Abstract
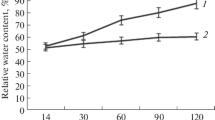
The association of bean yellow mosaic potyvirus (BYMV) was investigated earlier with the severe mosaic and stunting disease of V. faba. In the present study, we mechanically transmitted BYMV on V. faba to assess the impact on physiological, biochemical, and nutritional attributes. BYMV-inoculated plants exhibited severe symptoms, and their height, length of the pod, and seed yield (size and number) were reduced to half of the mock-inoculated V. faba. In BYMV-inoculated V. faba, chlorophyll a, b, and total (Chl a + Chl b) were lowered to 66.70%, 64.94%, and 66.19% respectively, and corroborated with the decrease in photosynthetic efficiency (Fv/Fm) from 0.36 to 0.26. An increase in membrane ion leakage and malondialdehyde was observed in inoculated V. faba indicating virus-induced physiological stress. The non-structural carbohydrates, total protein, and free proline contents were also significantly altered. More, the high accumulation of salicylic acid and other defense-related antioxidant enzymes (ascorbate peroxidase, guaiacol peroxidase, superoxide dismutase, and catalase) was observed to ameliorate virus-induced stress. An increase in polyphenols and flavonoids: gallic acid, protocatechuic acid, chlorogenic acid, caffeic acid, rutin, ferulic acid, quercetin, and kaempferol suggests the proactive action by BYMV-infected V. faba. A reduced accumulation of nutrition-related parameters while induced accumulation of anti-nutritional factors like tannin (as tannic acid) and phytate observed is suggestive of poor nutritional quality of the plants. The present study comprehensively elucidates the BYMV-induced perturbations in physio-biochemical and nutritional attributes of V. faba, diminishing the quality of plants and seeds, and raises serious concerns for the management measures against the BYMV.





Similar content being viewed by others
Data availability
The data are available in the laboratory.
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Ananthu N, Umamaheswaran K (2019) Effect of viral infection on carbohydrate and chlorophyll contents in ginger (Zingiber officinale Rosc.). Int J Curr Microbiol App Sci 8:862–867
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399. https://doi.org/10.1146/annurev.arplant.55.031903.141701
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207. https://doi.org/10.1007/BF00018060
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assay and an assay applicable to acrylamide gels. Anal Biochem 44:276–287. https://doi.org/10.1016/0003-2697(71)90370-8
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Cai YZ, Luo Q, Sun M, Corke H (2004) Antioxidant activity and phenolic compounds of 112 traditional chinese medicinal plants associated with anticancer. Life Sci 74:2157–2184. https://doi.org/10.1016/j.lfs.2003.09.047
Chaerle L, Lenk S, Hagenbeek D, Buschmann C, van Der Straeten D (2007) Multicolor fluorescence imaging for early detection of the hypersensitive reaction to tobacco mosaic virus. J Plant Physiol 164:253–326. https://doi.org/10.1016/j.jplph.2006.01.011
Chen C, Dickman MB (2005) Proline suppresses apoptosis in the fungal pathogen Colletotrichum trifolii. Proc Natl Acad Sci USA 102:3459–3464. https://doi.org/10.1073/pnas.040796010
Cheng Y, Jones RAC (2000) Biological properties of necrotic and non-necrotic strains of bean yellow mosaic virus in cool season grain legumes. Ann Appl Biol 36:215–227. https://doi.org/10.1111/j.1744-7348.2000.tb00028.x
Chojnacka K, Skrzypczak D, Izydorczyk G, Mikula K, Szopa D, Witek-Krowiak A (2021) Antiviral properties of polyphenols from plants. Foods 10:2277. https://doi.org/10.3390/foods10102277
Dat J, Vandenabeele S, Vranová E, Van Montagu M, Inzé D, van Breusegem F (2000) Dual action of the active oxygen species during plant stress responses. Cell Mol Life Sci 57:779–795. https://doi.org/10.1007/s000180050041
Dhull SB, Kidwai MK, Noor R, Chawla P, Rose PK (2021) A review of nutritional profile and processing of faba bean (Vicia faba L.). Legume Sci. https://doi.org/10.1002/leg3.129
Elbeshehy EF, Almaghrabi OA, Mahmoud WMA, Elazzazy A (2014) Effect of biosynthesized silver nanoparticles on physiological parameters of Vicia faba infected by Bean yellow mosaic virus. J Pure App Microbiol 8:803–812
Epple P, Mack AA, Morris VR, Dangl JL (2003) Antagonistic control of oxidative stress-induced cell death in Arabidopsis by two related, plant-specific zinc finger proteins. Proc Natl Acad Sci USA 100:6831–6836. https://doi.org/10.1073/pnas.1130421100
Fan L, Zheng S, Wang X (1997) Antisense suppression of phospholipase D retards abscisic acid- and ethylene-promoted senescence of postharvest arabidopsis leaves. Plant Cell 9:2183–2196. https://doi.org/10.1105/tpc.9.12.2183
Folin O, Ciocalteu V (1927) On tyrosine and tryptophane determinations in proteins. J Biol Chem 73:627–650. https://doi.org/10.1016/S0021-9258(18)84277-6
Fraser RSS (1982) Are pathogenesis-related proteins involved in acquired systemic resistance of tobacco plants to tobacco mosaic virus? J Gen Virol 58:305–313. https://doi.org/10.1099/0022-1317-58-2-305
Gachon C, Baltz R, Saindrenan P (2004) Over-expression of a scopoletin glucosyltransferase in Nicotiana tabacum leads to precocious lesion formation during the hypersensitive response to tobacco mosaic virus but does not affect virus resistance. Plant Mol Biol 541:137–146. https://doi.org/10.1023/B:PLAN.0000028775.58537.fe
Gerbling KP, Grahame JK, Fisher KH, Latzko E (1984) Partial purification and properties of soluble ascorbate peroxidases from pea leaves. J Plant Physiol 115:59–67. https://doi.org/10.1016/S0176-1617(84)80051-6
Gerchacov SM, Hatcher PG (1972) Improved technique for analysis of carbohydrates in sediments. Limnol Oceanogr 17:938–143. https://doi.org/10.4319/lo.1972.17.6.0938
Goldberg DM, Spooner RJ (1984) Glutathione reductase. In: Bergmeyer HU (Ed). Methods in enzymatic analysis. Enzymes 1: Oxidoreductases, transferases. vol. III. Verlag Chemie, Weinheim. pp 43–49
Ha C, Coombs S, Revill PA, Harding RM, Vu M, Dale JL (2008) Design and application of two novel degenerate primer pairs for the detection and complete genomic characterization of potyviruses. Arch Virol 153:25–36. https://doi.org/10.1007/s00705-007-1053-7
Hare PD, Cress WA (1997) Metabolic implications of stress induced proline accumulation in plants. Plant Growth Regul 21:79–102
Hemidia SK (2005) Effect of Bean yellow mosaic virus on physiological parameters of Vicia faba and Phaseolus vulgaris. Int J Agric Biol 7:154–157
Huang YC, Chang YH, Shao YY (2006) Effects of genotype and treatment on the antioxidant activity of sweet potato in Taiwan. Food Chem 98:529–538. https://doi.org/10.1016/j.foodchem.2005.05.083
Huseynova IM, Mirzayeva SM, Sultanova NF, Aliyeva DR, Mustafayev NSh, Aliyev JA (2018) Virus-induced changes in photosynthetic parameters and peroxidase isoenzyme contents in tomato (Solanum lycopersicum L.) plants. Photosynthetica 56:841–885. https://doi.org/10.1007/s11099-017-0737-9
Islam MDT, Hossain T, Ahmed JU, Karim S (2003) Biochemical changes in tomato fruits caused by tomato yellow leaf curl virus. Bangladesh J Bot 32:81–84
Karkanis A, Ntatsi G, Lepse L, Fernández JA, Vågen IM, Rewald B et al (2018) Faba bean cultivation-revealing novel managing practices for more sustainable and competitive European cropping systems. Front Plant Sci 9:1115. https://doi.org/10.3389/fpls.2018.01115
Katoch M, Abdin MZ, Ram R, Zaidi AA (2003) An overview of diagnostics for viruses infecting gladiolus. Crop Prot 22:153–156. https://doi.org/10.1016/S0261-2194(02)00139-4
Kaur C, Kumar S, Raj SK (2013) New record of association of Bean yellow mosaic virus with mosaic disease of Vicia faba in India. Ind J Virol 24:95–96. https://doi.org/10.1007/s13337-013-0128-1
Khan M, Rauf W, Fazal-e-Habib MR, Iqbal M (2020) Screening and identification of bioactive compounds from citrus against non-structural protein 3 protease of hepatitis C virus genotype 3a by fluorescence resonance energy transfer assay and mass spectrometry. World J Hepatol 12:976–992. https://doi.org/10.4254/wjh.v12.i11.976
Kim H, Chung MS (2018) Antiviral activities of mulberry (Morus alba) juice and seed against influenza viruses. Evid Based Complement Altern Med 2018:2606583. https://doi.org/10.1155/2018/2606583
Kumar S, Chauhan PS, Agrawal L, Raj R, Srivastava A, Gupta S et al (2016) Paenibacillus lentimorbus inoculation enhances tobacco growth and extenuates the virulence of Cucumber mosaic virus. PLoS ONE 11:e0149980. https://doi.org/10.1371/journal.pone.0149980
Kumari SG, Makkouk KM (2007) Virus diseases of faba bean (Vicia faba L.) in Asia and Africa. Plant Viruses 1:93–105
Labba ICM, Frøkiær H, Sandberg AS (2021) Nutritional and antinutritional composition of fava bean (Vicia faba L., var. minor) cultivars. Food Res Int. https://doi.org/10.1016/j.foodres.2020.110038
Lichtenthaler HK, Wellbum AR (1983) Determinations of total carotenoids and chlorophylls a and b of leaf extracts in different solvents. Biochem Soc Transactions 11:591–592. https://doi.org/10.1042/bst0110591
Luo Y, Gu Z, Han Y, Chen Z (2008) The impact of processing on phytic acid, in vitro soluble iron and Phy/Fe molar ratio of faba bean (Vicia faba L.). J Agric Food Chem 89:861–866. https://doi.org/10.1002/jsfa.3525
Maalouf F, Ahmed S, Bishaw Z (2021) Faba bean. In: Pratap A, Gupta S (Eds.), The beans and the peas From orphan to mainstream crops. Woodhead Publishing. pp 105–131 https://doi.org/10.1016/B978-0-12-821450-3.00008-1
Makkouk KM, Rizkallah L, Kumari SG, Zaki M, Abul Enien R (2003) First record of Chickpea chlorotic dwarf virus (CpCDV) affecting faba bean (Vicia faba) crops in Egypt. Plant Pathol 52:413. https://doi.org/10.1046/j.1365-3059.2003.00834.x
Mazid M, Khan TA, Khan ZH, Quddusi S, Mohammad F (2011) Occurrence, biosynthesis and potentialities of ascorbic acid in plants. Int J Plant Animal Env Sci 1(2):167–184
Mínguez MI, Rubiales D (2021) Faba bean. In: Sadras VO, Calderini DF (Eds.), Crop physiology case histories for major crops. Cambridge, MA, USA: Elsevier. pp 452–481 https://doi.org/10.1016/B978-0-12-819194-1.00015-3
Parvez MK, Tabish Rehman M, Alam P, Al-Dosari MS, Alqasoumi SI, Alajmi MF (2019) Plant-derived antiviral drugs as novel hepatitis B virus inhibitors: Cell culture and molecular docking study. Saudi Pharm J 27:389–400. https://doi.org/10.1016/j.jsps.2018.12.008
Qiu ZB, Liu X, Tian XJ, Yue M (2007) Effects of CO2 laser pretreatment on drought stress resistance in wheat. J Photochem Photobiol B 90:17–25
Radwan DEM, Fayez KA, Mahmoud SY, Lu G (2010) Modifications of antioxidant activity and protein composition of bean leaf due to Bean yellow mosaic virus infection and salicylic acid treatments. Acta Physiol Plant 32:891–904. https://doi.org/10.1007/s11738-010-0477-y
Radwana DEM, Lu G, Fayez KA, Mahmoud S (2008) Protective action of salicylic acid against bean yellow mosaic virus infection in Vicia faba leaves. J Plant Physiol 165:845–857. https://doi.org/10.1016/j.jplph.2007.07.012
Rohani B, Habibi MK, Mosahebi G (2008) Nodule infection by Bean yellow mosaic virus in Vicia faba and molecular characterization of it. Commun Agric Appl Biol Sci 73:303–306 (PMID:19226767)
Savicka M, Škute N (2010) Effects of high temperature on malondialdehyde content, superoxide production and growth changes in wheat seedlings (Triticum aestivum L.). Ekologija 56:26–33. https://doi.org/10.2478/v10055-010-0004-x
Schwinghamer MW, Nicholas AH, Schilg MA (2009) Three aphid vectors of faba bean (Vicia faba) viruses in northern New South Wales and occurrence of Acyrthosiphon pisum-transmitted isolates of Soybean dwarf virus. Austral Plant Pathol 38:262–269. https://doi.org/10.1071/AP09001
Serraj R, Sinclair TR (2002) Osmolyte accumulation: can it really help increase crop yield under drought conditions? Plant Cell Environ 25:333–341. https://doi.org/10.1046/j.1365-3040.2002.00754.x
Shaik R, Ramakrishna W (2014) Machine learning approaches distinguish multiple stress conditions using stress-responsive genes and identify candidate genes for broad resistance in rice. Plant Physiol 164:481–495. https://doi.org/10.1104/pp.113.225862
Shalitin D, Wolf S (2000) Cucumber mosaic virus infection affects sugar transport in melon plants. Plant Physiol 123:597–604
Singh AK, Bhardwaj R, Singh IS (2014) Assessment of nutritional quality of developed faba bean (Vicia faba L.) lines. J AgriSearch 1:96–101. https://doi.org/10.1007/s11738-010-0477-y
Tecsi LI, Maule AJ, Smith AM, Leegood RC (1996) A spatial analysis of physiological changes associated with infection of cotyledons of marrow plants with cucumber mosaic virus. Plant Physiol 111:975–985. https://doi.org/10.1104/pp.111.4.975
Towo E, Matuschek E, Svanberg U (2006) Fermentation and enzyme treatment of tannin sorghum gruels: effects on phenolic compounds, phytate and in vitro accessible iron. Food Chem 94:369–376. https://doi.org/10.1016/j.foodchem.2004.11.027
Wang C-J, Yang W, Wang C, Gu C, Niu D-D (2012) Induction of drought tolerance in cucumber plants by a consortium of three plant growth-promoting rhizobacterium strains. PLoS ONE. https://doi.org/10.1371/journal.pone.0052565
Whitham SA, Yang C, Goodin MM (2006) Global impact: elucidating plant responses to viral infection. Mol Plant Microbe Interact 19:1207–1215. https://doi.org/10.1094/MPMI-19-1207
Younes HA, El-Aziz MHA, Zeid AH (2021) Detection of Bean yellow mosaic potyvirus in infected faba bean plants (Vicia faba L.) from Northern Egypt. Arch Phytopathol Plant Prot 54:19–20. https://doi.org/10.1080/03235408.2021.1932285
Acknowledgements
The authors are thankful to the Director, CSIR-NBRI, Lucknow, for the facilities. Author C. Kaur is thankful to the University Grants Commission, India for her fellowship.
Funding
The authors are thankful to the Director, CSIR-National Botanical Research Institute, Lucknow, India for laboratory facilities and financial support.
Author information
Authors and Affiliations
Contributions
S.K.R conceived the experiment. C.K, R.R, and A.S and A.N did all the experiments. A.N and A.L analyzed and interpreted the biochemical and specific phenolic-related results. S.K and S.K.R wrote the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Communicated by M. Capuana.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kaur, C., Srivastava, A., Raj, R. et al. Physio-biochemical and nutritional alterations in faba bean due to bean yellow mosaic virus infection. Acta Physiol Plant 46, 54 (2024). https://doi.org/10.1007/s11738-024-03681-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-024-03681-5