression. Neurochem Res 41(10):2559–2572. https://doi.org/10.1007/s11064-016-1968-z
Mao J, Yang J, Zhang Y, Li T, Wang C, Xu L, Hu Q, Wang X, Jiang S, Nie X, Chen G (2016) Arsenic trioxide mediates HAPI microglia inflammatory response and subsequent neuron apoptosis through p38/JNK MAPK/STAT3 pathway. Toxicol Appl Pharmacol 303:79–89. https://doi.org/10.1016/j.taap.2016.05.003
Bortolotti P, Faure E, Kipnis E (2018) Inflammasomes in tissue damages and immune disorders after trauma. Front Immunol 9:1900. https://doi.org/10.3389/fimmu.2018.01900
Yang Y, Wang Q, Wang W, Wei S, Zeng Q, Zhang A (2020) Semaphorin 4A antibody alleviates arsenic-induced hepatotoxicity in mice via inhibition of AKT2/NF-κB inflammatory signaling. Toxicol Appl Pharmacol 1(410):115364. https://doi.org/10.1016/j.taap.2020.115364
Huang R, Hou L, Zhai X, Ruan Z, Sun W, Zhang D, Zhao X, Wang Q (2021) 2,5-hexanedione induces NLRP3 inflammasome activation and neurotoxicity through NADPH oxidase-dependent pathway. Free Radic Biol Med 162:561–570. https://doi.org/10.1016/j.freeradbiomed.2020.11.013
Daneman R, Prat A (2015) The blood-brain barrier. Cold Spring Harb Perspect Biol 7(1):a020412. https://doi.org/10.1101/cshperspect.a020412
Kim S, Kim GH (2017) Roles of claudin-2, ZO-1 and occludin in leaky HK-2 cells. PLoS One 12(12):e0189221. https://doi.org/10.1371/journal.pone.0189221
Manthari RK, Tikka C, Ommati MM, Niu R, Sun Z, Wang J, Zhang J, Wang J (2018) Arsenic induces autophagy in developmental mouse cerebral cortex and hippocampus by inhibiting PI3K/Akt/mTOR signaling pathway: involvement of blood-brain barrier’s tight junction proteins. Arch Toxicol 92(11):3255–3275. https://doi.org/10.1007/s00204-018-2304-y
Wang Y, Zhao H, Liu Y, Guo M, Tian Y, Huang P, Xing M (2021) Arsenite induce neurotoxicity of common carp: involvement of blood brain barrier, apoptosis and autophagy, and subsequently relieved by zinc (II) supplementation. Aquat Toxicol 232:105765. https://doi.org/10.1016/j.aquatox.2021.105765
Huang Y, Chen S, Luo Y, Han Z (2020) Crosstalk between inflammation and the BBB in stroke. Curr Neuropharmacol 18(12):1227–1236. https://doi.org/10.2174/1570159X18666200620230321
DiStasio MM, Nagakura I, Nadler MJ, Anderson MP (2019) T lymphocytes and cytotoxic astrocyte blebs correlate across autism brains. Ann Neurol 86(6):885–898. https://doi.org/10.1002/ana.25610
Shin JS, Kim I, Moon JS, Ho CC, Choi MS, Ghosh S, Lee SK (2021) Intranuclear Delivery of HIF-1α-TMD Alleviates EAE via Functional Conversion of TH17 Cells. Front Immunol 12:741938. https://doi.org/10.3389/fimmu.2021.741938
Xiao Z, Juan L, Song Y, Zhijian Z, Jing J, Kun Y, Yuna H, Dongfa D, Lili D, Liuxin T, Fei L, Nan L, Fang Y, Yuying S, Yongzhi X (2015) Evaluation of humoral and cellular immune responses to a DNA vaccine encoding chicken type II collagen for rheumatoid arthritis in normal rats. Hum Vaccin Immunother 11(4):938–945. https://doi.org/10.1080/21645515.2015.1010977
Bakheet SA, Alzahrani MZ, Ansari MA, Nadeem A, Zoheir KMA, Attia SM, Al-Ayadhi LY, Ahmad SF (2017) Resveratrol ameliorates dysregulation of Th1, Th2, Th17, and T regulatory cell-related transcription factor signaling in a BTBR T + tf/J Mouse Model of Autism. Mol Neurobiol 54(7):5201–5212. https://doi.org/10.1007/s12035-016-0066-1
Banerjee N, Nandy S, Kearns JK, Bandyopadhyay AK, Das JK, Majumder P, Basu S, Banerjee S, Sau TJ, States JC, Giri AK (2011) Polymorphisms in the TNF-α and IL10 gene promoters and risk of arsenic-induced skin lesions and other nondermatological health effects. Toxicol Sci 121(1):132–139. https://doi.org/10.1093/toxsci/kfr046
Lee GR (2018) The balance of Th17 versus Treg cells in autoimmunity. Int J Mol Sci 19(3):730. https://doi.org/10.3390/ijms19030730
Huang Y, Xiao M, Ye Z, Han Z, Reinach PS, Cheng J (2020) 2-(-2-Benzofuranyl)-2-imidazoline reciprocally regulates Th17/Treg balance induced by ischemic stroke in rats. Pharmazie 75(11):586–589. https://doi.org/10.1691/ph.2020.0634
Funding
This work was supported by grants from the National Natural Science Foundation of China (NSFC) (no. 81803180 and no. 81803200), the Basic Research Project of Liaoning Provincial Department of Education Youth Project (no. LJKQZ2021173), Shenyang Middleyounger Scientific and Technological Innovation Support Plan (no. RC190479 and no. RC200238), Liaoning Province Key Research and Development Program Guidance Plan (no. 2019JH8/10300047), Shenyang Science and Technology Plan Project Social Governance Science and Technology Special Project (no. 21–108-9–11), and Science and Technology Innovation Fund of Shenyang Medical College (no. Y20210513).
Author information
Authors and Affiliations
Contributions
Conceptualization, funding acquisition, data curation, formal analysis, visualization, and writing—original draft, H.J.; software, data curation, and validation, R.H.F.; formal analysis and software, N.Y.; investigation, Z.L.; methodology, Q.W., K.J.X., X.K.H.; resources, validation, writing—review, supervision and editing, L.F.Z.; funding acquisition, writing—review and editing, supervision, project administration, X.X.D.
Corresponding authors
Ethics declarations
Ethics Approval
Animal use has been approved by the Animal Use and Care Committee at Shenyang Medical College (protocol number: SYYXY2020031201).
Consent for Publication
All authors have read and agreed to the published version of the manuscript.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jing, H., Yan, N., Fan, R. et al. Arsenic Activates the NLRP3 Inflammasome and Disturbs the Th1/Th2/Th17/Treg Balance in the Hippocampus in Mice. Biol Trace Elem Res 201, 3395–3403 (2023). https://doi.org/10.1007/s12011-022-03421-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-022-03421-1