Abstract
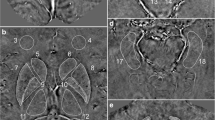
The current study was aimed to ascertain the effect of blood lead level on brain tissues in patients with type 2 diabetes. A total of 300 human participants ages 27 to 60 years with type 2 diabetes (n = 150) and healthy individuals (n = 150) were included in this study. The serum samples were used for measuring HbA1c and fasting blood glucose. Blood lead level was measured using flame atomic absorption spectrophotometer. Magnetic resonance imaging sub-analysis was used to assess the brain hyperintensities. Brain hyperintensities were found in 55% of patients with diabetes and 6% of non-diabetic control group subjects. The deep white matter hyperintensities were observed in 45% of diabetic patients, while the subcortical hyperintensities were noted in 10% of cases. Entorhinal cortex changes (31%) and hippocampus changes (42%) were noted in diabetic patients with brain hyperintensities. Diabetic patients with brain hyperintensities showed higher blood lead levels, HbA1c, and fasting blood sugar (p < 0.0001) as compared with healthy volunteers. A higher correlation (R2 = 0.8922) was found between deep white matter hyperintensities’ size and blood lead levels. In nutshell, persistence of high blood lead level in diabetic patients may progress to brain hyperintensities which may consequently lead to cognitive, behavioral changes and Alzheimer’s disease.










Similar content being viewed by others
References
(2014) Diagnosis and classification of diabetes mellitus. Diabetes Care 37(Suppl 1):S81–S90. https://doi.org/10.2337/dc14-S081
Acharjee S, Ghosh B, Al-Dhubiab BE, Nair AB (2013) Understanding type 1 diabetes: etiology and models. Can J Diabetes 37(4):269–276. https://doi.org/10.1016/j.jcjd.2013.05.001
Fayfman M, Pasquel FJ, Umpierrez GE (2017) Management of hyperglycemic crises: diabetic ketoacidosis and hyperglycemic hyperosmolar state. Med Clin North Am 101(3):587–606. https://doi.org/10.1016/j.mcna.2016.12.011
Thakur AK, Tyagi S, Shekhar N (2019) Comorbid brain disorders associated with diabetes: therapeutic potentials of prebiotics, probiotics and herbal drugs. Transl Med Commun 4(1):12. https://doi.org/10.1186/s41231-019-0043-6
Nair AB, Gupta S, Al-Dhubiab BE, Jacob S, Shinu P, Shah J, Morsy MA, SreeHarsha N, Attimarad M, Venugopala KN, Akrawi SH (2019) Effective therapeutic delivery and bioavailability enhancement of pioglitazone using drug in adhesive transdermal patch. Pharmaceutics 11(7):359. https://doi.org/10.3390/pharmaceutics11070359
Nelson PT, Smith CD, Abner EA, Schmitt FA, Scheff SW, Davis GJ, Keller JN, Jicha GA, Davis D, Wang-Xia W, Hartman A, Katz DG, Markesbery WR (2009) Human cerebral neuropathology of type 2 diabetes mellitus. Biochim Biophys Acta 1792(5):454–469. https://doi.org/10.1016/j.bbadis.2008.08.005
Peila R, Rodriguez BL, Launer LJ (2002) Type 2 diabetes, APOE gene, and the risk for dementia and related pathologies: the Honolulu-Asia Aging Study. Diabetes 51(4):1256–1262. https://doi.org/10.2337/diabetes.51.4.1256
Wani AL, Ara A, Usmani JA (2015) Lead toxicity: a review. Interdiscip Toxicol 8(2):55–64. https://doi.org/10.1515/intox-2015-0009
Obeng-Gyasi E (2019) Sources of lead exposure in various countries. Rev Environ Health 34(1):25–34. https://doi.org/10.1515/reveh-2018-0037
Qu CS, Ma ZW, Yang J, Liu Y, Bi J, Huang L (2012) Human exposure pathways of heavy metals in a lead-zinc mining area, Jiangsu Province, China. PLoS One 7(11):e46793. https://doi.org/10.1371/journal.pone.0046793
Jarrar BM, Mahmoud ZN (2000) Histochemical demonstration of changes in the activity of hepatic phosphatases induced by experimental lead poisoning in male white rats (Rattus norvegicus). Toxicol Ind Health 16(1):7–15. https://doi.org/10.1177/074823370001600102
Bonini MG, Sargis RM (2018) Environmental toxicant exposures and type 2 diabetes mellitus: two interrelated public health problems on the rise. Curr Opin Toxicol 7:52–59. https://doi.org/10.1016/j.cotox.2017.09.003
Heindel JJ, Blumberg B, Cave M, Machtinger R, Mantovani A, Mendez MA, Nadal A, Palanza P, Panzica G, Sargis R, Vandenberg LN, vom Saal F (2017) Metabolism disrupting chemicals and metabolic disorders. Reprod Toxicol 68:3–33. https://doi.org/10.1016/j.reprotox.2016.10.001
Jan AT, Azam M, Siddiqui K, Ali A, Choi I, Haq QM (2015) Heavy metals and human health: mechanistic insight into toxicity and counter defense system of antioxidants. Int J Mol Sci 16(12):29592–29630. https://doi.org/10.3390/ijms161226183
Poole LB (2015) The basics of thiols and cysteines in redox biology and chemistry. Free Radic Biol Med 80:148–157. https://doi.org/10.1016/j.freeradbiomed.2014.11.013
Leff T, Stemmer P, Tyrrell J, Jog R (2018) Diabetes and exposure to environmental Lead (Pb). Toxics 6(3):54. https://doi.org/10.3390/toxics6030054
Mostafalou S, Baeeri M, Bahadar H, Soltany-Rezaee-Rad M, Gholami M, Abdollahi M (2015) Molecular mechanisms involved in lead induced disruption of hepatic and pancreatic glucose metabolism. Environ Toxicol Pharmacol 39(1):16–26. https://doi.org/10.1016/j.etap.2014.11.001
Sun H, Wang N, Nie X, Zhao L, Li Q, Cang Z, Chen C, Lu M, Cheng J, Zhai H, Xia F, Ye L, Lu Y (2017) Lead exposure induces weight gain in adult rats, accompanied by DNA hypermethylation. PLoS One 12(1):e0169958. https://doi.org/10.1371/journal.pone.0169958
Collin F (2019) Chemical basis of reactive oxygen species reactivity and involvement in neurodegenerative diseases. Int J Mol Sci 20(10):2407. https://doi.org/10.3390/ijms20102407
Ercal N, Gurer-Orhan H, Aykin-Burns N (2001) Toxic metals and oxidative stress part I: mechanisms involved in metal-induced oxidative damage. Curr Top Med Chem 1(6):529–539. https://doi.org/10.2174/1568026013394831
Patrick L (2006) Lead toxicity part II: the role of free radical damage and the use of antioxidants in the pathology and treatment of lead toxicity. Altern Med Rev 11(2):114–127
Kim GH, Kim JE, Rhie SJ, Yoon S (2015) The role of oxidative stress in neurodegenerative diseases. Exp Neurobiol 24(4):325–340. https://doi.org/10.5607/en.2015.24.4.325
Salim S (2017) Oxidative stress and the central nervous system. J Pharmacol Exp Ther 360(1):201–205. https://doi.org/10.1124/jpet.116.237503
Mason LH, Harp JP, Han DY (2014) Pb neurotoxicity: neuropsychological effects of lead toxicity. Biomed Res Int 2014:840547–840548. https://doi.org/10.1155/2014/840547
Flora G, Gupta D, Tiwari A (2012) Toxicity of lead: a review with recent updates. Interdiscip Toxicol 5(2):47–58. https://doi.org/10.2478/v10102-012-0009-2
Searle AK, Baghurst PA, van Hooff M, Sawyer MG, Sim MR, Galletly C, Clark LS, McFarlane AC (2014) Tracing the long-term legacy of childhood lead exposure: a review of three decades of the port Pirie cohort study. Neurotoxicology 43:46–56. https://doi.org/10.1016/j.neuro.2014.04.004
Ris MD, Dietrich KN, Succop PA, Berger OG, Bornschein RL (2004) Early exposure to lead and neuropsychological outcome in adolescence. J Int Neuropsychol Soc 10(2):261–270. https://doi.org/10.1017/s1355617704102154
Surkan PJ, Zhang A, Trachtenberg F, Daniel DB, McKinlay S, Bellinger DC (2007) Neuropsychological function in children with blood lead levels <10 microg/dL. Neurotoxicology 28(6):1170–1177. https://doi.org/10.1016/j.neuro.2007.07.007
Jacob S, Morsy MA, Nair A (2018) An Overview on the Insulin Preparations and Devices. Indian J Pharm Educ Res 52(4):550–557
Ishak I, Rosli FD, Mohamed J, Mohd Ismail MF (2015) Comparison of digestion methods for the determination of trace elements and heavy metals in human hair and nails. Malays J Med Sci 22(6):11–20
Zinterhofer LJ, Jatlow PI, Fappiano A (1971) Atomic absorption determination of lead in blood and urine in the presence of EDTA. J Lab Clin Med 78(4):664–674
Assi MA, Hezmee MNM, Haron AW, Sabri MYM, Rajion MA (2016) The detrimental effects of lead on human and animal health. Vet World 9(6):660–671. https://doi.org/10.14202/vetworld.2016.660-671
Kosnett MJ, Wedeen RP, Rothenberg SJ, Hipkins KL, Materna BL, Schwartz BS, Hu H, Woolf A (2007) Recommendations for medical management of adult lead exposure. Environ Health Perspect 115(3):463–471. https://doi.org/10.1289/ehp.9784
Chen YW, Yang CY, Huang CF, Hung DZ, Leung YM, Liu SH (2009) Heavy metals, islet function and diabetes development. Islets 1(3):169–176. https://doi.org/10.4161/isl.1.3.9262
Kolachi NF, Kazi TG, Afridi HI, Kazi N, Khan S, Kandhro GA, Shah AQ, Baig JA, Wadhwa SK, Shah F, Jamali MK, Arain MB (2011) Status of toxic metals in biological samples of diabetic mothers and their neonates. Biol Trace Elem Res 143(1):196–212. https://doi.org/10.1007/s12011-010-8879-7
Roberts RO, Knopman DS, Przybelski SA, Mielke MM, Kantarci K, Preboske GM, Senjem ML, Pankratz VS, Geda YE, Boeve BF, Ivnik RJ, Rocca WA, Petersen RC, Jack CR Jr (2014) Association of type 2 diabetes with brain atrophy and cognitive impairment. Neurology 82(13):1132–1141. https://doi.org/10.1212/wnl.0000000000000269
Li W, Risacher SL, Huang E, Saykin AJ, Alzheimer's Disease Neuroimaging I (2016) Type 2 diabetes mellitus is associated with brain atrophy and hypometabolism in the ADNI cohort. Neurology 87(6):595–600. https://doi.org/10.1212/WNL.0000000000002950
Dunham CM, Cook AJ 2nd, Paparodis AM, Huang GS (2016) Practical one-dimensional measurements of age-related brain atrophy are validated by 3-dimensional values and clinical outcomes: a retrospective study. BMC Med Imaging 16:32–32. https://doi.org/10.1186/s12880-016-0136-x
Hoffmann M (2013) The human frontal lobes and frontal network systems: an evolutionary, clinical, and treatment perspective. ISRN Neurol 2013:892459–892434. https://doi.org/10.1155/2013/892459
Duan Y, Lin Y, Rosen D, Du J, He L, Wang Y (2020) Identifying morphological patterns of hippocampal atrophy in patients with mesial temporal lobe epilepsy and Alzheimer disease. Front Neurol 11:21–21. https://doi.org/10.3389/fneur.2020.00021
Hilal S, Xin X, Ang SL, Tan CS, Venketasubramanian N, Niessen WJ, Vrooman H, Wong TY, Chen C, Ikram MK (2015) Risk factors and consequences of cortical thickness in an Asian population. Medicine 94(23):e852. https://doi.org/10.1097/md.0000000000000852
Kaup AR, Mirzakhanian H, Jeste DV, Eyler LT (2011) A review of the brain structure correlates of successful cognitive aging. J Neuropsychiatry Clin Neurosci 23(1):6–15. https://doi.org/10.1176/jnp.23.1.jnp6
Acknowledgments
The authors thanks Al-Kadhimiya Teaching Hospital, Baghdad, Iraq, and all study participants for the support in completing this study.
Author information
Authors and Affiliations
Contributions
Study concept and design: H.S.N.A, D.K.I., M.K.H., Q.A.A.A., and P.S. Methodology, formal analysis, investigation, data curation, original draft preparation, writing, review editing and final approval of this version: H.S.N.A, D.K.I., M.K.H., Q.A.A.A, P.S., and A.B.N.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Al-Anbari, H.S.N., Ismail, D.K., Hasan, M.K. et al. High Blood Lead Levels: An Increased Risk for Development of Brain Hyperintensities among Type 2 Diabetes Mellitus Patients. Biol Trace Elem Res 199, 2149–2157 (2021). https://doi.org/10.1007/s12011-020-02359-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-020-02359-6