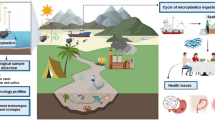
Abstract
Environmental pollutants and especially metal trace elements remain an unmitigated threat to the overall life support system. Their chemical stability and accumulation pattern in the ecosystem make them a persistent hazard. This study aims to characterize the early cadmium (Cd) histological and hematological alterations and their corresponding plasma indicators in the Mediterranean sea bass (Dicentrarchus labrax). We also assessed garlic potential to prevent cadmium toxicity. For this purpose, 200 fish of 55 g mean weight were separated into 3 cylindroconical fiberglass tanks of 500-L capacity, each with a stocking density of 4 kg m−3. The fish were regularly hand-fed 0% (control group), 2%, and 6% garlic-supplemented diets to apparent satiation twice a day for 1 month. At the end of the experiment, we injected 22.2 mM cadmium (CdCl2) intraperitoneally to the experimental groups and a placebo solution (9% NaCl) to the control groups; liver, kidney, heart, and blood tissue alterations were monitored with a full screening of their plasmatic indicators, 24 h before and 48 h after Cd injection. Subsequently, whole blood count and blood smears were performed to follow up on Cd-induced vascular damages. Our data showed that Cd induced thrombotic thrombocytopenic purpura, leading to widespread bleeding and cellular alterations in the targeted tissues. These alterations were associated with an obvious normochromic normocytic anemia in favor of microangiopathic hemolytic anemia. Cd injection has also seriously inhibited the overall enzymatic activities triggering a metabolic shift. Although garlic supplementation had little effect on cadmium-induced alterations, it significantly reduced biomass dispersion. Our data is the first evidence of the cadmium versatile toxicity involving vascular alterations as a central and a leading cause of the overall parenchymal lesions. Cd toxicity was associated with a specific enzymatic signature, which must be considered during the interpretation.
Graphical abstract






Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author on request.
References
Yuan Z, Luo T, Liu X, Hua H, Zhuang Y, Zhang X, Zhang L, Zhang Y, Xu W, Ren J (2019) Tracing anthropogenic cadmium emissions: from sources to pollution. Sci Total Environ 676:87–96. https://doi.org/10.1016/j.scitotenv.2019.04.250
Khan MZH, Hasan MR, Khan M, Aktar S, Fatema K (2017) Distribution of heavy metals in surface sediments of the Bay of Bengal Coast. J Toxicol 2017:1–7. https://doi.org/10.1155/2017/9235764
Zhang H, Reynolds M (2019) Cadmium exposure in living organisms: a short review. Sci Total Environ 678:761–767. https://doi.org/10.1016/j.scitotenv.2019.04.395
Fatoki O, Mathabatha S (2011) An assessment of heavy metal pollution in the East London and Port Elizabeth harbours | Fatoki | Water SA
Zohra BS, Habib A (2016) Assessment of heavy metal contamination levels and toxicity in sediments and fishes from the Mediterranean Sea (southern coast of Sfax, Tunisia). Environ Sci Pollut Res 23:13954–13963. https://doi.org/10.1007/s11356-016-6534-3
Macken A, Giltrap M, Ryall K, Foley B, McGovern E, McHugh B, Davoren M (2009) A test battery approach to the ecotoxicological evaluation of cadmium and copper employing a battery of marine bioassays. Ecotoxicology 18:470–480. https://doi.org/10.1007/s10646-009-0305-6
Tan WH, Tair R, Ali SAM et al (2016) Distribution of heavy metals in seawater and surface sediment in coastal area of Tuaran, Sabah. Trans Sci Technol 3:114–122
Raj VM, Thirunavukkarasu AR, Kailasam M et al (2013) Acute toxicity bioassays of cadmium and mercury on the juveniles of Asian seabass Lates calcarifer (Bloch). Indian J Sci Technol 6:4329–4335
Aly FM, Kotb AM, Haridy MAM, Hammad S (2018) Impacts of fullerene C60 and virgin olive oil on cadmium-induced genotoxicity in rats. Sci Total Environ 630:750–756. https://doi.org/10.1016/j.scitotenv.2018.02.205
Celik A, Büyükakilli B, Cimen B et al (2009) Assessment of cadmium genotoxicity in peripheral blood and bone marrow tissues of male Wistar rats. Toxicol Mech Methods 19:135–140. https://doi.org/10.1080/15376510802354979
Nemmiche S (2017) Oxidative signaling response to cadmium exposure. Toxicol Sci kfw222. https://doi.org/10.1093/toxsci/kfw222
Yu C, Yang C, Song X, Li J, Peng H, Qiu M, Yang L, du H, Jiang X, Liu Y (2020) Long non-coding RNA expression profile in broiler liver with cadmium-induced oxidative damage. Biol Trace Elem Res. https://doi.org/10.1007/s12011-020-02436-w
Salazar-Lugo R, Vargas A, Rojas L, Lemus M (2013) Histopathological changes in the head kidney induced by cadmium in a neotropical fish Colossoma macropomum. Open Vet J 3:145–150
Vazzana M, Celi M, Vazzana M, et al (2009) Effect of in vitro exposure to cadmium and copper on sea bass blood cells. 884–887. https://doi.org/10.4081/ijas.2009.s2.884
Vazzana M, Celi M, Tramati C, Ferrantelli V, Arizza V, Parrinello N (2014) In vitro effect of cadmium and copper on separated blood leukocytes of Dicentrarchus labrax. Ecotoxicol Environ Saf 102:113–120. https://doi.org/10.1016/j.ecoenv.2014.01.018
Annabi A, Kessabi K, Kerkeni A, Said K, Messaoudi I (2012) Influence of cadmium exposure on growth and fecundity of freshwater Mosquitofish Gambusia affinis: in situ and in vivo studies. Biol Trace Elem Res 148:345–355
Ben Ameur W, de Lapuente J, El Megdiche Y et al (2012) Oxidative stress, genotoxicity and histopathology biomarker responses in mullet (Mugil cephalus) and sea bass (Dicentrarchus labrax) liver from Bizerte Lagoon (Tunisia). Mar Pollut Bull 64:241–251. https://doi.org/10.1016/j.marpolbul.2011.11.026
Hyder O, Chung M, Cosgrove D, Herman JM, Li Z, Firoozmand A, Gurakar A, Koteish A, Pawlik TM (2013) Cadmium exposure and liver disease among US adults. J Gastrointest Surg 17:1265–1273. https://doi.org/10.1007/s11605-013-2210-9
Messaoudi I, Hammouda F, El Heni J et al (2010) Reversal of cadmium-induced oxidative stress in rat erythrocytes by selenium, zinc or their combination. Exp Toxicol Pathol 62:281–288. https://doi.org/10.1016/j.etp.2009.04.004
Romeo M, Mathieu A, Gnassia-Barelli M et al (1994) Heavy metal content and biotransformation enzymes in two fish species from the NW Mediterranean. Mar Ecol Prog Ser 107:15–22
Tinkov AA, Filippini T, Ajsuvakova OP, Aaseth J, Gluhcheva YG, Ivanova JM, Bjørklund G, Skalnaya MG, Gatiatulina ER, Popova EV, Nemereshina ON, Vinceti M, Skalny AV (2017) The role of cadmium in obesity and diabetes. Sci Total Environ 601–602:741–755. https://doi.org/10.1016/j.scitotenv.2017.05.224
Thongra-ar W, Parkpian P, Tang A (2003) Toxicity of mercury to growth and survival of seabass larvae, Lates calcarifer and the modifying effects of salinity. ScienceAsia 29:209. https://doi.org/10.2306/scienceasia1513-1874.2003.29.209
Nishijo M, Nakagawa H, Suwazono Y, Nogawa K, Kido T (2017) Causes of death in patients with Itai-itai disease suffering from severe chronic cadmium poisoning: a nested case-control analysis of a follow-up study in Japan. BMJ Open 7:1–7. https://doi.org/10.1136/bmjopen-2016-015694
Banni M, Chouchene L, Said K (2011) Mechanisms underlying the protective effect of zinc and selenium against cadmium-induced oxidative stress in zebrafish Danio rerio. 981–992. https://doi.org/10.1007/s10534-011-9456-z
De Smet H, Blust R (2001) Stress responses and changes in protein metabolism in carp Cyprinus carpio during cadmium exposure. Ecotoxicol Environ Saf 48:255–262. https://doi.org/10.1006/eesa.2000.2011
Pereira LS, Ribas JLC, Vicari T, Silva SB, Stival J, Baldan AP, Valdez Domingos FX, Grassi MT, Cestari MM, Silva de Assis HC (2016) Effects of ecologically relevant concentrations of cadmium in a freshwater fish. Ecotoxicol Environ Saf 130:29–36. https://doi.org/10.1016/j.ecoenv.2016.03.046
Arroyo VS, Flores KM, Ortiz LB et al (2013) Liver and cadmium toxicity. J Drug Metab Toxicol 03. https://doi.org/10.4172/2157-7609.S5-001
Gill TS, Tewari H, Pande J (1991) In vivo and in vitro effects of cadmium on selected enzymes in different organs of the fish Barbus conchonius Ham.(rosy barb). Comp Biochem Physiol Part C Comp Pharmacol 100:501–505
Al-asgah NA (2015) Haematological and biochemical parameters and tissue accumulations of cadmium in Oreochromis niloticus exposed to various concentrations of cadmium chloride. Saudi J Biol Sci 22:543–550. https://doi.org/10.1016/j.sjbs.2015.01.002
Javed M, Ahmad I, Ahmad A, Usmani N, Ahmad M (2016) Studies on the alterations in haematological indices, micronuclei induction and pathological marker enzyme activities in Channa punctatus (spotted snakehead) perciformes, channidae exposed to thermal power plant effluent. Springerplus. https://doi.org/10.1186/s40064-016-2478-9, 5, 761
McGill MR (2016) The past and present of serum aminotransferases and the future of liver injury biomarkers. EXCLI J 15:817–828. https://doi.org/10.17179/excli2016-800
Guo JJ, Kuo CM, Chuang YC, Hong JW, Chou RL, Chen TI (2012) The effects of garlic-supplemented diets on antibacterial activity against Streptococcus iniae and on growth in orange-spotted grouper, Epinephelus coioides. Aquaculture 364–365:33–38. https://doi.org/10.1016/j.aquaculture.2012.07.023
Moore PK (2013) EP03 therapeutic outlook for hydrogen sulfide donors. Nitric Oxide 31:S18–S19. https://doi.org/10.1016/j.niox.2013.06.029
Talpur AD, Ikhwanuddin M (2012) Dietary effects of garlic (Allium sativum) on haemato-immunological parameters, survival, growth, and disease resistance against Vibrio harveyi infection in Asian sea bass, Lates calcarifer (Bloch). Aquaculture 364–365:6–12. https://doi.org/10.1016/j.aquaculture.2012.07.035
Militz TA, Southgate PC, Carton AG, Hutson KS (2013) Dietary supplementation of garlic (Allium sativum) to prevent monogenean infection in aquaculture. Aquaculture 408–409:95–99. https://doi.org/10.1016/j.aquaculture.2013.05.027
Yildiz HY, Van QP, Parisi G, Sao MD (2019) Anti-parasitic activity of garlic (Allium sativum) and onion (Allium cepa) juice against crustacean parasite, Lernantropus kroyeri, found on European sea bass (Dicentrarchus labrax). Ital J Anim Sci 18:833–837
Hosseini A, Hosseinzadeh H (2015) A review on the effects of Allium sativum (garlic) in metabolic syndrome. J Endocrinol Investig 38:1147–1157. https://doi.org/10.1007/s40618-015-0313-8
Liu H, Mao P, Wang J, Wang T, Xie CH (2015) Allicin protects PC12 cells against 6-OHDA-induced oxidative stress and mitochondrial dysfunction via regulating mitochondrial dynamics. Cell Physiol Biochem 36:966–979. https://doi.org/10.1159/000430271
Liu S-G, Ren P-Y, Wang G-Y, Yao SX, He XJ (2014) Allicin protects spinal cord neurons from glutamate-induced oxidative stress through regulating the heat shock protein 70/inducible nitric oxide synthase pathway. Food Funct 6:320–329. https://doi.org/10.1039/C4FO00761A
Lanzotti V (2006) The analysis of onion and garlic. 1112:3–22. https://doi.org/10.1016/j.chroma.2005.12.016
Liang D, Wang C, Tocmo R, Wu H, Deng LW, Huang D (2015) Hydrogen sulphide (H2S) releasing capacity of essential oils isolated from organosulphur rich fruits and vegetables. J Funct Foods 14:634–640. https://doi.org/10.1016/j.jff.2015.02.007
Guan MY, Zhang HH, Pan W, Jin CW, Lin XY (2018) Sulfide alleviates cadmium toxicity in Arabidopsis plants by altering the chemical form and the subcellular distribution of cadmium. Sci Total Environ 627:663–670. https://doi.org/10.1016/j.scitotenv.2018.01.245
Manera M, Sayyaf Dezfuli B, DePasquale JA, Giari L (2016) Multivariate approach to gill pathology in European sea bass after experimental exposure to cadmium and terbuthylazine. Ecotoxicol Environ Saf 129:282–290. https://doi.org/10.1016/j.ecoenv.2016.03.039
Heydarnejad MS, Khosravian-hemamai M, Nematollahi A (2013) Effects of cadmium at sub-lethal concentration on growth and biochemical parameters in rainbow trout (Oncorhynchus mykiss). Vet J 66:1–7
Chen S, Shen X, Cheng S, Li P, du J, Chang Y, Meng H (2013) Evaluation of garlic cultivars for polyphenolic content and antioxidant properties 8:. https://doi.org/10.1371/journal.pone.0079730
Amiard JC, Pineau A, Boiteau HL, Metayer C, Amiard-Triquet C (1987) Application de la spectrometrie d’absorption atomique zeeman aux dosages de huit elements traces (Ag, Cd, Cr, Cu, Mn, Ni, Pb et Se) dans des matrices biologiques solides. Water Res 21:693–697. https://doi.org/10.1016/0043-1354(87)90081-9
Mosbah A, Guerbej H, Boussetta H, Bouraoui Z, Banni M (2018) Protective effects of dietary garlic powder against cadmium-induced toxicity in sea bass liver: a chemical, biochemical, and transcriptomic approach. Biol Trace Elem Res 183:370–378. https://doi.org/10.1007/s12011-017-1146-4
Kilkenny C, Browne WJ, Cuthill IC et al (2010) Animal research: reporting in vivo experiments. Anim Res 3
Rosenfeld A, Dial S (2010) Clinical pathology for the veterinary team
Şahan A, Yılmaz HA, Eroldoğan OT (2017) Determination of hematological status as health indicator of European sea bass (Dicentrarchus labrax) fed different dietary fatty acids 5:445–450
Anonymos (2009) Clinical chemistry reagents product. Thermo Fish, In https://assets.thermofisher.com/TFS-Assets/CDD/Specification-Sheets/D14489~.pdf
Coeurdacier J-L, Dutto G, Gasset E, Blancheton J-P (2011) Is total serum protein a good indicator for welfare in reared sea bass (Dicentrarchus labrax)? Aquat Living Resour 24:121–127. https://doi.org/10.1051/alr/2011130
Froese R (2006) Cube law, condition factor and weight-length relationships: history, meta-analysis and recommendations. J Appl Ichthyol 22:241–253. https://doi.org/10.1111/j.1439-0426.2006.00805.x
Nalawade VB, Bhilave MP (2011) Protein efficiency ratio (PER) and gross food conversion efficiency (GFCE) of freshwater fish Labeo rohita fed on formulated feed. 4
Feys J (2016) Nonparametric tests for the interaction in two-way factorial designs using R. R J 8:367–378. https://doi.org/10.32614/rj-2016-027
Salazar-lugo R (2013) Histopathological changes in the head kidney induced by cadmium in a neotropical fish Colossoma macropomum
Fagerberg B, Bergström G, Borén J, Barregard L (2012) Cadmium exposure is accompanied by increased prevalence and future growth of atherosclerotic plaques in 64-year-old women. J Intern Med 272:601–610. https://doi.org/10.1111/j.1365-2796.2012.02578.x
Rikans LE, Yamano T (2000) Mechanisms of cadmium-mediated acute hepatotoxicity 14:110–117
Bain BJ (2005) Diagnosis from the blood smear. 498–507
Messaoudi I, Hammouda F, Sa K (2010) Reversal of cadmium-induced oxidative stress in rat erythrocytes by selenium, zinc or their combination. 62:281–288. https://doi.org/10.1016/j.etp.2009.04.004
Horiguchi H, Oguma E, Kayama F (2017) Cadmium induces anemia through interdependent progress of hemolysis, body iron accumulation, and insufficient erythropoietin production in rats. 122:198–210. https://doi.org/10.1093/toxsci/kfr100
Cucu D, D’Haese PC, De Beuf A, Verhulst A (2011) Low doses of cadmium chloride and methallothionein-1-bound cadmium display different accumulation kinetics and induce different genes in cells of the human nephron. Nephron Extra 1:24–37. https://doi.org/10.1159/000330069
Sabolic I, Ljubojevic M, Herak-Kramberger CM, Brown D (2002) Cd-MT causes endocytosis of brush-border transporters in rat renal proximal tubules. Am J Physiol Ren Physiol 283:F1389–F1402. https://doi.org/10.1152/ajprenal.00066.2002
McGeer JC, Niyogi S, Scott Smith D (2011) Cadmium. Fish Physiol 31:125–184. https://doi.org/10.1016/S1546-5098(11)31025-4
Heydarnejad MS, Khosravian-Hemamai M, Nematollahi A (2013) Effects of cadmium at sub-lethal concentration on growth and biochemical parameters in rainbow trout (Oncorhynchus mykiss). Ir Vet J 66:11
Al-Asgah NA, Abdel-Warith A-WA, Younis E-SM, Allam HY (2015) Haematological and biochemical parameters and tissue accumulations of cadmium in Oreochromis niloticus exposed to various concentrations of cadmium chloride. Saudi J Biol Sci 22:543–550. https://doi.org/10.1016/j.sjbs.2015.01.002
Alhazza IM (2008) Cadmium-induced hepatotoxicity and oxidative stress in rats: protection by selenium
Dubale MS, Shah P (1981) Biochemical alterations induced by cadmium in the liver of Channa punctatus. Environ Res 26:110–118
El-Naga EHA, El-Moselhy KM, Hamed MA (2005) Toxicity of cadmium and copper and their effect on some biochemical parameters of marine fish Mugil Seheli. Egypt J Aquat Res 31:60–71
Öner M, Atli G, Canli M (2008) Changes in serum biochemical parameters of freshwater fish Oreochromis niloticus following prolonged metal (Ag, Cd, Cr, Cu, Zn) exposures. Environ Toxicol Chem 27:360–366. https://doi.org/10.1897/07-281R.1
Peres H (2013) Selected plasma biochemistry parameters in gilthead seabream (Sparus aurata) juveniles. https://doi.org/10.1111/j.1439-0426.2012.02049.x
Levesque HM, Moon TW, Campbell PGC, Hontela A (2002) Seasonal variation in carbohydrate and lipid metabolism of yellow perch (Perca flavescens) chronically exposed to metals in the field. Aquat Toxicol 60:257–267. https://doi.org/10.1016/S0166-445X(02)00012-7
Wieland M, Levin MK, Hingorani KS, Biro FN, Hingorani MM (2009) Mechanism of cadmium-mediated inhibition of Msh2-Msh6 function in DNA mismatch repair. Biochemistry 48:9492–9502. https://doi.org/10.1021/bi9001248
Almeida JA, Diniz YS, Marques SFG, Faine LA, Ribas BO, Burneiko RC, Novelli ELB (2002) The use of the oxidative stress responses as biomarkers in Nile tilapia (Oreochromis niloticus) exposed to in vivo cadmium contamination. Environ Int 27:673–679
Valvona CJ, Fillmore HL, Nunn PB, Pilkington GJ (2016) The regulation and function of lactate dehydrogenase A: therapeutic potential in brain tumor. Brain Pathol 26:3–17. https://doi.org/10.1111/bpa.12299
Lionetto MG, Giordano ME, Vilella S, Schettino T (2000) Inhibition of eel enzymatic activities by cadmium 48:561–571
Ghaffari MA, Ph D, Abromand M, et al (2008) In vitro inhibition of human sperm creatine kinase by nicotine , Cotinine and Cadmium , as a Mechanism in Smoker Men Infertility. 2:125–130
Muia J, Zhu J, Greco SC, et al (2019) Phylogenetic and functional analysis of ADAMTS13 identifies highly conserved domains essential for allosteric regulation. 133:12–15. https://doi.org/10.1182/blood-2018-11-886275
Ugwuja EI, Erejuwa OO, Ugwu NC (2016) Spices mixture containing garlic, ginger and nutmeg has protective effects on the kidneys and liver of cadmium exposed rats. Tabriz Univ Med Sci 6:271–274. https://doi.org/10.15171/apb.2016.038
Ibrahim NM, Eweis EA, El-Beltagi HS, Abdel-Mobdy YE (2012) Effect of lead acetate toxicity on experimental male albino rat. Asian Pac J Trop Biomed 2:41–46. https://doi.org/10.1016/S2221-1691(11)60187-1
Boonpeng S, Siripongvutikorn S, Sae-wong C, Sutthirak P (2014) The antioxidant and anti-cadmium toxicity properties of garlic extracts. Food Sci Nutr 2:792–801. https://doi.org/10.1002/fsn3.164
Obioha UE, Suru SM, Ola-Mudathir KF, Faremi TY (2009) Hepatoprotective potentials of onion and garlic extracts on cadmium-induced oxidative damage in rats. Biol Trace Elem Res 129:143–156. https://doi.org/10.1007/s12011-008-8276-7
Ereño-Orbea J, Majtan T, Oyenarte I, et al (2013) Structural basis of regulation and oligomerization of human cystathionine β-synthase, the central enzyme of transsulfuration. Proc Natl Acad Sci U S A 110:undefined-undefined. https://doi.org/10.1073/pnas.1313683110
Cheung SH, Lau JYW (2018) Hydrogen sulfide mediates athero-protection against oxidative stress via S-sulfhydration. PLoS One 13:e0194176. https://doi.org/10.1371/journal.pone.0194176
Ferriero R, Nusco E, De Cegli R et al (2018) Pyruvate dehydrogenase complex and lactate dehydrogenase are targets for therapy of acute liver failure. J Hepatol 69:325–335. https://doi.org/10.1016/j.jhep.2018.03.016
XieZhi-Zhong, ShiMei-Mei, XieLi, et al (2014) Sulfhydration of p66Shc at cysteine59 mediates the antioxidant effect of hydrogen sulfide. Antioxid Redox Signal https://doi.org/10.1089/ars.2013.5604, 21, 2531, 2542
Benavides GA, Squadrito GL, Mills RW, Patel HD, Isbell TS, Patel RP, Darley-Usmar VM, Doeller JE, Kraus DW (2007) Hydrogen sulfide mediates the vasoactivity of garlic. Proc Natl Acad Sci 104:17977–17982
Bełtowski J (2015) Hydrogen sulfide in pharmacology and medicine–an update. Pharmacol Reports 67:647–658. https://doi.org/10.1016/j.pharep.2015.01.005
Xie Z-Z, Liu Y, Bian J-S (2016) Hydrogen sulfide and cellular redox homeostasis. Oxidative Med Cell Longev 2016:6043038–6043012. https://doi.org/10.1155/2016/6043038
Torres-Palazzolo C, Ramirez D, Locatelli D, Manucha W, Castro C, Camargo A (2018) Bioaccessibility and permeability of bioactive compounds in raw and cooked garlic. J Food Compos Anal 70:49–53. https://doi.org/10.1016/j.jfca.2018.03.008
Zeb I, Ahmadi N, Flores F, Budoff MJ (2018) Randomized trial evaluating the effect of aged garlic extract with supplements versus placebo on adipose tissue surrogates for coronary atherosclerosis progression. Coron Artery Dis 29:325–328. https://doi.org/10.1097/MCA.0000000000000587
Labrador JRP, Guiares RC, Hontiveros GJS (2016) Effect of garlic powder-supplemented diets on the growth and survival of Pacific white leg shrimp (Litopenaeus vannamei). Cogent Food Agric 2:. https://doi.org/10.1080/23311932.2016.1210066
Metwally MAA (2009) Effects of garlic (Allium sativum) on some antioxidant activities in tilapia nilotica (Oreochromis niloticus). World J fish Mar Sci 1:56–64
Saleh NE, Michael FR, Toutou MM (2015) Evaluation of garlic and onion powder as phyto-additives in the diet of sea bass (Dicentrarcus labrax). Egypt J Aquat Res 41:211–217. https://doi.org/10.1016/j.ejar.2015.03.008
Sh D, Seidgar M, Nekuiefard A et al (2019) Oral administration of garlic powder (Allium sativum) on growth performance and survival rate of Carassius auratus fingerlings. Iran J Fish Sci 18:71–82. https://doi.org/10.22092/ijfs.2018.117478
Yılmaz S, Ergün S (2012) Effects of garlic and ginger oils on hematological and biochemical variables of sea bass Dicentrarchus labrax. J Aquat Anim Health 24:219–224. https://doi.org/10.1080/08997659.2012.711266
Juveniles BD, Irkin LC, Yigit M, et al (2014) Toxicological evaluation of dietary garlic (Allium sativum) powder in European sea bass Dicentrarchuslabrax juveniles. 989–996
Satheeshkumar P, Ananthan G, Kumar DS, Jagadeesan L (2012) Haematology and biochemical parameters of different feeding behaviour of teleost fishes from Vellar estuary, India. Comp Clin Path 21:1187–1191. https://doi.org/10.1007/s00580-011-1259-7
Fazio F, Piccione G (2015) Relationship between blood parameters and biometric indices of Sparus aurata and Dicentrarcus labrax cultured in onshore tanks. https://doi.org/10.1080/10236244.2015.1041239
Hellal MEA, Hellal F, El Khemissi Z et al (2011) Trace metals in algae and sediments from the North-Eastern Tunisian lagoons. Bull Environ Contam Toxicol 86:194–198. https://doi.org/10.1007/s00128-010-0175-x
Acknowledgments
We thank the Tunisia Ministry of Defense and the Tunisian Higher Education and Scientific Research along with Mr. Kathy Newman.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Cadmium toxicity is mediated through endothelial alterations.
• Cadmium induced an overall inhibition in plasma enzymatic activity.
• Garlic supplementation significantly improved growth and feed efficiency.
Supplementary Information
ESM 1
Approximate feed (base) and water compositions (DOC 45 kb)
ESM 2
Garlic phytochemicals assessments, Values represent the mean ± standard error (SE) of 3 replicates (DOCX 14 kb)
ESM 3
Analysis of the liver histological changes before and after Cd injection in different experimental groups. (DOCX 17 kb)
ESM 4
Analysis of the pancreas and the kidney histological changes before and after Cd injection, in different experimental groups (DOCX 17 kb)
ESM 5
Analysis of blood cell and cardiac histological changes before and after cadmium injection (DOCX 14 kb)
ESM 6
Biochemical indicators measured in fish blood. (a) Heat map of plasmatic indicators, and (b) bars plot of alanine aminotransferase (ALT), aspartate aminotransferase (AST); lactate dehydrogenase (LDH); creatine kinase (CK); gamma-glutamyl-transferase (GGT); blood urea noytogen (BUN); creatinine (CRT) and alkaline phosphatase (ALP). Color intensity correlates with the parameter s’ activity, Values represent the mean ± standard error (SE) of at least 10 replicates, * Significant differences using two ways robust ANOVA test (horizontal comparison). (PDF 707 kb)
ESM 7
Biochemical indicators measured in fish blood. (a) Heat map and (b) bars plot of total cholesterol (CHL); triglycerides (TG);total protein (TP);total bilirubin (TB);calcium(Ca);phosphorus (P) and Ca.P ratio. Color intensity correlates with the parameter s’ activity . Values represent the mean ± standard error (SE) of at least 10. * Significant differences using two ways robust ANOVA test (horizontal comparison). (PDF 713 kb)
ESM 8
Fish growth parameters. (SGR) specific growth rate, (K.factor) fulton’s condition factor, (FCR) feed conversion rate; Values represent the mean ± standard error (SE) of at least 10 replicates, a,b: groups with the same letter have significant differences using one way robust ANOVA test with p < 0.05 (horizontal comparison). (DOCX 22 kb)
ESM 9
Fish growth parameters. (SGR) specific growth rate, (K.factor) fulton’s condition factor, (FCR) feed conversion rate; Values represent the mean ± standard error (SE) of at least 10 replicates, a,b: groups with the same letter have significant differences using one way robust ANOVA test with p < 0.05 (horizontal comparison). (PDF 1790 kb)
ESM 10
Metals accumulation profile in liver tissue results published in (Mosbah et al., 2017) (DOCX 17 kb)
Rights and permissions
About this article
Cite this article
Mosbah, A., Dhaouadi, R., Abdeljelil, N.B. et al. Multifactorial Screening Reveals New Insight into Early Cadmium Exposure and Garlic Interactions in Dicentrarchus labrax. Biol Trace Elem Res 199, 4759–4771 (2021). https://doi.org/10.1007/s12011-021-02592-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-021-02592-7