Abstract
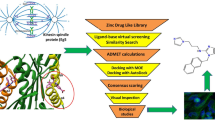
The study aimed to evaluate the potential of piperidine-based 2H chromen-2-one derivatives against targeted enzymes, i.e., cholinesterase’s and monoamine oxidase enzymes. The compounds were divided into three groups, i.e., 4a–m ((3,4-dimethyl-7-((1-methylpiperidin-4-yl)oxy)-2H-chromen-2-one derivatives), 5a–e (3,4-dimethyl-7-((1-methypipridin-3-yl)methoxy)-2H-chromen-2-one derivatives), and 7a–b (7-(3-(3,4-dihydroisoquinolin-2(1H)-yl)propoxy)-3,4-dimethyl-2H-chromen-2-one derivatives) with slight difference in the basic structure. The comprehensive computational investigations were conducted including density functional theories studies (DFTs), 2D-QSAR studies, molecular docking, and molecular dynamics simulations. The QSAR equation revealed that the activity of selected chromen-2-one-based piperidine derivatives is being affected by the six descriptors, i.e., Nitrogens Count, SdssCcount, SssOE-Index, T-2–2-7, ChiV6chain, and SssCH2E-Index. These descriptor values were further used for the preparation of chromen-2-one based piperidine derivatives. Based on this, 83 new derivatives were created from 7 selected parent compounds. The QSAR model predicted their IC50 values, with compound 4 k and 4kk as the most potent multi-targeted derivative. Molecular docking results exhibited these compounds as the best inhibitors; however, 4kk exhibited greater activity than the parent compounds. The results were further validated by molecular dynamic simulation studies along with the suitable physicochemical properties. These results prove to be an essential guide for the further design and development of new piperidine based chromen-2-one derivatives having better activity against neurodegenerative disorder.












Similar content being viewed by others
Data Availability
All the associated data will be available on request from corresponding author.
References
Lau, A., et al. (2021). Alzheimer’s disease-related metabolic pattern in diverse forms of neurodegenerative diseases. Diagnostics, 11(11), 2023.
Atri, A. (2019). The Alzheimer’s disease clinical spectrum: Diagnosis and management. Medical Clinics, 103(2), 263–293.
Perl, D. P. (2010). Neuropathology of Alzheimer’s disease. Mount Sinai Journal of Medicine: A Journal of Translational and Personalized Medicine: A Journal of Translational and Personalized Medicine, 77(1), 32–42.
Huat, T. J., et al. (2019). Metal toxicity links to Alzheimer’s disease and neuroinflammation. Journal of Molecular Biology, 431(9), 1843–1868.
Jakovljevic, M., et al. (2021). Aging and global health (pp. 73–102). Handbook of global health.
Jia, J., et al. (2018). The cost of Alzheimer’s disease in China and re-estimation of costs worldwide. Alzheimer’s & Dementia, 14(4), 483–491.
Eckroat, T. J., Manross, D. L., & Cowan, S. C. (2020). Merged tacrine-based, multitarget-directed acetylcholinesterase inhibitors 2015–present: Synthesis and biological activity. International Journal of Molecular Sciences, 21(17), 5965.
Fitzgerald, P. J., et al. (2020). The cholinesterase inhibitor donepezil has antidepressant-like properties in the mouse forced swim test. Translational Psychiatry, 10(1), 255.
Crews, F. T., et al. (2021). Loss of basal forebrain cholinergic neurons following adolescent binge ethanol exposure: Recovery with the cholinesterase inhibitor galantamine. Frontiers in Behavioral Neuroscience, 15, 652494.
Schneider, L. S. (2022). A critical review of cholinesterase inhibitors as a treatment modality in Alzheimer’s disease. Dialogues in clinical neuroscience, 2(2), 111–128.
.Jha, A., & Singh, A. (2021). Cholinesterase inhibitors used for the management of Alzheimer’s disease: A review. Journal of Pharmaceutical Research International, 33(60A), 121–128.
Szökő, É., et al. (2018). Pharmacological aspects of the neuroprotective effects of irreversible MAO-B inhibitors, selegiline and rasagiline, in Parkinson’s disease. Journal of Neural Transmission, 125, 1735–1749.
Bortolami, M., et al. (2021). Acetylcholinesterase inhibitors for the treatment of Alzheimer’s disease–A patent review (2016–present). Expert Opinion on Therapeutic Patents, 31(5), 399–420.
Chen, Z.-R., et al. (2022). Role of cholinergic signaling in Alzheimer’s disease. Molecules, 27(6), 1816.
Yang, W., et al. (2020). Current and projected future economic burden of Parkinson’s disease in the US. npj Parkinson’s Disease, 6(1), 15.
Monzio Compagnoni, G., et al. (2020). The role of mitochondria in neurodegenerative diseases: The lesson from Alzheimer’s disease and Parkinson’s disease. Molecular neurobiology, 57, 2959–2980.
Cummings, J. (2021). The role of neuropsychiatric symptoms in research diagnostic criteria for neurodegenerative diseases. The American Journal of Geriatric Psychiatry, 29(4), 375–383.
Post, B., et al. (2020). Young onset Parkinson’s disease: A modern and tailored approach. Journal of Parkinson’s Disease, 10(s1), S29–S36.
Delavar, M. R., et al. (2018). Differential expression of miR-34a, miR-141, and miR-9 in MPP+-treated differentiated PC12 cells as a model of Parkinson’s disease. Gene, 662, 54–65.
Friedlander, A. H., et al. (2009). Parkinson disease: Systemic and orofacial manifestations, medical and dental management. The Journal of the American Dental Association, 140(6), 658–669.
Santin, Y., et al. (2020). Mitochondrial 4-HNE derived from MAO-A promotes mitoCa2+ overload in chronic postischemic cardiac remodeling. Cell Death & Differentiation, 27(6), 1907–1923.
Pathania, A., Kumar, R., & Sandhir, R. (2021). Hydroxytyrosol as anti-parkinsonian molecule: Assessment using in-silico and MPTP-induced Parkinson’s disease model. Biomedicine & Pharmacotherapy, 139, 111525.
Binde, C. D., et al. (2018). A multiple treatment comparison meta-analysis of monoamine oxidase type B inhibitors for Parkinson’s disease. British Journal of Clinical Pharmacology, 84(9), 1917–1927.
Aksoz, B. E., & Aksoz, E. (2020). Vital role of monoamine oxidases and cholinesterases in central nervous system drug research: A sharp dissection of the pathophysiology. Combinatorial Chemistry & High Throughput Screening, 23(9), 877–886.
Löscher, W., & Potschka, H. (2005). Drug resistance in brain diseases and the role of drug efflux transporters. Nature Reviews Neuroscience, 6(8), 591–602.
Grygorenko, O. O., Volochnyuk, D. M., & Vashchenko, B. V. (2021). Emerging building blocks for medicinal chemistry: Recent synthetic advances. European Journal of Organic Chemistry, 2021(47), 6478–6510.
Haider, S., et al. (2014). Emerging pharmaceutical applications of piperidine, pyrrolidine and its derivatives. World Journal of Pharmaceutical Research, 3(7), 2277–7105.
Szczepańska, K., et al. (2021). Structural and molecular insight into piperazine and piperidine derivatives as histamine H3 and sigma-1 receptor antagonists with promising antinociceptive properties. ACS Chemical Neuroscience, 13(1), 1–15.
Nasab, N. H., et al. (2022). Coumarin-chalcones generated from 3-acetylcoumarin as a promising agent: Synthesis and pharmacological properties. ChemistrySelect, 7(11), e202200238.
Pisani, L., et al. (2016). Exploring basic tail modifications of coumarin-based dual acetylcholinesterase-monoamine oxidase B inhibitors: Identification of water-soluble, brain-permeant neuroprotective multitarget agents. Journal of Medicinal Chemistry, 59(14), 6791–6806.
Heo, J. H., et al. (2020). Acetylcholinesterase and butyrylcholinesterase inhibitory activities of khellactone coumarin derivatives isolated from Peucedanum japonicum Thurnberg. Scientific Reports, 10(1), 1–11.
Aljohani, F. S., et al. (2021). Structural inspection for novel Pd (II), VO (II), Zn (II) and Cr (III)-azomethine metal chelates: DNA interaction, biological screening and theoretical treatments. Journal of Molecular Structure, 1246, 131139.
Ejaz, S. A., et al. (2022). Identification of N-(4-acetyl-4, 5-dihydro-5-(7, 8, 9-substituted-tetrazolo [1, 5-a]-quinolin-4-yl)-1, 3, 4-thiadiazol-2-yl) acetamide derivatives as potential caspase-3 inhibitors via detailed computational investigations. Structural Chemistry, 34(2), 1–14.
Mahesar, P. A., et al. (2023). Potential role of hydrazinyl 1, 2, 4-triazoles derivatives as acetylcholinesterase inhibitors: Synthesis, biological evaluation, kinetics mechanism and molecular docking and simulation studies. Chemical Papers, 77(1), 1–13.
Nachon, F., et al. (2013). Crystal structures of human cholinesterases in complex with huprine W and tacrine: Elements of specificity for anti-Alzheimer’s drugs targeting acetyl-and butyryl-cholinesterase. Biochemical Journal, 453(3), 393–399.
Ahmed, M., et al. (2006). Inhibition of two different cholinesterases by tacrine. Chemico-Biological Interactions, 162(2), 165–171.
Son, S. (2002). Crystal structure of human monoamine oxidase A. Neuroscience, 114(4), 825–835.
Binda, C., et al. (2007). Structures of human monoamine oxidase B complexes with selective noncovalent inhibitors: Safinamide and coumarin analogs. Journal of Medicinal Chemistry, 50(23), 5848–5852.
Aziz, M., et al. (2022). Identification of potent inhibitors of NEK7 protein using a comprehensive computational approach. Scientific Reports, 12(1), 6404.
Ejaz, S. A., et al. (2022). In silico prospects and therapeutic applications of ouabagenin and hydroxylated corticosteroid analogues in the treatment of lung cancer. Applied Biochemistry and Biotechnology, 194(12), 6106–6125.
Aziz, M., et al. (2022). Deep learning and structure-based virtual screening for drug discovery against NEK7: A novel target for the treatment of cancer. Molecules, 27(13), 4098.
Ejaz, S. A., et al. (2022). 4-Phthalimidobenzenesulfonamide derivatives as acetylcholinesterase and butyrylcholinesterase inhibitors: DFTs, 3D-QSAR, ADMET, and molecular dynamics simulation. Neurodegenerative Diseases, 22(3–4), 1–17.
Ahmed, A., et al. (2022). Design, synthesis, kinetic analysis and pharmacophore-directed discovery of 3-ethylaniline hybrid imino-thiazolidinone as potential inhibitor of carbonic anhydrase II: An emerging biological target for treatment of cancer. Biomolecules, 12(11), 1696.
Khan, I., et al. (2015). Investigation of quinoline-4-carboxylic acid as a highly potent scaffold for the development of alkaline phosphatase inhibitors: Synthesis, SAR analysis and molecular modelling studies. RSC advances, 5(79), 64404–64413.
Channar, S. A., et al. (2022). Exploring thiazole-linked thioureas using alkaline phosphatase assay, biochemical evaluation, computational analysis and structure–activity relationship (SAR) studies. Medicinal Chemistry Research, 31(10), 1792–1802.
Hansson, T., Oostenbrink, C., & van Gunsteren, W. (2002). Molecular dynamics simulations. Current opinion in structural biology, 12(2), 190–196.
Deb, S., & Reeves, A. A. (2021). Simulation of remdesivir pharmacokinetics and its drug interactions. Journal of Pharmacy and Pharmaceutical Sciences., 24, 277–291.
Acknowledgements
We acknowledge Al Ain University for providing facilities and resources to rum the in silico simulation using Gastro plus Software.
Funding
Arab Emirates University for awarding Research Start-up grant No.: 700032854.
Author information
Authors and Affiliations
Contributions
M.S.: methodology, investigations. M.K.I.: methodology, experimental material design, investigations. S.A.E..: conceptualization, methodology, supervision, investigation, writing review and editing. M.A.: methodology, experimental material design, investigations. H. M.A.: methodology, investigations, writing review and editing. M.A.: investigation, writing-review and editing. T.S.: methodology, formal analysis. T.R.: investigation, writing-review and editing. H.K.M.: investigation, writing-review and editing. M.E.: revision and resources. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethical Approval
Not applicable
Consent to Participate
Not applicable
Consent for Publication
All authors agreed for the publication of the final version of the manuscript.
Competing Interests
All the authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sarfraz, M., Ibrahim, M.K., Ejaz, S.A. et al. An Integrated Computational Approaches for Designing of Potential Piperidine based Inhibitors of Alzheimer Disease by Targeting Cholinesterase and Monoamine Oxidases Isoenzymes. Appl Biochem Biotechnol (2024). https://doi.org/10.1007/s12010-023-04815-0
Accepted:
Published:
DOI: https://doi.org/10.1007/s12010-023-04815-0