Abstract
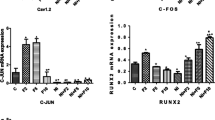
A low concentration of boron (B) accelerates the proliferation and differentiation of mammalian osteoblasts. The aim of this study was to investigate the effects of 0.1 mM of B on the membrane function of osteoblastic cells in vitro. Genes involved in cell activity were investigated using gene expression microarray analyses. The Ca2+ influx and efflux were evaluated to demonstrate the activation of L-type Ca2+ channel for the Ca2+ influx, and that of Na+/K+-ATPase for the Ca2+ efflux. A real-time PCR analysis revealed that the messenger RNA (mRNA) expression of four mineralization-related genes was clearly increased after 3 days of culture with a B-supplemented culture medium. Using microarray analyses, five genes involved in cell proliferation and differentiation were upregulated compared to the control group. Regarding the Ca2+ influx, in the nifedipine-pretreated group, the relative fluorescence intensity for 1 min after adding B solution did not increase compared with that for 1 min before addition. In the control group, the relative fluorescence intensity was significantly increased compared with the experimental group (P < 0.05). Regarding the Ca2+ efflux, in the experimental group cultured in 0.1 mM of B-supplemented medium, the relative fluorescence intensity for 10 min after ouabain treatment revealed a significantly lower slope value compared with the control group (P < 0.01). This is the first study to demonstrate the acceleration of Ca2+ flux by B supplementation in osteoblastic cells. Cell membrane stability is related to the mechanism by which a very low concentration of B promotes the proliferation and differentiation of mammalian osteoblastic cells in vitro.






Similar content being viewed by others
References
Hunt CD, Nielsen FH (1981) Interaction between boron and cholecalciferol in the chick. In: McCHowell J, Gawthorne JM, White CL (eds) Trace element metabolism in man and animals (TEMA-4). Australia: Australia Academy of Science, Canberra, pp. 597–600
Nielsen FH (1998) The justification for providing dietary guidance for the nutritional intake of boron. Biol Trace Elem Res 66(1–3):271–298
Nielsen FH (2000) The emergence of boron as nutritionally important throughout the life cycle. Nutrition 16(7–8):512–514
Nielsen FH (2004) Dietary fat composition modifies the effect of boron on bone characteristics and plasma lipids in rats. Biofactors 20(3):161–171
Gallardo-Williams MT, Maronpot RR, Turner CH, Johnson CS, Harris MW, Jayo MJ, Chapin RE (2003) Effects of boric acid supplementation on bone histomorphometry, metabolism, and biomechanical properties in aged female F-334 rats. Biol Trace Elem Res 93(1–3):155–170
Devirian TA, Volpe SL (2003) The physiological effects of dietary boron. Crit Rev Food Sci Nutr 43(2):219–231
Gorustovich AA, Steimetz T, Nielsen FH, Guglielmotti MB (2008) A histomorphometric study of alveolar bone modeling and remodeling in mice fed a boron-deficient diet. 53(7):677–682
Brown PH, Bellaloui N, Wimmer MA, Bassil ES, Ruiz J, Hu H, Pfeffer H, Dannel F, Römheld V (2002) Boron in plant biology. Plant Biol 4(2):205–223
Gonzalez-Fontes A, Navarro-Gochicoa MT, Camacho-Cristobal JJ, Herrera-Rodrigeuz MB, Quiles-Pando C, Rexach J (2014) Is Ca2+ involved in the signal transduction pathway of boron deficiency? New hypotheses for sensing boron deprivation. Plant Sci 217-218:135–139
Sanders D, Pelloux J, Brownlee C, Harper JF (2002) Calcium at the crossroads of signaling. Plant Cell 14(Suppl 1):401–417
Dodd AN, Kudla J, Sanders D (2002) The language of calcium signaling. Annu Rev Plant Biol 61:593–620
Blastic O, Kudla J (2012) Analysis of calcium signaling pathways in plants. Biochim Biophys Acta 1820(8):1283–1293
Su B, Jacinto E, Hihi M, Kallunki T, Karin M, Ben-Neriah Y (1994) JNK is involved in signal integration during costimulation of lymphocytes. Cell 77(5):727–736
Egea J, Espinet C, Comella JX (1999) Calcium influx activates extracellular-regulated kinase/mitogen-activated protein kinase pathway through a calmodulin-sensitive mechanism in PC12 cells. J Biol Chem 274(1):75–85
Apati A, Janossy J, Brozik A, Bauer PI, Magocsi M (2003) Calcium induces cell survival and proliferation through the activation of the MAPK pathway in a human hormone-dependent leukemia cell lone, TF-1. J Biol Chem 278(11):9235–9243
Hakki SS, Bozkurt BS, Hakki EE (2010) Boron regulates mineralized tissue-associated proteins in osteoblasts (MC3T3-E1). J Trace Elem Med Biol 24(4):243–250
Ying X, Cheng S, Wang W, Lin Z, Chen Q, Zhang W, Kou D, Shen Y, Cheng X, Rompis FA, Peng L, Lu CZ (2011) Effect of boron on osteogenic differentiation of human bone marrow stromal cells. Biol Trace Elem Res 144(1–3):306–315
Hotta T, Motoyama T, Watanabe H (1992) Three human osteosarcoma cell lines exhibiting different phenotypic expressions. Acta Phatol Jpn 42(8):595–603
Kawasaki A, Hayashi Y, Yanagiguchi K, Yamada S, Syudo M, Igawa K, Ikeda T, Kubo S, Fujiwara M (2012) Effects of eluted components from 4-META/MMA-TBB adhesive resin sealer on osteoblastic cell proliferation. J Dent Sci 7(2):94–98
Bolastad BM, Irizarry RA, Astrand M, Speed TP (2003) A comparison of normalization methods for high density oligonucleotide array data based on variance and bias. Bioinformatics 19(2):185–193
Quackenbush J (2002) Microarray data normalization and transformation. Nat Genet 32(Suppl):496–501
Owen TA, Aronow M, Shalhoub V, Barone LM, Wilming L, Tassinari MS, Kennedy MB, Pockwinse S, Lian JS, Stein GS (1990) Progressive development of the rat osteoblast phenotype in vitro: reciprocal relationships in expression of genes associated with osteoblast proliferation and differentiation during formation of the bone extracellular matrix. J Cell Physiol 143(3):420–430
Aubin JE, Liu F, Malaval L, Gupta AK (1995) Osteoblast and chondroblast differentiation. Bone 17(2 Suppl):77S–83S
Lian JB, Stein GS (1995) Development of the osteoblast phenotype: molecular mechanisms mediating osteoblast growth and differentiation. Iowa Orthop J 15:118–140
Duncan RL, Akanbi KA, Farach-Carson MC (1998) Calcium signals and calcium channels in osteoblastic cells. Semin Nephrol 18(2):178–190
Yamaguchi DT, Hahn TJ, Iida-Klein A, Kleeman CR, Muallem S (1987) Parathyroid hormone-activated calcium channels in an osteoblast-loke clonal osteosarcome cell line. cAMP-dependent and cAMP-independent calcium channels. J Biol Chem 260(16):7711–7718
Lieberherr M (1987) Effects of vitamin D3 metabolites on cytosolic free calcium in confluent mouse osteoblasts. J Biol Chem 262(27):13168–13173
Bidwell JP, Carter WB, Fryer MJ, Health H (1991) Parathyriod hormone (PTH)-induced intracellular Ca2+ signaling in native and PTH-desensitized osteoblast-like cells (POS 17/2.8): pharmacological characterization and evidence for synchronous oscillation of intracellular Ca2+. Endocrinology 129(6):2993–3000
Caffrey JM, Farach-Carson MC (1989) Vitamin D3 metabolites modulate dihydropyridine-sensitive calcium currents in clonal rat osteosarcoma cells. J Biol Cem 264(34):20265–20274
Civitelli R, Kim YS, Guncten SL, Fujimori A, Huskey M, Avioli LV, Hruska KA (1990) Nongenomic activation of the calcium message by vitamin D metabolites in osteoblast-like cells. Endocrinology 127(5):2253–2262
Duncan RL, Hruska KA, Misler S (1992) Parathyroid hormone activation of stretch-activated cation channels in oesteosarcoma cells (UMR-106.01). FEBS Lett 307(2):219–223
Yamaguchi DT, Green J, Kleeman CR, Muallem S (1989) Characterization of volume-sensitive, calcium-permeating pathways in the osteosarcoma cell line UMR-106-01. J Biol Chem 264(8):4383–4390
Meszaros JG, Karin NJ, Frarach-Carson MC (1996) Voltage-sensitive calcium channels in osteoblasts: mediators of plasma membrane signaling events. Connect Tissue Res 35(1–4):107–111
Loza JC, Carpio LC, Bradford PG, Dziak R (1999) Molecular characterization of the α1 subunit of the L-type voltage calcium channel expressed in rat calvarial osteoblasts. J Bone Miner Res 14(3):386–395
Fleckenstein A (1984) Calcium antagonist: history and prospects for a multifaceted pharmacodynamic principle. In: Opie LH (ed) Calcium antagonists and cardiovascular disease. Raven Press, New York, p. 9
Katz AM, Hager WD, Messineo FC, Pappano AJ (1984) Cellular actions and pharmacology of the calcium channel blocking drugs. Am J Med 77(2B):2–10
Nishiya Y, Kosaka N, Uchii M, Sugimoto S (2002) A potent 1,4-dihudropyridine L-type calcium channel blocker, benidipine, promotes osteoblast differentiation. Calcif Tissue Int 70(1):30–39
Lingrel JB, Kuntzweiler T (1994) Na+,K+-ATPase. 269(31):19659–19662
Xie Z, Askari A (2002) Na+/K+-ATPase as asignal transducer. Eur J Biochem 269(10):2434–2439
Schwartz A, Grupp G, Wallick E, Grupp IL, Ball WJ Jr (1988) Role of the Na+K+-ATPase in the cardiotonic action of cardiac glycosides. Prog Clin Biol Res 268B:321–338
Ajera T, NG YC (1991) Digitalis sensitivity of Na+,K+-ATPase, myocytes and heart. Life Sci 48(2):97–106
Acknowledgments
All of authors except for the first two authors were actively involved in the execution of the study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by the Japan Society for the Promotion of Science (JSPS) KAKENHI (Grant Number 25670812).
Conflict of Interest
Yoshihiko Hayashi has received research grants from the JSPS. The authors certify that there are no conflicts of interest to declare.
Additional information
Mark Luigi Fabian Capati and Ayako Nakazono contributed equally to this work.
Rights and permissions
About this article
Cite this article
Capati, M.L.F., Nakazono, A., Igawa, K. et al. Boron Accelerates Cultured Osteoblastic Cell Activity through Calcium Flux. Biol Trace Elem Res 174, 300–308 (2016). https://doi.org/10.1007/s12011-016-0719-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-016-0719-y