Abstract
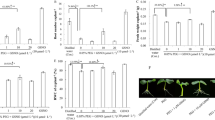
Fourteen-day-old bean seedlings were cultured in nutrient solution containing Cu2+ ions at various concentrations (50 and 75 µM of CuSO4) for 3 days. This excess of copper induced a reduction in the water volume absorbed by the plants. Moreover, this reduction was accompanied by an increase of the amount of copper taken up by the roots. Analysis by native gel electrophoresis of cell wall peroxidase activities in the roots revealed a stimulation of two anionic isoforms (A2 and A3) under cupric stress conditions. Moreover, the activity of phenylalanine ammonia lyase (EC. 4.3.1.5), which plays an important role in plant defense, was enhanced. Copper-treated bean roots showed modifications in the cell walls of various tissues. Label for lignin, callose, and suberin with berberine-aniline blue revealed abnormal cell wall thickenings in the endodermis, the phloem, and the xylem, especially in plants treated with 75 µM CuSO4.






Similar content being viewed by others
References
Bouazizi H, Jouili H, Geitmann A, El Ferjani E (2008) Effect of copper excess on H2O2 accumulation and peroxidase activities in bean roots. Acta Biol Hung 59(2):233–245
Tadeo RR, Primo-Millo E (1990) Peroxidase activity changes and lignin deposition during the senescence process in Citrus stigmas and styles. Plant Sci 68:47–56
Delannoy E, Marmey P, Penel C et al (2004) Les peroxydases végétales de classe III. Acta Bot Gall 151(4):353–380
De Jaegher G (1986) La thigmomorphogenèse de la bryone, lignification et métabolisme de l’éthylène. PhD thesis, Université de Clermont-Ferrand II, U.F.R de recherche scientifique et technique. p 195
Guttenberg H (1968) Der primäre Bau der Angiospermenwurzel. Encyclopedia of Plant Anatomy. Gebüder Borntraeger, Berlin
Haas DL, Carothers ZB, Robbins RR (1976) Observations on the phi thickening and Casparian strips in pelargonium roots. Am J Bot 63:863–867
Degenhardt B, Gimmler H (2000) Cell wall adaptation to multiple environmental stresses in maize roots. J Exp Bot 51:595–603
Schützendübel A, Schwanz P, Teichmann T et al (2001) Cadmium-induced changes in antioxidative systems, hydrogen peroxide content and differentiation in scots pine roots. Plant Physiol 127:887–898
Chen EL, Chen YA, Chen LM et al (2002) Effect of copper on peroxidase activity and lignin content in Raphanus sativus. Plant Physiol Biochem 40:439–444
Mazhoudi S, Chaoui A, Ghorbal MH et al (1997) Response of antioxidant enzymes to excess copper in 252 tomato (Lycopersicon esculentum, Mill.) Plant Sci 127:182–186
Jouili H, El Ferjani E (2003) Changes in antioxidant and lignifying enzyme activities in sunflower roots (Helianthus annuus L.) stressed with copper excess. CR Biologies 326:639–644
Swain T, Hillis WE (1959) The phenolic constituents of Prunus domestica 1. The quantitative analysis of phenolic constituent. J Sci Food Agric 10:63–68
Brundrett MC, Enstone DE, Peterson CA (1988) A berberine–aniline blue fluorescent staining procedure for suberin, lignin, and callose in Plant Tissue. Protoplasma 146:133–142
Reichman SM (2002) The responses of plants to metal toxicity: a review focusing on copper, manganese and zinc. Aust Miner Energy Environ Found 14:1–54
Baker AJ (1981) Accumulators and excluders strategies in the response of plants to heavy Metals. J Plant Nutr 3:611–654
CH LCH, Chen LM, Liu ZH (2005) Rapid effect of copper on lignin biosynthesis in soybean roots. Plant Sci 168:855–861
Bouazizi H, Jouili H, Geitmann A et al (2007) Copper-induce oxidative stress in maize shoots (Zea mays L.) H2O2 accumulation and peroxidases modulation. Acta Biol Hung 58(2):209–218
Santiago LMS, Louro RP, Dulce E (2000) Compartmentation of phenolic compounds and phenylalanine ammonia-lyase in leaves of Phyllanthus tenellus Roxb and their induction by copper sulfate. Ann Bot 86:1023–1032
Nicholson RL, Hammerschmid TR (1992) Phenolic compounds and their role in disease resistance. Annu Rev Phytopathol 30:369–389
Ganeva G, Zozikova E (2007) Effect of increasing Cu2+ concentrations on growth and content of free phenols in two lines of wheat (Triticum aestivum) with different tolerance. Gen Appl Plant Physiol 33(1–2):75–82
Sgherri C, Coisantd E, Navari-Izzo F (2003) Phenols and oxidative status of Raphus sativus grown in copper excess. Physiol Plant 118:21–28
Van Tunen AJ, Mol JNM (1991) Control of favonoid synthesis and manipulation of flower colour. In: Blackie DG (ed) Developmental regulation of plant gene expression, Plant Biotechnology Series. New York: Chapman and Hall
Booker FL, Antonnen S, Heagle AS (1996) Catechin, and lignin contents of loblolly pine (Pinus taeda L.) needles after chronic exposure to ozone. New Phytol 132:483–492
Barceló J, Poschenrieder CH (1990) Plant water relations as affected by heavy metal stress: a review. J Plant Nutr 13:1–37
Barceló J, Vázquez MD, Pschenrieder CH (1988) Cadmium-induced structural and ultrastructural changes in the vascular system of bush bean stems. Bot Acta 101:254–261
Romero-Puertas MC (2002) Metabolismo de especies de oxigeno reaction en plantas de guisante (Pisum sativum L.) y en peroxisomas de hojas en condiciones de estrès pur cadmio. PhD thesis, University of Granada, Spain
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bouazizi, H., Jouili, H., Geitmann, A. et al. Structural Changes of Cell Wall and Lignifying Enzymes Modulations in Bean Roots in Response to Copper Stress. Biol Trace Elem Res 136, 232–240 (2010). https://doi.org/10.1007/s12011-009-8530-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-009-8530-7