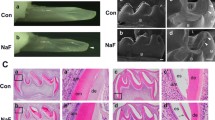
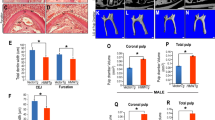
Abstract
Fluoride can alter the formation of mineralized tissues, including enamel, dentin, and bone. Dentin fluorosis occurs in tandem with enamel fluorosis. However, the pathogenesis of dentin fluorosis and its mechanisms are poorly understood. In this study, we report the effects of fluoride on the initiation of dentin matrix formation and odontoblast function. Mice from two enamel fluorosis susceptible strains (A/J and C57BL/6J) were given either 0 or 50 ppm fluoride in drinking water for 4 weeks. In both mouse strains, there was no overall change in dentin thickness, but fluoride treatment resulted in a significant increase in the thickness of the predentin layer. The lightly mineralized layer (LL), which lies at the border between predentin and fully mineralized dentin and is associated with dentin phosphoprotein (DPP), was absent in fluoride exposed mice. Consistent with a possible reduction of DPP, fluoride-treated mice showed reduced immunostaining for dentin sialoprotein (DSP). Fluoride reduced RUNX2, the transcription regulator of dentin sialophosphoprotein (DSPP), that is cleaved to form both DPP and DSP. In fluoride-treated mouse odontoblasts, the effect of fluoride was further seen in the upstream of RUNX2 as the reduced nuclear translocation of β-catenin and phosphorylated p65/NFκB. In vitro, MD10-F2 pre-odontoblast cells showed inhibition of the Dspp mRNA level in the presence of 10 μM fluoride, and qPCR analysis showed a significantly downregulated level of mRNAs for RUNX2, β-catenin, and Wnt10b. These findings indicate that in mice, systemic exposure to excess fluoride resulted in reduced Wnt/β-catenin signaling in differentiating odontoblasts to downregulate DSPP production via RUNX2.








Similar content being viewed by others
References
Fejerskov O, Larsen MJ, Josephsen K, Thylstrup A (1979) Effect of long-term administration of fluoride on plasma fluoride and calcium in relation to forming enamel and dentin in rats. Scand J Dent Res 87(2):98–104
Vieira A, Hancock R, Dumitriu M, Schwartz M, Limeback H, Grynpas M (2005) How does fluoride affect dentin microhardness and mineralization? J Dent Res 84(10):951–957
Kierdorf U, Kierdorf H, Fejerskov O (1993) Fluoride-induced developmental changes in enamel and dentine of European roe deer (Capreolus capreolus L.) as a result of environmental pollution. Arch Oral Biol 38(12):1071–1081
Yaeger JA (1966) The effects of high fluoride diets on developing enamel and dentin in the incisors of rats. Am J Anat 118(2):665–683. https://doi.org/10.1002/aja.1001180219
Fejerskov O, Thylstrup A, Larsen MJ (1977) Clinical and structural features and possible pathogenic mechanisms of dental fluorosis. Scand J Dent Res 85(7):510–534
Fejerskov O, Yaeger JA, Thylstrup A (1979) Microradiography of the effect of acute and chronic administration of fluoride on human and rat dentine and enamel. Arch Oral Biol 24(2):123–130
Rojas-Sanchez F, Alaminos M, Campos A, Rivera H, Sanchez-Quevedo MC (2007) Dentin in severe fluorosis: a quantitative histochemical study. J Dent Res 86(9):857–861
Nelson DG, Coote GE, Vickridge IC, Suckling G (1989) Proton microprobe determination of fluorine profiles in the enamel and dentine of erupting incisors from sheep given low and high daily doses of fluoride. Arch Oral Biol 34(6):419–429
Waidyasekera K, Nikaido T, Weerasinghe D, Watanabe A, Ichinose S, Tay F, Tagami J (2010) Why does fluorosed dentine show a higher susceptibility for caries: an ultra-morphological explanation. J Med Dent Sci 57(1):17–23
Waidyasekera PG, Nikaido T, Weerasinghe DD, Wettasinghe KA, Tagami J (2007) Caries susceptibility of human fluorosed enamel and dentine. J Dent 35(4):343–349. https://doi.org/10.1016/j.jdent.2006.10.008
Milan AM, Waddington RJ, Embery G (1999) Altered phosphorylation of rat dentine phosphoproteins by fluoride in vivo. Calcif Tissue Int 64(3):234–238
Li P, Xue Y, Zhang W, Teng F, Sun Y, Qu T, Chen X, Cheng X, Song B, Luo W, Yu Q (2013) Sodium fluoride induces apoptosis in odontoblasts via a JNK-dependent mechanism. Toxicology 308:138–145. https://doi.org/10.1016/j.tox.2013.03.016
Cvikl B, Lussi A, Carvalho TS, Moritz A, Gruber R (2018) Stannous chloride and stannous fluoride are inhibitors of matrix metalloproteinases. J Dent 78:51–58. https://doi.org/10.1016/j.jdent.2018.08.002
Wurtz T, Houari S, Mauro N, MacDougall M, Peters H, Berdal A (2008) Fluoride at non-toxic dose affects odontoblast gene expression in vitro. Toxicology 249(1):26–34. https://doi.org/10.1016/j.tox.2008.04.013
Houari S, Wurtz T, Ferbus D, Chateau D, Dessombz A, Berdal A, Babajko S (2014) Asporin and the mineralization process in fluoride-treated rats. J Bone Miner Res 29(6):1446–1455. https://doi.org/10.1002/jbmr.2153
Ruch JV, Lesot H, Begue-Kirn C (1995) Odontoblast differentiation. Int J Dev Biol 39(1):51–68
Miyazaki T, Kanatani N, Rokutanda S, Yoshida C, Toyosawa S, Nakamura R, Takada S, Komori T (2008) Inhibition of the terminal differentiation of odontoblasts and their transdifferentiation into osteoblasts in Runx2 transgenic mice. Arch Histol Cytol 71(2):131–146
Yun C-Y, Choi H, You Y-J, Yang J-Y, Baek J-A, Cho E-S (2016) Requirement of Smad4-mediated signaling in odontoblast differentiation and dentin matrix formation. Anat Cell Biol 49(3):199–205
Yan D, Willett TL, Gu XM, Martinez-Mier EA, Sardone L, McShane L, Grynpas M, Everett ET (2011) Phenotypic variation of fluoride responses between inbred strains of mice. Cells Tissues Organs 194(2–4):261–267. https://doi.org/10.1159/000324224
Everett ET (2011) Fluoride’s effects on the formation of teeth and bones, and the influence of genetics. J Dent Res 90(5):552–560. https://doi.org/10.1177/0022034510384626
Kawamoto T (2003) Use of a new adhesive film for the preparation of multi-purpose fresh-frozen sections from hard tissues, whole-animals, insects and plants. Arch Histol Cytol 66(2):123–143
Taves DR (1968) Separation of fluoride by rapid diffusion using hexamethyldisiloxane. Talanta 15:969–974
Bawden JW, Deaton TG, Koch GG, Crawford BP (1989) Effect of an acute maternal fluoride dose on fetal plasma fluoride levels and enamel fluoride uptake in guinea pigs. J Dent Res 68(7):1169–1172
Gomez S, Boyde A (1994) Correlated alkaline phosphatase histochemistry and quantitative backscattered electron imaging in the study of rat incisor ameloblasts and enamel mineralization. Microsc Res Tech 29(1):29–36. https://doi.org/10.1002/jemt.1070290105
Symons NB (1955) Alkaline phosphatase activity in the developing teeth of the rat. J Anat 89(2):238–245
Burstone S (1962) Enzyme histochemistry, and its application in the study of neoplasms. Academic Press
Chen S, Rani S, Wu Y, Unterbrink A, Gu TT, Gluhak-Heinrich J, Chuang H-H, MacDougall M (2005) Differential regulation of dentin sialophosphoprotein expression by Runx2 during odontoblast cytodifferentiation. J Biol Chem 280(33):29717–29727. https://doi.org/10.1074/jbc.M502929200
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Zhang Y, Kim JY, Horst O, Nakano Y, Zhu L, Radlanski RJ, Ho S, Den Besten PK (2014) Fluorosed mouse ameloblasts have increased SATB1 retention and Galphaq activity. PLoS One 9(8):e103994. https://doi.org/10.1371/journal.pone.0103994
Guy WS, Taves DR, Brey WS (1976) Organic Fluorocompounds in human plasma: prevalence and characterization. In: Biochemistry involving carbon-fluorine bonds, vol 28. ACS Symposium Series, vol 28. AMERICAN CHEMICAL SOCIETY, pp 117-134. https://doi.org/10.1021/bk-1976-0028.ch007
Ahmad M, Iseki H, Abduweli D, Baba O, Tabata MJ, Takano Y (2011) Ultrastructural and histochemical evaluation of appositional mineralization of circumpulpal dentin at the crown- and root-analog portions of rat incisors. J Electron Microsc 60(1):79–87. https://doi.org/10.1093/jmicro/dfq075
Goldberg M, Septier D (1996) A comparative study of the transition between predentin and dentin, using various preparative procedures in the rat. Eur J Oral Sci 104(3):269–277
Suzuki M, Shin M, Simmer JP, Bartlett JD (2014) Fluoride affects enamel protein content via TGF-beta1-mediated KLK4 inhibition. J Dent Res 93(10):1022–1027. https://doi.org/10.1177/0022034514545629
Braut A, Kollar EJ, Mina M (2003) Analysis of the odontogenic and osteogenic potentials of dental pulp in vivo using a Col1a1-2.3-GFP transgene. Int J Dev Biol 47(4):281–292
Yamakoshi Y, Hu JC-C, Fukae M, Zhang H, Simmer JP (2005) Dentin glycoprotein: the protein in the middle of the dentin sialophosphoprotein chimera. J Biol Chem 280(17):17472–17479. https://doi.org/10.1074/jbc.M413220200
Sreenath T, Thyagarajan T, Hall B, Longenecker G, D'Souza R, Hong S, Wright JT, MacDougall M, Sauk J, Kulkarni AB (2003) Dentin sialophosphoprotein knockout mouse teeth display widened predentin zone and develop defective dentin mineralization similar to human dentinogenesis imperfecta type III. J Biol Chem 278(27):24874–24880. https://doi.org/10.1074/jbc.M303908200
Suzuki S, Sreenath T, Haruyama N, Honeycutt C, Terse A, Cho A, Kohler T, Muller R, Goldberg M, Kulkarni AB (2009) Dentin sialoprotein and dentin phosphoprotein have distinct roles in dentin mineralization. Matrix Biol 28(4):221–229. https://doi.org/10.1016/j.matbio.2009.03.006
Oh H-J, Lee H-K, Park S-J, Cho Y-S, Bae H-S, Cho M-I, Park J-C (2012) Zinc balance is critical for NFI-C mediated regulation of odontoblast differentiation. J Cell Biochem 113(3):877–887. https://doi.org/10.1002/jcb.23421
Narayanan K, Gajjeraman S, Ramachandran A, Hao J, George A (2006) Dentin matrix protein 1 regulates dentin sialophosphoprotein gene transcription during early odontoblast differentiation. J Biol Chem 281(28):19064–19071. https://doi.org/10.1074/jbc.M600714200
Yang G, Yuan G, MacDougall M, Zhi C, Chen S (2017) BMP-2 induced Dspp transcription is mediated by Dlx3/Osx signaling pathway in odontoblasts. Sci Rep 7(1):10775. https://doi.org/10.1038/s41598-017-10908-8
Chen S, Gluhak-Heinrich J, Wang YH, Wu YM, Chuang HH, Chen L, Yuan GH, Dong J, Gay I, MacDougall M (2009) Runx2, Osx, and Dspp in tooth development. J Dent Res 88(10):904–909. https://doi.org/10.1177/0022034509342873
Lee D-S, Choung H-W, Kim H-J, Gronostajski RM, Yang Y-I, Ryoo H-M, Lee ZH, Kim H-H, Cho E-S, Park J-C (2014) NFI-C regulates osteoblast differentiation via control of osterix expression. Stem Cells 32(9):2467–2479. https://doi.org/10.1002/stem.1733
Gaur T, Lengner CJ, Hovhannisyan H, Bhat RA, Bodine PVN, Komm BS, Javed A, van Wijnen AJ, Stein JL, Stein GS, Lian JB (2005) Canonical WNT signaling promotes osteogenesis by directly stimulating Runx2 gene expression. J Biol Chem 280(39):33132–33140. https://doi.org/10.1074/jbc.M500608200
Yamashiro T, Zheng L, Shitaku Y, Saito M, Tsubakimoto T, Takada K, Takano-Yamamoto T, Thesleff I (2007) Wnt10a regulates dentin sialophosphoprotein mRNA expression and possibly links odontoblast differentiation and tooth morphogenesis. Differentiation 75(5):452–462. https://doi.org/10.1111/j.1432-0436.2006.00150.x
Sagomonyants K, Mina M (2014) Biphasic effects of FGF2 on odontoblast differentiation involve changes in the BMP and Wnt signaling pathways. Connect Tissue Res 55(sup1):53–56. https://doi.org/10.3109/03008207.2014.923867
Cawthorn WP, Bree AJ, Yao Y, Du B, Hemati N, Martinez-Santibanez G, MacDougald OA (2012) Wnt6, Wnt10a and Wnt10b inhibit adipogenesis and stimulate osteoblastogenesis through a beta-catenin-dependent mechanism. Bone 50(2):477–489. https://doi.org/10.1016/j.bone.2011.08.010
Qurrat Ul A, Seemab U, Nawaz S, Rashid S (2011) Integrative analyses of conserved WNT clusters and their co-operative behaviour in human breast cancer. Bioinformation 7(7):339–346
Katoh M, Katoh M (2007) AP1- and NF-kappaB-binding sites conserved among mammalian WNT10B orthologs elucidate the TNFalpha-WNT10B signaling loop implicated in carcinogenesis and adipogenesis. Int J Mol Med 19(4):699–703
Li J, Peet GW, Balzarano D, Li X, Massa P, Barton RW, Marcu KB (2001) Novel NEMO/IkappaB kinase and NF-kappa B target genes at the pre-B to immature B cell transition. J Biol Chem 276(21):18579–18590. https://doi.org/10.1074/jbc.M100846200
Everett ET, McHenry MA, Reynolds N, Eggertsson H, Sullivan J, Kantmann C, Martinez-Mier EA, Warrick JM, Stookey GK (2002) Dental fluorosis: variability among different inbred mouse strains. J Dent Res 81(11):794–798
Yan D, Gurumurthy A, Wright M, Pfeiler TW, Loboa EG, Everett ET (2007) Genetic background influences fluoride’s effects on osteoclastogenesis. Bone 41(6):1036–1044. https://doi.org/10.1016/j.bone.2007.07.018
Mousny M, Banse X, Wise L, Everett ET, Hancock R, Vieth R, Devogelaer JP, Grynpas MD (2006) The genetic influence on bone susceptibility to fluoride. Bone 39(6):1283–1289. https://doi.org/10.1016/j.bone.2006.06.006
Beertsen W, Niehof A (1986) Root-analogue versus crown-analogue dentin: a radioautographic and ultrastructural investigation of the mouse incisor. Anat Rec 215(2):106–118. https://doi.org/10.1002/ar.1092150204
Boskey AL, Maresca M, Doty S, Sabsay B, Veis A (1990) Concentration-dependent effects of dentin phosphophoryn in the regulation of in vitro hydroxyapatite formation and growth. Bone Miner 11(1):55–65. https://doi.org/10.1016/0169-6009(90)90015-8
Milan AM, Waddington RJ, Embery G (2001) Fluoride alters casein kinase II and alkaline phosphatase activity in vitro with potential implications for dentine mineralization. Arch Oral Biol 46(4):343–351
Dimuzio MT, Veis A (1978) Phosphophoryns-major noncollagenous proteins of rat incisor dentin. Calcif Tissue Res 25(2):169–178
Butler WT (1985) The chemistry and biology of mineralized tissues: proceedings of the Second International Conference on the Chemistry and Biology of Mineralized Tissues, held in Gulf Shores, Alabama, September 9-14, 1984. vol 436 p. s.n.], [S.l
Yamakoshi Y, Hu JC, Fukae M, Iwata T, Kim JW, Zhang H, Simmer JP (2005) Porcine dentin sialoprotein is a proteoglycan with glycosaminoglycan chains containing chondroitin 6-sulfate. J Biol Chem 280(2):1552–1560. https://doi.org/10.1074/jbc.M409606200
MacDougall M, Simmons D, Luan X, Nydegger J, Feng J, Gu TT (1997) Dentin Phosphoprotein and dentin sialoprotein are cleavage products expressed from a single transcript coded by a gene on human chromosome 4: dentin phosphoprotein dna sequence determination. J Biol Chem 272(2):835–842. https://doi.org/10.1074/jbc.272.2.835
Sun Y, Lu Y, Chen S, Prasad M, Wang X, Zhu Q, Zhang J, Ball H, Feng J, Butler WT, Qin C (2010) Key proteolytic cleavage site and full-length form of DSPP. J Dent Res 89(5):498–503. https://doi.org/10.1177/0022034510363109
Tsuchiya S, Simmer JP, Hu JCC, Richardson AS, Yamakoshi F, Yamakoshi Y (2011) Astacin proteases cleave dentin sialophosphoprotein (Dspp) to generate dentin phosphoprotein (Dpp). J Bone Miner Res 26(1):220–228. https://doi.org/10.1002/jbmr.202
Zhang Y, Song Y, Ravindran S, Gao Q, Huang CC, Ramachandran A, Kulkarni A, George A (2014) DSPP contains an IRES element responsible for the translation of dentin phosphophoryn. J Dent Res 93(2):155–161. https://doi.org/10.1177/0022034513516631
Lim WH, Liu B, Cheng D, Hunter DJ, Zhong Z, Ramos DM, Williams BO, Sharpe PT, Bardet C, Mah SJ, Helms JA (2014) Wnt signaling regulates pulp volume and dentin thickness. J Bone Miner Res 29(4):892–901. https://doi.org/10.1002/jbmr.2088
Chen LF, Williams SA, Mu Y, Nakano H, Duerr JM, Buckbinder L, Greene WC (2005) NF-kappaB RelA phosphorylation regulates RelA acetylation. Mol Cell Biol 25(18):7966–7975. https://doi.org/10.1128/MCB.25.18.7966-7975.2005
Arab-Nozari M, Mohammadi E, Shokrzadeh M, Ahangar N, Amiri FT, Shaki F (2020) Co-exposure to non-toxic levels of cadmium and fluoride induces hepatotoxicity in rats via triggering mitochondrial oxidative damage, apoptosis, and NF-kB pathways. Environ Sci Pollut Res Int 27(19):24048–24058. https://doi.org/10.1007/s11356-020-08791-4
Refsnes M, Skuland T, Lag M, Schwarze PE, Ovrevik J (2014) Differential NF-kappaB and MAPK activation underlies fluoride- and TPA-mediated CXCL8 (IL-8) induction in lung epithelial cells. J Inflamm Res 7:169–185. https://doi.org/10.2147/JIR.S69646
Baskiewicz-Masiuk M, Rybicka M, Gutowska I, Bober J, Grymula K, Dziedziejko V (2004) Sodium fluoride enhancement of monocyte differentiation via nuclear factor Kappa B mechanism. In
Zhang M, Wang A, Xia T, He P (2008) Effects of fluoride on DNA damage, S-phase cell-cycle arrest and the expression of NF-kappaB in primary cultured rat hippocampal neurons. Toxicol Lett 179(1):1–5. https://doi.org/10.1016/j.toxlet.2008.03.002
Rani CSS, MacDougall M (2000) Dental cells express factors that regulate bone resorption. Mol Cell Biol Res Commun 3(3):145–152. https://doi.org/10.1006/mcbr.2000.0205
Kobayashi CAN, Leite AL, Peres-Buzalaf C, Carvalho JG, Whitford GM, Everett ET, Siqueira WL, Buzalaf MAR (2014) Bone response to fluoride exposure is influenced by genetics. PLoS One 9(12):e114343. https://doi.org/10.1371/journal.pone.0114343
Cheng PT, Bader SM, Grynpas MD (1995) Biphasic sodium fluoride effects on bone and bone mineral: a review. Cells Mater 5(3):271–282
Smalley JW, Embery G (1980) The influence of fluoride administration on the structure of proteoglycans in the developing rat incisor. Biochem J 190(2):263–272. https://doi.org/10.1042/bj1900263
Waddington RJ, Embery G, Hall RC (1993) The influence of fluoride on proteoglycan structure using a rat odontoblast in vitro system. Calcif Tissue Int 52(5):392–398. https://doi.org/10.1007/BF00310205
Waddington RJ, Moseley R, Smith AJ, Sloan AJ, Embery G (2004) Fluoride-induced changes to proteoglycan structure synthesised within the dentine–pulp complex in vitro. Biochim Biophys Acta (BBA) - Mol Basis Dis 1689(2):142–151. https://doi.org/10.1016/j.bbadis.2004.03.003
Susheela AK, Sharma K (1988) Fluoride-induced changes in the tooth glycosaminoglycans: an in vivo study in the rabbit. Arch Toxicol 62(4):328–330. https://doi.org/10.1007/BF00332496
Hall RC, Embery G, Waddington RJ (1996) Modification of the proteoglycans of rat incisor dentin-predentin during in vivo fluorosis. Eur J Oral Sci 104(3):285–291. https://doi.org/10.1111/j.1600-0722.1996.tb00079.x
Goldberg M, Takagi M (1993) Dentine proteoglycans: composition, ultrastructure and functions. Histochem J 25(11):781–806. https://doi.org/10.1007/BF02388111
Embery G, Hall R, Waddington R, Septier D, Goldberg M (2001) Proteoglycans in dentinogenesis. Crit Rev Oral Biol Med 12(4):331–349. https://doi.org/10.1177/10454411010120040401
de Mattos Pimenta Vidal C, Leme-Kraus AA, Rahman M, Farina AP, Bedran-Russo AK (2017) Role of proteoglycans on the biochemical and biomechanical properties of dentin organic matrix. Arch Oral Biol 82:203–208. https://doi.org/10.1016/j.archoralbio.2017.06.020
Funding
This study was supported by funding from the Division of Pediatric Dentistry, Department of Orofacial Sciences, and the UCSF Center for Children’s Oral Health Research.
Author information
Authors and Affiliations
Contributions
YN designed and coordinated the study, contributed to data analysis and writing of the manuscript; YK contributed to histological data gathering and analysis and writing of the manuscript; NI contributed to histological data gathering and analysis; DU contributed to in vitro data analysis; ZU and YZ completed fluoride assay; HO contributed to EMPA analysis; MD contributed to data discussion; YT completed TEM analysis and contributed to the writing of the manuscript; PDB contributed to study design and writing of the manuscript.
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kao, YH., Igarashi, N., Abduweli Uyghurturk, D. et al. Fluoride Alters Signaling Pathways Associated with the Initiation of Dentin Mineralization in Enamel Fluorosis Susceptible Mice. Biol Trace Elem Res 199, 3021–3034 (2021). https://doi.org/10.1007/s12011-020-02434-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-020-02434-y