Abstract
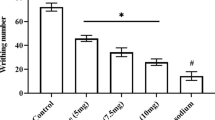
Pain management has been a severe public health issue throughout the world. Acute pain if not treated at the appropriate time can lead to chronic pain that can cause psychological and social distress. Nothing can be more rewarding than treating pain successfully for a physician. However, the use of chemical NSAIDs and opiate drugs has taken a toll on the patients with their unavoidable side effects. This study intends to explore the potential to treat pain by inhibiting nociception and inflammation with a safer, non-addictive, effective, and low-cost alternative agent from a natural source, visnagin. In vivo studies have been conducted using male Swiss albino mice as models for this research. Nociception was induced using different chemical and thermal stimuli such as acetic acid, glutamate, capsaicin, and formalin. To check for the anti-inflammatory properties, carrageenan was used to induce inflammation and the activity was assayed using peritoneal cavity leukocyte infiltration analysis and pro-inflammatory cytokine level comparison with the supplementation of visnagin at three different dosages. The findings of this study revealed that the visnagin treatment effectively attenuated the acetic acid-induced writhing response, glutamate-induced paw licking numbers, capsaicin-induced pain response, and formalin-induced biphasic licking incidences in the experimental mice models. Furthermore, the visnagin treatment remarkably suppressed the carrageenan-induced inflammation in mice, which is evident from the decreased leukocytes, mononuclear, and polymorphonuclear cell numbers in the mice. The levels of cytokines such as TNF-α, IL-1β, and IL-6 were effectively reduced by the visnagin treatment in the experimental mice. The results of open field test proved that the visnagin showed a better locomotor movement in the experimental mice. These results provided evidence for the potential activity of the visnagin against inflammatory and nociceptive responses in the mice.








Similar content being viewed by others
Data Availability
Not applicable
References
Carr, F. B., & Zachariou, V. (2014). Nociception and pain: Lessons from optogenetics. Frontiers in Behavioral Neuroscience, 8(69), 1–6.
Dubin, A. E., & Patapoutian, A. (2010). Nociceptors: The sensors of the pain pathway. The Journal of Clinical Investigation, 120(11), 3760–3772.
Fürst, S. (1999). Transmitters involved in antinociception in the spinal cord. Brain Research Bulletin, 48(2), 129–141.
Cervero, F., & Merskey, H. (1996). What is a noxious stimulus? Pain Forum, 5(3), 157–161.
Omoigui, S. (2007). The biochemical origin of pain: The origin of all pain is inflammation and the inflammatory response. Part 2 of 3–Inflammatory profile of pain syndromes. Medical Hypotheses, 69, 1169–1178.
Baron, R., Maier, C., & Attal, N. (2017). Peripheral neuropathic pain: A mechanism-related organizing principle based on sensory profiles. Pain, 158(2), 261–272.
Kim, K.-H., Seo, H.-J., Abdi, S., & Huh, B. (2020). All about pain pharmacology: What pain physicians should know. Korean J Pain, 33(2), 108–120.
Maroon, J. C., Bost, J. W., & Maroon, A. (2010). Natural anti-inflammatory agents for pain relief. Surgical Neurology International, 1, 80.
Li, J.W.-H., & Vederas, J. C. (2009). Drug discovery and natural products: End of an era or an endless frontier? Science, 325, 161–165.
Parveen, A., Kim, J. H., Oh, B. G., Subedi, L., Khan, Z., & Kim, S. Y. (2018). Phytochemicals: Target-based therapeutic strategies for diabetic retinopathy. Molecules, 23(7), 1519.
Akram, M., Asif, H. M., & Usmanghani, K. (2013). Anti-nociceptive activities of medicinal plants: A review. Journal of Pharmacy and Pharmaceutical Sciences, 4(1), 50–58.
Vanachayangkul, P., Byer, K., Khan, S., & Butterweck, V. (2010). An aqueous extract of Ammi visnaga fruits and its constituents khellin and visnagin prevent cell damage caused by oxalate in renal epithelial cells. Phytomedicine, 17, 653–658.
Bhagavathula, A. S., Al-Khatib, A. J. M., & Elnour, A. A. (2015). Ammi Visnaga in treatment of urolithiasis and hypertriglyceridemia. Pharmacognosy Research, 7(4), 397–400.
Rauwald, H. W., Brehm, O., & Odenthal, K. P. (1994). The involvement of a Ca2+ channel blocking mode of action in the pharmacology of Ammi visnaga fruits. Planta Medica, 60, 101–105.
Duarte, J., Perez-Vizcaino, F., Torres, A. I., Zarzuelo, A., Jimenez, J., & Tamargo, J. (1995). Vasodilator effects of visnagin in isolated rat vascular smooth muscle. European Journal of Pharmacology, 286, 115–122.
Hassan, J., Mhamed, M., & Mohammed, E. (2002). Hypoglycemic effect of aqueous extract of Ammi visnaga in normal and streptozotocin-induced diabetic rats. Journal of Herbal Pharmacotherapy, 2, 19–29.
Lee, J., Jung, J., Park, S., Sim, Y., Kim, S., Ha, T., & Suh, H. (2010). Anti-inflammatory effect of visnagin in lipopolysaccharide-stimulated BV-2 microglial cells. Archives of Pharmacal Research, 33, 1843–1850.
Kwon, M., Lee, J., Park, S., Sim, Y., Jung, J., Won, M., Kim, S., & Suh, H. (2010). Neuroprotective effect of visnagin on kainic acid-induced neuronal cell death in the mice hippocampus. The Korean Journal of Physiology and Pharmacology, 14, 257–263.
Duarte, J., Torres, A. I., & Zarzuelo, A. (2000). Cardiovascular effects of visnagin on rats. Planta Medica, 66, 35–39.
Khalil, N., Bishr, M., Desouky, S., & Salama, O. (2020). Ammi Visnaga L., A potential medicinal plant: A review. Molecules, 25, 301.
Koster, R., Anderson, M., & de Beer, E. F. (1952). Acetic acid for analgesic screening. Federation Proceedings, 18, 412.
Beirith, A., Santos, A. R., & Calixto, J. B. (2002). Mechanisms underlying the nociception and paw oedema caused by injection of glutamate into the mouse paw. Brain Research, 924(2), 219–228.
Luiz, A. P., Moura, J. D., Meotti, F. C., Guginski, G., Guimaraes, C. L., Azevedo, M. S., Rodrigues, A. L., & Santos, A. R. (2007). Antinociceptive action of ethanolic extract obtained from roots of Humirianthera ampla Miers. Journal of Ethnopharmacology, 114(3), 355–363.
Gomes, N. M., Rezende, C. M., Fontes, S. P., Matheus, M. E., & Fernandes, P. D. (2007). Antinociceptive activity of Amazonian Copaiba oils. Journal of Ethnopharmacology, 109(3), 486–492.
Vinegar, R., Truax, J. F., & Selph, J. L. (1973). Some quantitative temporal characteristics of carrageenan-induced pleurisy in the rat. Proceedings of the Society for Experimental Biology and Medicine, 143(3), 711–714.
Edwards, J. C., Sedgwick, A. D., & Willoughby, D. A. (1981). The formation of a structure with the features of synovial lining by subcutaneous injection of air: An in vivo tissue culture system. The Journal of Pathology, 134(2), 147–156.
De Mattos, E. S., Frederico, M. J., Colle, T. D., De Pieri, D. V., Peters, R. R., & Piovezan, A. P. (2007). Evaluation of antinociceptive activity of Casearia sylvestris and possible mechanism of action. Journal of Ethnopharmacology, 112(1), 1–6.
Gonzalez-Velasco, H. E., Perez-Gutierrez, M., Alonso-Castro, A. J., Zapata-Morales, J. R., Nino-Moreno, P. C., Campos-Xolalpa, N., & Gonzalez-Chavez, M. M. (2022). Anti-inflammatory and antinociceptive activities of the essential oil of Tagetes parryi A. Gray (Asteraceae) and Verbenone. Molecules, 27(9), 2612.
Wang, M., & Thyagarajan, B. (2022). Pain pathways and potential new targets for pain relief. Biotechnology and Applied Biochemistry, 69(1), 110–123.
Gold, M. S., & Gebhart, G. F. (2010). Nociceptor sensitization in pain pathogenesis. Nature Medicine, 16(11), 1248–57.
Cashman, J. N. (1996). The mechanisms of action of NSAIDs in analgesia. Drugs, 52(Suppl. 5), 13–23.
Su, L. Y., Liu, Q., Jiao, L., & Yao, Y. G. (2021). Molecular mechanism of neuroprotective effect of melatonin on morphine addiction and analgesic tolerance: An update. Molecular Neurobiology, 58(9), 4628–4638.
Veeresham, C. (2012). Natural products derived from plants as a source of drugs. Journal of Advanced Pharmaceutical Technology & Research, 3, 200–201.
Taur, D. J., Waghmare, M. G., Bandal, R. S., & Patil, R. Y. (2011). Antinociceptive activity of Ricinus communis L. leaves. Asian Pacific Journal of Tropical Biomedicine, 1(2), 139–41. https://doi.org/10.1016/S2221-1691(11)60012-9
Abu-Hashem, A. A., & Youssef, M. M. (2011). Synthesis of new visnagen and khellin furochromone pyrimidine derivatives and their anti-inflammatory and analgesic activity. Molecules, 16, 1956–1972.
Fundytus, M. E. (2001). Glutamate receptors and nociception implications for the drug treatment of pain. CNS Drugs, 15(1), 29–58.
Frias, B., & Merighi, A. (2016). Review capsaicin, nociception and pain. Molecules, 21, 797.
Karim, N., Khan, I., Khan, W., Khan, I., Khan, A., Halim, S. A., Khan, H., Hussain, J., & Al-Harrasi, A. (2019). Anti-nociceptive and anti-inflammatory activities of asparacosin a involve selective cyclooxygenase 2 and inflammatory cytokines inhibition: An in-vitro, in-vivo, and in-silico approach. Frontiers in Immunology, 26(10), 581.
Lee, J. K., Jung, J. S., Park, S. H., Park, S. H., Sim, Y. B., Kim, S. M., Ha, T. S., & Suh, H. W. (2010). Anti-inflammatory effect of visnagin in lipopolysaccharide-stimulated BV-2 microglial cells. Archives of Pharmacal Research, 33(11), 1843–1850.
El-Sawy, E. R., Ebaid, M. S., Abo-Salem, H. M., Al-Sehemi, A. G., & Mandour, A. H. (2014). Synthesis, anti-inflammatory, analgesic and anticonvulsant activities of some new 4,6-dimethoxy-5-(heterocycles) benzofuran starting from naturally occurring visnagin. Arabian Journal of Chemistry, 7(6), 914–923.
Khalil, H. S., Sedky, N. K., Amin, K. M., Abd Elhafez, O. M., & Arafa, R. K. (2019). Visnagin and benzofuran scaffold-based molecules as selective cyclooxygenase-2 inhibitors with anti-inflammatory and analgesic properties: Design, synthesis and molecular docking. Future Medicinal Chemistry, 11(7), 659–676.
Author information
Authors and Affiliations
Contributions
Xiaobing Qi and Sattam Khulaif Alenezi drafted the manuscript; Ibrahim M. Alanazi and Mohammed S. Alshammari drafted the manuscript; Ibrahim Abdel Aziz Ibrahim and Kalaivani Aiyasamy conceptualized and corrected/revised the manuscript.
Corresponding author
Ethics declarations
Ethics Approval
All the animal experiments were done under the guidelines of the Institutional Animal Ethics Committee (Animal ethical approval number: IMBH20211128).
Consent to Participate
All authors have their consent to participate.
Consent for Publication
All authors have their consent to publish their work.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Qi, X., Aiyasamy, K., Alenezi, S.K. et al. Anti-nociceptive and Anti-inflammatory Activities of Visnagin in Different Nociceptive and Inflammatory Mice Models. Appl Biochem Biotechnol (2023). https://doi.org/10.1007/s12010-023-04677-6
Accepted:
Published:
DOI: https://doi.org/10.1007/s12010-023-04677-6