Abstract
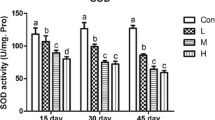
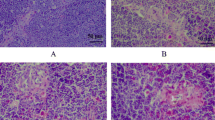
The purpose of this 42-day study was to investigate the effects of low selenium (Se) on histopathological changes of spleen, cell cycle and apoptosis of splenocytes, and oxidative status of the spleen. One hundred twenty 1-day-old avian broilers were randomly divided into two groups of 60 each and were fed on a low Se diet (0.0342 mg/kg Se) or a control diet (0.2 mg/kg Se), respectively. The weight and relative weight of the spleen were significantly decreased in the low Se group when compared with those of the control group. Histopathologically, splenic lesions in low-Se chicken were characterized by lymphocyte depletion and congestion of red pulp. As measured by flow cytometry, splenocytes in G0/G1 phase were significantly increased, while splenocytes in S phase and G2 + M phase were obviously decreased in the low Se group. The percentage of apoptotic splenocytes was greatly increased in the low Se group when compared with that of control group. At the same time, the occurrence frequencies of apoptotic splenocytes was markedly increased in the low Se group with the appearance of condensed nucleus ultrastructurally. Oxidative stress in the spleens of the low Se group was evidenced by decrease in glutathione peroxidase and catalase activities and increase in malondialdehyde contents. The results showed that low Se diet intake caused increased apoptosis, arrested cell cycle, and obvious oxidative stress, which provided a possible pathway for the injured structure and immune function of the spleen in chickens.

Similar content being viewed by others
References
McKenzie RC, Rafferty TS, Beckett GJ (1998) Selenium: an essential element for immune function. Immunol Today 19:342–345
Brown KM, Arthur JR (2001) Selenium, selenoproteins and human health: a review. Public Health Nutr 4:593–599
Bhaskaram P (2002) Micronutrient malnutrition, infection, and immunity: an overview. Nutr Rev 60:S40–S45
Marsh JA, Combs GF Jr, Whitacre ME et al (1986) Effect of selenium and vitamin E dietary deficiencies on chick lymphoid organ development. Proc Soc Exp Biol Med 182:425–436
Cui HM (1988) The histopathologic study of selenium deficiency in chicks. Acta Vet Zootech Sin 1:52–56
Peng X, Cui Y, Cui W et al (2011) The cell cycle arrest and apoptosis of bursa of Fabrcius induced by low selenium in chickens. Biol Trace Elem Res 139:32–40
Peng X, Cui H, Deng J et al (2011) Low dietary selenium induce increased apoptotic thymic cells and alter peripheral blood T cell subsets in chicken. Biol Trace Elem Res 142:167–173
Kiremidjian-Schumacher L, Roy M, Wishe HI et al (1994) Supplementation with selenium and human immune cell functions, II: effect on cytotoxic lymphocytes and natural killer cells. Biol Trace Elem Res 41:115–127
Vega L, Rodríguez-Sosa M, García-Montalvo EA et al (2007) Non-optimal levels of dietary selenomethionine alter splenocyte response and modify oxidative stress markers in female mice. Food Chem Toxicol 45:1147–1153
Spallholz JE, Stewart JE (1989) Advances in the role of minerals in immunobiology. Biol Trace Elem Res 19:129–151
Marsh JR, Dietert RR, Combs GF Jr (1981) Influence of dietary selenium and vitamin E on the humoral immune response of the chick. Proc Soc Exp Biol Med 166:228–236
Serfass RE, Ganther HE (1975) Defective microbicidal activity in glutathione peroxidase-deficient neutrophils of selenium-deficient rats. Nature 255:640–641
Gladyshev VM, Hatfield DL (1999) Selenocysteine-containing proteins in mammals. J Biomed Sci 6:151–160
Chow CK, Tappel AL (1974) Response of glutathione peroxidase to dietary selenium in rats. J Nutr 104:444–451
Knight SAB, Sunde RA (1988) Effect of selenium repletion on gluthione peroxidase protein level in rat liver. J Nutr 118:853–858
Rotruck JT, Pope AL, Ganther HE, Swanson AB, Hafeman DG, Hoekstra WG (1973) Selenium: biochemical role as a component of glutathione peroxidase. Science 179:588–590
Peng X, Cui Y, Cui W, Deng JL, Cui HM (2009) The decrease of relative weight, lesions, and apoptosis of Bursa of Fabricius induced by excess dietary selenium in chickens. Biol Trace Elem Res 131(1):33–42
Cui HM, Fang J, Peng X (2003) Pathology of the thymus, spleen and bursa of Fabricius in zinc-deficient ducklings. Avian Pathol 32:259–263
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Chen T, Cui HM, Cui Y, Gong T, Caimin B, Peng X (2010) Decreased antioxidase activities and oxidative stress in the spleen of chickens fed on high fluorine diets. Hum Exp Toxicol 30(9):1282–1286
Riddell C (1987) Avian histopathology. In: Lymphoid system. American Association of Avian Pathologists, Kansas
Rao L, Puschner B, Prolla TA (2001) Gene exprssion profiling of low selenium status in the mouse intestine: transcriptional activation of genes linked to DNA damage, cell cycle control and oxidative stress. J Nutr 131:3175–3181
Saito Y, Yoshida Y, Akazawa T, Takahashi K, Niki E (2003) Cell death caused by selenium deficiency and protective effect of antioxidants. J Biol Chem 278(41):39428–39434
Zeng H (2002) Selenite and selenomethionine promote HL-60 cell cycle progression. J Nutr 132:74–679
Cheng WH, Quimby FW, Lei XG (2003) Impacts of glutathione peroxidase-1 knockout on the protection by injected selenium against the pro-oxidant-induced liver aponecrosis and signaling in selenium-deficient mice. Free Radic Bio Med 34(7):918–927
Spallholz JE (1990) Selenium and glutathione peroxidase: essential nutrient and antioxidant component of the immune system. Adv Exp Med Biol 262:145–158
Green DR (2000) Apoptotic pathways: paper wraps stone blunts scissors. Cell 102:1–4
Mona MAH (2011) Toxicological study of sodium selenite on fetal development and DNA fragmentation in liver cells of pregnant rats. Biol Trace Elem Res 140(1):114–126
Halliwell B, Chirico S (1993) Lipid peroxidation: its mechanism, measurement, and significance. Am J Clin Nutr 57(suppl):715S–725S
Acknowledgments
This study was supported by Program for Changjiang Scholars and Innovative Research Team in University (IRT 0848) and Education Department of Sichuan Province (09ZZ017).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Peng, X., Cui, H., Fang, J. et al. Low Selenium Diet Alters Cell Cycle Phase, Apoptotic Population and Modifies Oxidative Stress Markers of Spleens in Broilers. Biol Trace Elem Res 148, 182–186 (2012). https://doi.org/10.1007/s12011-012-9357-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-012-9357-1