Abstract
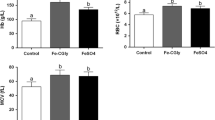
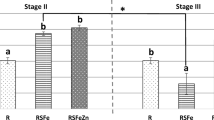
Ferrous glycinate (Fe-Gly) maintains high bioavailability in animals, but its exact absorption mechanism is still unknown. Here, we studied on the absorption kinetics of ferrous glycinate and its impact on the relevant transport protein in Sprague-Dawley (SD) rats. A total of 72 SD rats (male, BW 100 ± 6.25 g) were randomly allotted to three treatments. These treatments were perfused with 1 mL of normal saline, ferrous sulfate (FeSO4), and ferrous glycinate (71.35 mg/L as iron) separately. Four rats were selected from each treatment for collection of blood from the tails at certain times (15, 30, 45, 60, 75, 90, 120, 240, and 360 min) after gavage. Moreover, other six rats selected from each treatment were slaughtered for sampling after gavage at 2, 4, and 6 h to evaluate the expression of intestinal transport protein. Pharmacokinetic parameters of iron were determined by one-compartmental analysis. Compared with FeSO4, the peak plasma concentration of iron (C max) is higher in the rats given gavage with Fe-Gly (P < 0.05). Four hours after gavage with Fe-Gly, the expression of divalent metal transporter 1 (DMT1) in the duodenum is significantly decreased (P < 0.05), but the expression of ferroportin 1 (Fpn1) is significantly increased (P < 0.05). This study indicates that Fe-Gly as iron sources can be absorbed more and utilized faster than FeSO4, and they had different effects on the expression of intestinal transport protein.



Similar content being viewed by others
References
Hentze MW, Muckenthaler MU, Andrews NC (2004) Balancing acts: molecular control of mammalian iron metabolism. Cell 117(3):285–297
Andrews NC (2000) Intestinal iron absorption: current concepts circa 2000. Dig Liver Dis 32(1):56–61
Núñez MT, Tapia V, Rojas A, Aguirre P, Gómez F, Nualart F (2010) Iron supply determines apical/basolateral membrane distribution of intestinal iron transporters DMT1 and ferroportin 1. Am J Physiol Cell Physiol 298(3):C477–C485
World Health Organization (1999) Nutrition for health and development: progress and prospects on the eve of the 21st century. WHO, Geneva
Veum TL, Bollinger DW, Ellersieck M, Halley JT (1995) Proteinated trace minerals and condensed fish protein digest in weanling pig diets. J Anim Sci 73(Suppl 1):308
Bovell-Benjamin AC, Viteri FE, Allen LH (2000) Iron absorption from ferrous bisglycinate and ferric trisglycinate in whole maize is regulated by iron status. Am J Clin Nutr 71(6):1563–1569
Kegley EB, Spears JW, Flowers WL, Schoenherr WD (2002) Iron methionine as a source of iron for the neonatal pig. Nutr Res 22(10):1209–1217
Creech BL, Spears JW, Flowers WL, Hill GM, Lloyd KE, Armstrong TA, Engle TE (2004) Effect of dietary trace mineral concentration and source (inorganic vs. chelated) on performance, mineral status, and fecal mineral excretion in pigs from weaning through finishing. J Anim Sci 82(7):2140–2147
Feng J, Ma WQ, Niu HH, Wu XM, Wang Y, Feng J (2010) Effects of zinc glycine chelate on growth, hematological, and immunological characteristics in broilers. Biol Trace Elem Res 133(2):203–211
Ma WQ, Niu HH, Feng J, Wang Y, Feng J (2011) Effects of zinc glycine chelate on oxidative stress, contents of trace elements, and intestinal morphology in broilers. Biol Trace Elem Res 142(3):546–556
Ma WQ, Sun H, Zhou Y, Wu J, Feng J (2012) Effects of iron glycine chelate on growth, tissue mineral concentrations, fecal mineral excretion, and liver antioxidant enzyme activities in broilers. Biol Trace Elem Res 149(2):204–211
Fang CL, Zhuo Z, Fang SL, Yue M, Feng J (2013) Iron sources on iron status and gene expression of iron related transporters in iron-deficient piglets. Anim Feed Sci Technol 182:121–125
Layrisse M, García-Casal MN, Solano L, Barón MA, Arguello F, Llovera D et al (2000) Iron bioavailability in humans from breakfasts enriched with iron bis-glycine chelate, phytates and polyphenols. J Nutr 130(9):2195–2199
Pineda O, Ashmead HD (2001) Effectiveness of treatment of iron-deficiency anemia in infants and young children with ferrous bis-glycinate chelate. Nutr 17(5):381–384
Rodgerson DO, Helfer RE (1966) Determination of iron in serum or plasma by atomic absorption spectrophotometry. Clin Chem 12(6):338–349
Yong Z (2007) A data analysis program in pharmacokinetics base on Microsoft Excel—development and validation of PKSlover 1.0. J Math Med 1, 026
Design RTPP (2005) Facile means for quantifying microRNA expression by real-time PCR. Biotechniques 39(4):519–525
Dupic F, Fruchon S, Bensaid M, Loreal O, Brissot P, Borot N et al (2002) Duodenal mRNA expression of iron related genes in response to iron loading and iron deficiency in four strains of mice. Gut 51(5):648–653
Rio DC, Ares M, Hannon GJ, Nilsen TW (2010). Purification of RNA using TRIzol (TRI reagent). Cold Spring Harb Protoc, 2010(6): pdb-prot5439 doi: 10.1101/pdb.prot5439
Ammerman CB, Baker DH, Lewis AJ (1995) Bioavailability of Nutrients for Animals: Amino Acids, Minerals, and Vitamins. Academic, San Diego
Allen LH, Bovell-Benjamin AC, Viteri F (1998) Ferrous bis- and ferric tris-glycinates as iron fortificants for whole maize: bioavailability and regulation by iron status. FASEB J 12:A821
Langini S, Carbone N, Galdi M et al (1988) Ferric glycinate iron bioavailability for rats, as determined by extrinsic radioisotopic labeling of infant formulas. Nutr Rep Int 38:729–735
Aghazadeh-Habashi A, Sattari S, Pasutto F, Jamali F (2002) Single dose pharmacokinetics and bioavailability of glucosamine in the rat. J Pharm Pharm Sci 5(2):181–184
Daud AI, Xu C, Hwu WJ, Urbas P, Andrews S, Papadopoulos NE et al (2011) Pharmacokinetic/pharmacodynamic analysis of adjuvant pegylated interferon α-2b in patients with resected high-risk melanoma. Cancer Chemother Pharmacol 67(3):657–666
Zhang Y, Zhao D, Xu J, Xu C, Dong C, Liu Q et al (2013) Effects of dietary factors on the pharmacokinetics of 58Fe-labeled Hemin after oral administration in normal rats and the iron-deficient rats. Biol Trace Elem Res 1–8
Zoller H, Theurl I, Koch R, Kaser A, Weiss G (2002) Mechanisms of iron mediated regulation of the duodenal iron transporters divalent metal transporter 1 and ferroportin 1. Blood Cells Mol Dis 29(3):488–497
Foot NJ, Dalton HE, Shearwin-Whyatt LM, Dorstyn L, Tan SS, Yang B, Kumar S (2008) Regulation of the divalent metal ion transporter DMT1 and iron homeostasis by a ubiquitin-dependent mechanism involving Ndfips and WWP2. Blood 112(10):4268–4275
Abboud S, Haile DJ (2000) A novel mammalian iron-regulated protein involved in intracellular iron metabolism. J Biol Chem 275(26):19906–19912
Mackenzie EL, Iwasaki K, Tsuji Y (2008) Intracellular iron transport and storage: from molecular mechanisms to health implications. Antioxid Redox Signal 10(6):997–1030
Rolfs A, Bonkovsky HL, Kohlroser JG, McNeal K, Sharma A, Berger UV, Hediger MA (2002) Intestinal expression of genes involved in iron absorption in humans. Am J Physiol Gastrointest Liver Physiol 282(4):G598–G607
Zoller H, Koch RO, Theurl I, Obrist P, Pietrangelo A, Montosi G et al (2001) Expression of the duodenal iron transporters divalent-metal transporter 1 and ferroportin 1 in iron deficiency and iron overload. Gastroenterology 120(6):1412–1419
Oates PS, Trinder D, Morgan EH (2000) Gastrointestinal function, divalent metal transporter-1 expression and intestinal iron absorption. Pflugers Arch 440(3):496–502
Tandy S, Williams M, Leggett A, Lopez-Jimenez M, Dedes M, Ramesh B et al (2000) Nramp2 expression is associated with pH-dependent iron uptake across the apical membrane of human intestinal Caco-2 cells. J Biol Chem 275(2):1023–1029
Arredondo M, Orellana A, Garate MA, Nunez MT (1997) Intracellular iron regulates iron absorption and IRP activity in intestinal epithelial (Caco-2) cells. Am J Physiol Gastrointest Liver Physiol 273(2):G275–G280
van Campen DR, Mitchell EA (1965) Absorption of Cu64, Zn65, Mo99, and Fe59 from ligated segments of the rat gastrointestinal tract. J Nutr 86(2):120–124
Pantopoulos K, Porwal SK, Tartakoff A, Devireddy L (2012) Mechanisms of mammalian iron homeostasis. Biochem 51(29):5705–5724
Mena NP, Esparza A, Tapia V, Valdés P, Núñez MT (2008) Hepcidin inhibits apical iron uptake in intestinal cells. Am J Physiol Gastrointest Liver Physiol 294(1):G192–G198
Acknowledgments
This work was supported by a Key Science Project “973” Award from National Science and Technology Committee (no. 2012CB124705), National Natural Science Foundation (no. 31272398), New-Century Training Program Foundation for Talents from the Ministry of Education of China (no. NCET-10-0727), and the Natural Science Foundation for Distinguished Young Scholars of Zhejiang Province, China (no. R3110085).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhuo, Z., Fang, S., Yue, M. et al. Kinetics Absorption Characteristics of Ferrous Glycinate in SD Rats and Its Impact on the Relevant Transport Protein. Biol Trace Elem Res 158, 197–202 (2014). https://doi.org/10.1007/s12011-014-9906-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-014-9906-x