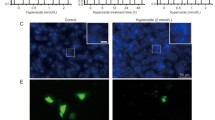
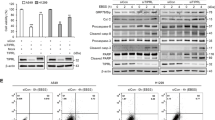
Abstract
The pathophysiology of lung cancer is dependent on the dysregulation in the apoptotic and autophagic pathways. The intricate link between apoptosis and autophagy through shared signaling pathways complicates our understanding of how lung cancer pathophysiology is regulated. As drug resistance is the primary reason behind treatment failure, it is crucial to understand how cancer cells may respond to different therapies and integrate crosstalk between apoptosis and autophagy in response to them, leading to cell death or survival. Thus, in this study, we have tried to evaluate the crosstalk between autophagy and apoptosis in A549 lung cancer cell line that could be modulated by employing a combination therapy of metformin (6 mM), an anti-diabetic drug, with gedunin (12 µM), an Hsp90 inhibitor, to provide insights into the development of new cancer therapeutics. Our results demonstrated that metformin and gedunin were cytotoxic to A549 lung cancer cells. Combination of metformin and gedunin generated ROS and promoted MMP loss and DNA damage. The combination further increased the expression of AMPKα1 and promoted the nuclear localization of AMPKα1/α2. The expression of Hsp90 was downregulated, further decreasing the expression of its clients, EGFR, PIK3CA, AKT1, and AKT3. Inhibition of the EGFR/PI3K/AKT pathway upregulated TP53 and inhibited autophagy. The combination was promoting nuclear localization of p53; however, some cytoplasmic signals were also detected. Further increase in the expression of caspase 9 and caspase 3 was observed. Thus, we concluded that the combination of metformin and gedunin upregulates apoptosis by inhibiting the EGFR/PI3K/AKT pathway and autophagy in A549 lung cancer cells.














Similar content being viewed by others
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
AACR Project GENIE Consortium. (2017). AACR Project GENIE: Powering Precision Medicine through an International Consortium. Cancer discovery, 7(8), 818–831. https://doi.org/10.1158/2159-8290.CD-17-0151
Anderson, E., Havener, T. M., Zorn, K. M., Foil, D. H., Lane, T. R., Capuzzi, S. J., Morris, D., Hickey, A. J., Drewry, D. H., & Ekins, S. (2020). Synergistic drug combinations and machine learning for drug repurposing in chordoma. Scientific reports, 10(1), 12982. https://doi.org/10.1038/s41598-020-70026-w
Arias, E., & Cuervo, A. M. (2011). Chaperone-mediated autophagy in protein quality control. Current Opinion in Cell Biology, 23(2), 184–189. https://doi.org/10.1016/j.ceb.2010.10.009
Bayat Mokhtari, R., Homayouni, T. S., Baluch, N., Morgatskaya, E., Kumar, S., Das, B., & Yeger, H. (2017). Combination therapy in combating cancer. Oncotarget, 8(23), 38022–38043. https://doi.org/10.18632/oncotarget.16723
Ben Sahra, I., Regazzetti, C., Robert, G., Laurent, K., Le Marchand-Brustel, Y., Auberger, P., Tanti, J. F., Giorgetti-Peraldi, S., & Bost, F. (2011). Metformin, independent of AMPK, induces mTOR inhibition and cell-cycle arrest through REDD1. Cancer research, 71(13), 4366–4372. https://doi.org/10.1158/0008-5472.CAN-10-1769
Bolte, S., & Cordelières, F. P. (2006). A guided tour into subcellular colocalization analysis in light microscopy. Journal of Microscopy, 224(Pt 3), 213–232. https://doi.org/10.1111/j.1365-2818.2006.01706.x
Bridges, H. R., Jones, A. J., Pollak, M. N., & Hirst, J. (2014). Effects of metformin and other biguanides on oxidative phosphorylation in mitochondria. The Biochemical journal, 462(3), 475–487. https://doi.org/10.1042/BJ20140620
Budanov, A. V., & Karin, M. (2008). p53 target genes sestrin1 and sestrin2 connect genotoxic stress and mTOR signaling. Cell, 134(3), 451–460. https://doi.org/10.1016/j.cell.2008.06.028
Cai, X., & Liu, X. (2008). Inhibition of Thr-55 phosphorylation restores p53 nuclear localization and sensitizes cancer cells to DNA damage. Proceedings of the National Academy of Sciences USA, 105(44), 16958–16963. https://doi.org/10.1073/pnas.0804608105
Chen, K., Qian, W., Li, J., Jiang, Z., Cheng, L., Yan, B., Cao, J., Sun, L., Zhou, C., Lei, M., Duan, W., Ma, J., Ma, Q., & Ma, Z. (2017). Loss of AMPK activation promotes the invasion and metastasis of pancreatic cancer through an HSF1-dependent pathway. Molecular Oncology, 11(10), 1475–1492. https://doi.org/10.1002/1878-0261.12116
Chen, D., Wang, Y., Wu, K., & Wang, X. (2018). Dual effects of metformin on adipogenic differentiation of 3T3-L1 preadipocyte in AMPK-dependent and independent manners. International Journal of Molecular Sciences, 19(6), 1547. https://doi.org/10.3390/ijms19061547
Cheng, M. H., Huang, H. L., Lin, Y. Y., Tsui, K. H., Chen, P. C., Cheng, S. Y., Chong, I. W., Sung, P. J., Tai, M. H., Wen, Z. H., Chen, N. F., & Kuo, H. M. (2019). BA6 induces apoptosis via stimulation of reactive oxygen species and inhibition of oxidative phosphorylation in human lung cancer cells. Oxidative Medicine and Cellular Longevity, 2019, 6342104. https://doi.org/10.1155/2019/6342104
Cho, D. H., Jo, Y. K., Hwang, J. J., Lee, Y. M., Roh, S. A., & Kim, J. C. (2009). Caspase-mediated cleavage of ATG6/Beclin-1 links apoptosis to autophagy in HeLa cells. Cancer Letters, 274(1), 95–100. https://doi.org/10.1016/j.canlet.2008.09.004
Chou, T. C. (2006). Theoretical basis, experimental design, and computerized simulation of synergism and antagonism in drug combination studies. Pharmacological Reviews, 58(3), 621–681. https://doi.org/10.1124/pr.58.3.10
Chou, T. C. (2010). Drug combination studies and their synergy quantification using the Chou-Talalay method. Cancer Research, 70(2), 440–446. https://doi.org/10.1158/0008-5472.CAN-09-1947
Ciocca, D. R., & Calderwood, S. K. (2005). Heat shock proteins in cancer: Diagnostic, prognostic, predictive, and treatment implications. Cell Stress & Chaperones, 10(2), 86–103. https://doi.org/10.1379/csc-99r.1
Colell, A., Ricci, J. E., Tait, S., Milasta, S., Maurer, U., Bouchier-Hayes, L., Fitzgerald, P., Guio-Carrion, A., Waterhouse, N. J., Li, C. W., Mari, B., Barbry, P., Newmeyer, D. D., Beere, H. M., & Green, D. R. (2007). GAPDH and autophagy preserve survival after apoptotic cytochrome c release in the absence of caspase activation. Cell, 129(5), 983–997. https://doi.org/10.1016/j.cell.2007.03.045
Corazzari, M., Gagliardi, M., Fimia, G. M., & Piacentini, M. (2017). Endoplasmic reticulum stress, unfolded protein response, and cancer cell fatE. Frontiers in Oncology, 7, 78. https://doi.org/10.3389/fonc.2017.00078
Cordelieres, F. P., Bolte, S. (2008). JACoP v2. 0: Improving the user experience with co-localization studies. Proceedings of the 2nd ImageJ user and developer conference pp. 6–7.
Daugan, M., Dufaÿ Wojcicki, A., d’Hayer, B., & Boudy, V. (2016). Metformin: An anti-diabetic drug to fight cancer. Pharmacological Research, 113(Pt A), 675–685. https://doi.org/10.1016/j.phrs.2016.10.006
Deng, J., Peng, M., Wang, Z., Zhou, S., Xiao, D., Deng, J., Yang, X., Peng, J., & Yang, X. (2019). Novel application of metformin combined with targeted drugs on anticancer treatment. Cancer Science, 110(1), 23–30. https://doi.org/10.1111/cas.13849
Dice, J. F. (2007). Chaperone-mediated autophagy. Autophagy, 3(4), 295–299. https://doi.org/10.4161/auto.4144
Dillon, R. L., Marcotte, R., Hennessy, B. T., Woodgett, J. R., Mills, G. B., & Muller, W. J. (2009). Akt1 and akt2 play distinct roles in the initiation and metastatic phases of mammary tumor progression. Cancer Research, 69(12), 5057–5064. https://doi.org/10.1158/0008-5472.CAN-08-4287
Djavaheri-Mergny, M., Maiuri, M. C., & Kroemer, G. (2010). Cross talk between apoptosis and autophagy by caspase-mediated cleavage of Beclin 1. Oncogene, 29(12), 1717–1719. https://doi.org/10.1038/onc.2009.519
Duan, W., Chen, K., Jiang, Z., Chen, X., Sun, L., Li, J., Lei, J., Xu, Q., Ma, J., Li, X., Han, L., Wang, Z., Wu, Z., Wang, F., Wu, E., Ma, Q., & Ma, Z. (2017). Desmoplasia suppression by metformin-mediated AMPK activation inhibits pancreatic cancer progression. Cancer Letters, 385, 225–233. https://doi.org/10.1016/j.canlet.2016.10.019
Dunn, C. J., & Peters, D. H. (1995). Metformin. A review of its pharmacological properties and therapeutic use in non-insulin-dependent diabetes mellitus. Drugs, 49(5), 721–749. https://doi.org/10.2165/00003495-199549050-00007
Durand, P. M., & Ramsey, G. (2019). The nature of programmed cell death. Biological Theory, 14(1), 30–41. https://doi.org/10.1007/s13752-018-0311-0
Fan, T. J., Han, L. H., Cong, R. S., & Liang, J. (2005). Caspase family proteases and apoptosis. Acta Biochimica et Biophysica Sinica (Shanghai), 37(11), 719–727. https://doi.org/10.1111/j.1745-7270.2005.00108.x
Fang, C., Gu, L., Smerin, D., Mao, S., & Xiong, X. (2017). The interrelation between reactive oxygen species and autophagy in neurological disorders. Oxidative Medicine and Cellular Longevity, 2017, 8495160. https://doi.org/10.1155/2017/8495160
Fato, R., Bergamini, C., Bortolus, M., Maniero, A. L., Leoni, S., Ohnishi, T., & Lenaz, G. (2009). Differential effects of mitochondrial Complex I inhibitors on production of reactive oxygen species. Biochimica et Biophysica Acta, 1787(5), 384–392. https://doi.org/10.1016/j.bbabio.2008.11.003
Gong, Y., Wang, C., Jiang, Y., Zhang, S., Feng, S., Fu, Y., & Luo, Y. (2020). Metformin inhibits tumor metastasis through suppressing Hsp90α secretion in an AMPKα1-PKCγ dependent manner. Cells, 9(1), 144. https://doi.org/10.3390/cells9010144
Gorrini, C., Harris, I. S., & Mak, T. W. (2013). Modulation of oxidative stress as an anticancer strategy. Nature Reviews Drug Discovery, 12(12), 931–947. https://doi.org/10.1038/nrd4002
Grabinski, N., Bartkowiak, K., Grupp, K., Brandt, B., Pantel, K., & Jücker, M. (2011). Distinct functional roles of Akt isoforms for proliferation, survival, migration and EGF-mediated signalling in lung cancer derived disseminated tumor cells. Cellular Signalling, 23(12), 1952–1960. https://doi.org/10.1016/j.cellsig.2011.07.003
Green, D. R., & Kroemer, G. (2009). Cytoplasmic functions of the tumour suppressor p53. Nature, 458(7242), 1127–1130. https://doi.org/10.1038/nature07986
Hardie, D. G., Schaffer, B. E., & Brunet, A. (2016). AMPK: An energy-sensing pathway with multiple inputs and outputs. Trends in Cell Biology, 26(3), 190–201. https://doi.org/10.1016/j.tcb.2015.10.013
Hasan, A., Haque, E., Hameed, R., Maier, P. N., Irfan, S., Kamil, M., Nazir, A., & Mir, S. S. (2020). Hsp90 inhibitor gedunin causes apoptosis in A549 lung cancer cells by disrupting Hsp90:Beclin-1:Bcl-2 interaction and downregulating autophagy. Life sciences, 256, 118000. https://doi.org/10.1016/j.lfs.2020.118000
He, G., Zhang, Y. W., Lee, J. H., Zeng, S. X., Wang, Y. V., Luo, Z., Dong, X. C., Viollet, B., Wahl, G. M., & Lu, H. (2014). AMP-activated protein kinase induces p53 by phosphorylating MDMX and inhibiting its activity. Molecular and Cellular Biology, 34(2), 148–157. https://doi.org/10.1128/MCB.00670-13
Hunziker, A., Jensen, M. H., & Krishna, S. (2010). Stress-specific response of the p53-Mdm2 feedback loop. BMC Systems Biology, 4, 94. https://doi.org/10.1186/1752-0509-4-94
Jafari, A., Rezaei-Tavirani, M., Farhadihosseinabadi, B., Taranejoo, S., & Zali, H. (2020). HSP90 and co-chaperones: Impact on tumor progression and prospects for molecular-targeted cancer therapy. Cancer Investigation, 38(5), 310–328. https://doi.org/10.1080/07357907.2020.1752227
Jhaveri, T. Z., Woo, J., Shang, X., Park, B. H., & Gabrielson, E. (2015). AMP-activated kinase (AMPK) regulates activity of HER2 and EGFR in breast cancer. Oncotarget, 6(17), 14754–14765. https://doi.org/10.18632/oncotarget.4474
Ji, L. L., & Mitchell, E. W. (1994). Effects of adriamycin on heart mitochondrial function in rested and exercised rats. Biochemical Pharmacology, 47(5), 877–885. https://doi.org/10.1016/0006-2952(94)90488-x
Jiang, B. H., & Liu, L. Z. (2009). PI3K/PTEN signaling in angiogenesis and tumorigenesis. Advances in Cancer Research, 102, 19–65. https://doi.org/10.1016/S0065-230X(09)02002-8
Jin, J., Xiong, Y., & Cen, B. (2017). Bcl-2 and Bcl-xL mediate resistance to receptor tyrosine kinase-targeted therapy in lung and gastric cancer. Anti-Cancer Drugs, 28(10), 1141–1149. https://doi.org/10.1097/CAD.0000000000000561
Jones, R. G., Plas, D. R., Kubek, S., Buzzai, M., Mu, J., Xu, Y., Birnbaum, M. J., & Thompson, C. B. (2005). AMP-activated protein kinase induces a p53-dependent metabolic checkpoint. Molecular Cell, 18(3), 283–293. https://doi.org/10.1016/j.molcel.2005.03.027
Ju, T. C., Chen, H. M., Lin, J. T., Chang, C. P., Chang, W. C., Kang, J. J., Sun, C. P., Tao, M. H., Tu, P. H., Chang, C., Dickson, D. W., & Chern, Y. (2011). Nuclear translocation of AMPK-alpha1 potentiates striatal neurodegeneration in Huntington’s disease. The Journal of Cell Biology, 194(2), 209–227. https://doi.org/10.1083/jcb.201105010
Kabir, M. F., Kim, H.-R., Han-Jung Chae, H.-J. (2018) Endoplasmic reticulum stress and autophagy. Endoplasmic Reticulum. IntechOpen. https://dpi.org/10.5772/intechopen.81381
Kamil, M., Haque, E., Irfan, S., Sheikh, S., Hasan, A., Nazir, A., Lohani, M., & Mir, S. S. (2017). ER chaperone GRP78 regulates autophagy by modulation of p53 localization. Frontiers in Bioscience (Elite edition), 9, 54–66. https://doi.org/10.2741/e785
Kaniak-Golik, A., & Skoneczna, A. (2015). Mitochondria-nucleus network for genome stability. Free Radical Biology & Medicine, 82, 73–104. https://doi.org/10.1016/j.freeradbiomed.2015.01.013
Kato, K., Gong, J., Iwama, H., Kitanaka, A., Tani, J., Miyoshi, H., Nomura, K., Mimura, S., Kobayashi, M., Aritomo, Y., Kobara, H., Mori, H., Himoto, T., Okano, K., Suzuki, Y., Murao, K., & Masaki, T. (2012). The antidiabetic drug metformin inhibits gastric cancer cell proliferation in vitro and in vivo. Molecular Cancer Therapeutics, 11(3), 549–560. https://doi.org/10.1158/1535-7163.MCT-11-0594
Kenyon, C. J. (2010). The genetics of ageing. Nature, 464(7288), 504–512. https://doi.org/10.1038/nature08980
Khurana, N., & Bhattacharyya, S. (2015). Hsp90, the concertmaster: Tuning transcription. Frontiers in Oncology, 5, 100. https://doi.org/10.3389/fonc.2015.00100
Kirkegaard, T., Witton, C. J., Edwards, J., Nielsen, K. V., Jensen, L. B., Campbell, F. M., Cooke, T. G., & Bartlett, J. M. (2010). Molecular alterations in AKT1, AKT2 and AKT3 detected in breast and prostatic cancer by FISH. Histopathology, 56(2), 203–211. https://doi.org/10.1111/j.1365-2559.2009.03467.x
Klionsky, D. J., & Emr, S. D. (2000). Autophagy as a regulated pathway of cellular degradation. Science (New York, N.Y.), 290(5497), 1717–1721. https://doi.org/10.1126/science.290.5497.1717
Kodiha, M., Rassi, J. G., Brown, C. M., & Stochaj, U. (2007). Localization of AMP kinase is regulated by stress, cell density, and signaling through the MEK–>ERK1/2 pathway. American Journal of Physiology Cell Physiology, 293(5), C1427–C1436. https://doi.org/10.1152/ajpcell.00176.2007
Koga, H., Martinez-Vicente, M., Arias, E., Kaushik, S., Sulzer, D., & Cuervo, A. M. (2011). Constitutive upregulation of chaperone-mediated autophagy in Huntington’s disease. The Journal of Neuroscience : The official journal of the Society for Neuroscience, 31(50), 18492–18505. https://doi.org/10.1523/JNEUROSCI.3219-11.2011
Kubbutat, M. H., Jones, S. N., & Vousden, K. H. (1997). Regulation of p53 stability by Mdm2. Nature, 387(6630), 299–303. https://doi.org/10.1038/387299a0
Lee, M. W., Kim, D. S., Lee, J. H., Lee, B. S., Lee, S. H., Jung, H. L., Sung, K. W., Kim, H. T., Yoo, K. H., & Koo, H. H. (2011). Roles of AKT1 and AKT2 in non-small cell lung cancer cell survival, growth, and migration. Cancer Science, 102(10), 1822–1828. https://doi.org/10.1111/j.1349-7006.2011.02025.x
Lev Bar-Or, R., Maya, R., Segel, L. A., Alon, U., Levine, A. J., & Oren, M. (2000). Generation of oscillations by the p53-Mdm2 feedback loop: A theoretical and experimental study. Proceedings of the National Academy of Sciences of the United States of America, 97(21), 11250–11255. https://doi.org/10.1073/pnas.210171597
Li, J., Ni, M., Lee, B., Barron, E., Hinton, D. R., & Lee, A. S. (2008). The unfolded protein response regulator GRP78/BiP is required for endoplasmic reticulum integrity and stress-induced autophagy in mammalian cells. Cell Death and Differentiation, 15(9), 1460–1471. https://doi.org/10.1038/cdd.2008.81
Li, P., Zhou, L., Zhao, T., Liu, X., Zhang, P., Liu, Y., Zheng, X., & Li, Q. (2017). Caspase-9: Structure, mechanisms and clinical application. Oncotarget, 8(14), 23996–24008. https://doi.org/10.18632/oncotarget.15098
Lianos, G. D., Alexiou, G. A., Mangano, A., Mangano, A., Rausei, S., Boni, L., Dionigi, G., & Roukos, D. H. (2015). The role of heat shock proteins in cancer. Cancer Letters, 360(2), 114–118. https://doi.org/10.1016/j.canlet.2015.02.026
Lin, S. C., & Hardie, D. G. (2018). AMPK: Sensing glucose as well as cellular energy status. Cell Metabolism, 27(2), 299–313. https://doi.org/10.1016/j.cmet.2017.10.009
Linnerth-Petrik, N. M., Santry, L. A., Moorehead, R., Jücker, M., Wootton, S. K., & Petrik, J. (2016). Akt isoform specific effects in ovarian cancer progression. Oncotarget, 7(46), 74820–74833. https://doi.org/10.18632/oncotarget.11204
Liu, K., Kang, M., Li, J., Qin, W., & Wang, R. (2019). Prognostic value of the mRNA expression of members of the HSP90 family in non-small cell lung cancer. Experimental and Therapeutic Medicine, 17(4), 2657–2665. https://doi.org/10.3892/etm.2019.7228
Liu, Z., Ding, Y., Ye, N., Wild, C., Chen, H., & Zhou, J. (2016). Direct activation of Bax protein for cancer therapy. Medicinal Research Reviews, 36(2), 313–341. https://doi.org/10.1002/med.21379
Lu, C. C., Chiang, J. H., Tsai, F. J., Hsu, Y. M., Juan, Y. N., Yang, J. S., & Chiu, H. Y. (2019). Metformin triggers the intrinsic apoptotic response in human AGS gastric adenocarcinoma cells by activating AMPK and suppressing mTOR/AKT signaling. International Journal of Oncology, 54(4), 1271–1281. https://doi.org/10.3892/ijo.2019.4704
Luo, S., & Rubinsztein, D. C. (2010). Apoptosis blocks Beclin 1-dependent autophagosome synthesis: An effect rescued by Bcl-xL. Cell Death and Differentiation, 17(2), 268–277. https://doi.org/10.1038/cdd.2009.121
Luo, Z., Zang, M., & Guo, W. (2010). AMPK as a metabolic tumor suppressor: Control of metabolism and cell growth. Future Oncology (London, England), 6(3), 457–470. https://doi.org/10.2217/fon.09.174
Ma, S. B., Nguyen, T. N., Tan, I., Ninnis, R., Iyer, S., Stroud, D. A., Menard, M., Kluck, R. M., Ryan, M. T., & Dewson, G. (2014). Bax targets mitochondria by distinct mechanisms before or during apoptotic cell death: A requirement for VDAC2 or Bak for efficient Bax apoptotic function. Cell Death and Differentiation, 21(12), 1925–1935. https://doi.org/10.1038/cdd.2014.119
Mariño, G., Niso-Santano, M., Baehrecke, E. H., & Kroemer, G. (2014). Self-consumption: The interplay of autophagy and apoptosis. Nature Reviews Molecular Cell Biology, 15(2), 81–94. https://doi.org/10.1038/nrm3735
McComb, S., Chan, P. K., Guinot, A., Hartmannsdottir, H., Jenni, S., Dobay, M. P., Bourquin, J. P., & Bornhauser, B. C. (2019). Efficient apoptosis requires feedback amplification of upstream apoptotic signals by effector caspase-3 or -7. Science Advances, 5(7), eaau9433. https://doi.org/10.1126/sciadv.aau9433
McIlwain, D. R., Berger, T., & Mak, T. W. (2015). Caspase functions in cell death and disease. Cold Spring Harbor Perspectives in Biology, 7(4), a026716. https://doi.org/10.1101/cshperspect.a026716
Menon, M. B., & Dhamija, S. (2018). Beclin 1 Phosphorylation - At the Center of Autophagy Regulation. Frontiers in Cell and Developmental Biology, 6, 137. https://doi.org/10.3389/fcell.2018.00137
Mogavero, A., Maiorana, M. V., Zanutto, S., Varinelli, L., Bozzi, F., Belfiore, A., Volpi, C. C., Gloghini, A., Pierotti, M. A., & Gariboldi, M. (2017). Metformin transiently inhibits colorectal cancer cell proliferation as a result of either AMPK activation or increased ROS production. Scientific Reports, 7(1), 15992. https://doi.org/10.1038/s41598-017-16149-z
Moretti, L., Cha, Y. I., Niermann, K. J., & Lu, B. (2007). Switch between apoptosis and autophagy: Radiation-induced endoplasmic reticulum stress? Cell cycle (Georgetown, Tex.), 6(7), 793798. https://doi.org/10.4161/cc.6.7.4036
Mori, M., Hitora, T., Nakamura, O., Yamagami, Y., Horie, R., Nishimura, H., & Yamamoto, T. (2015). Hsp90 inhibitor induces autophagy and apoptosis in osteosarcoma cells. International Journal of Oncology, 46(1), 47–54. https://doi.org/10.3892/ijo.2014.2727
Mukhopadhyay, S., Panda, P. K., Sinha, N., Das, D. N., & Bhutia, S. K. (2014). Autophagy and apoptosis: Where do they meet? Apoptosis : An International Journal on Programmed Cell Death, 19(4), 555–566. https://doi.org/10.1007/s10495-014-0967-2
Murphy, M. P. (2009). How mitochondria produce reactive oxygen species. The Biochemical Journal, 417(1), 1–13. https://doi.org/10.1042/BJ20081386
Nazim, U. M., & Park, S. Y. (2019). Attenuation of autophagy flux by 6-shogaol sensitizes human liver cancer cells to TRAIL-induced apoptosis via p53 and ROS. International Journal of Molecular Medicine, 43(2), 701–708. https://doi.org/10.3892/ijmm.2018.3994
Nieminen, A. I., Eskelinen, V. M., Haikala, H. M., Tervonen, T. A., Yan, Y., Partanen, J. I., & Klefström, J. (2013). Myc-induced AMPK-phospho p53 pathway activates Bak to sensitize mitochondrial apoptosis. Proceedings of the National Academy of Sciences of the United States of America, 110(20), E1839–E1848. https://doi.org/10.1073/pnas.1208530110
Nogueira, V., Park, Y., Chen, C. C., Xu, P. Z., Chen, M. L., Tonic, I., Unterman, T., & Hay, N. (2008). Akt determines replicative senescence and oxidative or oncogenic premature senescence and sensitizes cells to oxidative apoptosis. Cancer Cell, 14(6), 458–470. https://doi.org/10.1016/j.ccr.2008.11.003
Otomo, C., Metlagel, Z., Takaesu, G., & Otomo, T. (2013). Structure of the human ATG12~ATG5 conjugate required for LC3 lipidation in autophagy. Nature Structural & Molecular Biology, 20(1), 59–66. https://doi.org/10.1038/nsmb.2431
Owen, M. R., Doran, E., & Halestrap, A. P. (2000). Evidence that metformin exerts its anti-diabetic effects through inhibition of complex 1 of the mitochondrial respiratory chain. The Biochemical Journal, 348(Pt 3), 607–614.
Ozaki, T., & Nakagawara, A. (2011). Role of p53 in cell death and human cancers. Cancers, 3(1), 994–1013. https://doi.org/10.3390/cancers3010994
Pandey, S. S., Singh, S., Pathak, C., & Tiwari, B. S. (2018). “Programmed cell death: A process of death for survival” - How far terminology pertinent for cell death in unicellular organisms. Journal of Cell Death, 11, 1179066018790259. https://doi.org/10.1177/1179066018790259
Park, H. K., Yoon, N. G., Lee, J. E., Hu, S., Yoon, S., Kim, S. Y., Hong, J. H., Nam, D., Chae, Y. C., Park, J. B., & Kang, B. H. (2020). Unleashing the full potential of Hsp90 inhibitors as cancer therapeutics through simultaneous inactivation of Hsp90, Grp94, and TRAP1. Experimental & Molecular Medicine, 52(1), 79–91. https://doi.org/10.1038/s12276-019-0360-x
Peng, M., Darko, K. O., Tao, T., Huang, Y., Su, Q., He, C., Yin, T., Liu, Z., & Yang, X. (2017). Combination of metformin with chemotherapeutic drugs via different molecular mechanisms. Cancer Treatment Reviews, 54, 24–33. https://doi.org/10.1016/j.ctrv.2017.01.005
Petrovski, G., Zahuczky, G., Katona, K., Vereb, G., Martinet, W., Nemes, Z., Bursch, W., & Fésüs, L. (2007). Clearance of dying autophagic cells of different origin by professional and non-professional phagocytes. Cell Death and Differentiation, 14(6), 1117–1128. https://doi.org/10.1038/sj.cdd.4402112
Pillai, R. N., & Ramalingam, S. S. (2014). Heat shock protein 90 inhibitors in non-small-cell lung cancer. Current Opinion in Oncology, 26(2), 159–164. https://doi.org/10.1097/CCO.0000000000000047
Pizzo, P., & Pozzan, T. (2007). Mitochondria-endoplasmic reticulum choreography: Structure and signaling dynamics. Trends in Cell Biology, 17(10), 511–517. https://doi.org/10.1016/j.tcb.2007.07.011
Polivka, J., Jr., & Janku, F. (2014). Molecular targets for cancer therapy in the PI3K/AKT/mTOR pathway. Pharmacology & Therapeutics, 142(2), 164–175. https://doi.org/10.1016/j.pharmthera.2013.12.004
Rasmussen, A. K., Chatterjee, A., Rasmussen, L. J., & Singh, K. K. (2003). Mitochondria-mediated nuclear mutator phenotype in Saccharomyces cerevisiae. Nucleic Acids Research, 31(14), 3909–3917. https://doi.org/10.1093/nar/gkg446
Rikiishi, H. (2012). Novel Insights into the Interplay between apoptosis and autophagy. International Journal of Cell Biology, 2012, 317645. https://doi.org/10.1155/2012/317645
Rong, B., & Yang, S. (2018). Molecular mechanism and targeted therapy of Hsp90 involved in lung cancer: New discoveries and developments. International Journal of Oncology, 52(2), 321–336. https://doi.org/10.3892/ijo.2017.4214
Sahai, R., Bhattacharjee, A., Shukla, V. N., Yadav, P., Hasanain, M., Sarkar, J., Narender, T., & Mitra, K. (2020). Gedunin isolated from the mangrove plant Xylocarpus granatum exerts its anti-proliferative activity in ovarian cancer cells through G2/M-phase arrest and oxidative stress-mediated intrinsic apoptosis. Apoptosis : An international journal on programmed cell death, 25(7–8), 481–499. https://doi.org/10.1007/s10495-020-01605-5
Saitoh, M., Nagai, K., Nakagawa, K., Yamamura, T., Yamamoto, S., & Nishizaki, T. (2004). Adenosine induces apoptosis in the human gastric cancer cells via an intrinsic pathway relevant to activation of AMP-activated protein kinase. Biochemical Pharmacology, 67(10), 2005–2011. https://doi.org/10.1016/j.bcp.2004.01.020
Santucci, R., Sinibaldi, F., Cozza, P., Polticelli, F., & Fiorucci, L. (2019). Cytochrome c: An extreme multifunctional protein with a key role in cell fate. International Journal of Biological Macromolecules, 136, 1237–1246. https://doi.org/10.1016/j.ijbiomac.2019.06.180
Sato, S., Fujita, N., & Tsuruo, T. (2000). Modulation of Akt kinase activity by binding to Hsp90. Proceedings of the National Academy of Sciences of the United States of America, 97(20), 10832–10837. https://doi.org/10.1073/pnas.170276797
Scaltriti, M., & Baselga, J. (2006). The epidermal growth factor receptor pathway: A model for targeted therapy. Clinical Cancer Research : An official journal of the American Association for Cancer Research, 12(18), 5268–5272. https://doi.org/10.1158/1078-0432.CCR-05-1554
Schuler, M., & Green, D. R. (2001). Mechanisms of p53-dependent apoptosis. Biochemical Society Transactions, 29(Pt 6), 684–688. https://doi.org/10.1042/0300-5127:0290684
Shakeel, E., Akhtar, S., Khan, M., Lohani, M., Arif, J. M., & Siddiqui, M. H. (2017). Molecular docking analysis of aplysin analogs targeting survivin protein. Bioinformation, 13(9), 293–300. https://doi.org/10.6026/97320630013293
Shimamura, T., Li, D., Ji, H., Haringsma, H. J., Liniker, E., Borgman, C. L., Lowell, A. M., Minami, Y., McNamara, K., Perera, S. A., Zaghlul, S., Thomas, R. K., Greulich, H., Kobayashi, S., Chirieac, L. R., Padera, R. F., Kubo, S., Takahashi, M., Tenen, D. G., … Shapiro, G. I. (2008). Hsp90 inhibition suppresses mutant EGFR-T790M signaling and overcomes kinase inhibitor resistance. Cancer Research, 68(14), 5827–5838. https://doi.org/10.1158/0008-5472.CAN-07-5428
Shintani, T., & Klionsky, D. J. (2004). Autophagy in health and disease: A double-edged sword. Science (New York, N.Y.), 306(5698), 990–995. https://doi.org/10.1126/science.1099993
Song, X., Kim, S. Y., Zhang, L., Tang, D., Bartlett, D. L., Kwon, Y. T., & Lee, Y. J. (2014). Role of AMP-activated protein kinase in cross-talk between apoptosis and autophagy in human colon cancer. Cell death & disease, 5(10), e1504. https://doi.org/10.1038/cddis.2014.463
Sreedhar, A. S., & Csermely, P. (2004). Heat shock proteins in the regulation of apoptosis: New strategies in tumor therapy: A comprehensive review. Pharmacology & Therapeutics, 101(3), 227–257. https://doi.org/10.1016/j.pharmthera.2003.11.004
Stahl, J. M., Sharma, A., Cheung, M., Zimmerman, M., Cheng, J. Q., Bosenberg, M. W., Kester, M., Sandirasegarane, L., & Robertson, G. P. (2004). Deregulated Akt3 activity promotes development of malignant melanoma. Cancer Research, 64(19), 7002–7010. https://doi.org/10.1158/0008-5472.CAN-04-1399
Subramani, R., Gonzalez, E., Nandy, S. B., Arumugam, A., Camacho, F., Medel, J., Alabi, D., & Lakshmanaswamy, R. (2017). Gedunin inhibits pancreatic cancer by altering sonic hedgehog signaling pathway. Oncotarget, 8(7), 1089–10904. https://doi.org/10.18632/oncotarget.8055
Sun, W., Sanderson, P. E., & Zheng, W. (2016). Drug combination therapy increases successful drug repositioning. Drug Discovery Today, 21(7), 1189–1195. https://doi.org/10.1016/j.drudis.2016.05.015
Tasdemir, E., Maiuri, M. C., Galluzzi, L., Vitale, I., Djavaheri-Mergny, M., D’Amelio, M., Criollo, A., Morselli, E., Zhu, C., Harper, F., Nannmark, U., Samara, C., Pinton, P., Vicencio, J. M., Carnuccio, R., Moll, U. M., Madeo, F., Paterlini-Brechot, P., Rizzuto, R., … Kroemer, G. (2008). Regulation of autophagy by cytoplasmic p53. Nature Cell Biology, 10(6), 676–687. https://doi.org/10.1038/ncb1730
Tharmarajah, L., Samarakoon, S. R., Ediriweera, M. K., Piyathilaka, P., Tennekoon, K. H., Senathilake, K. S., Rajagopalan, U., Galhena, P. B., & Thabrew, I. (2017). In vitro anticancer effect of gedunin on human teratocarcinomal (NTERA-2) cancer stem-like cells. BioMed Research International, 2017, 2413197. https://doi.org/10.1155/2017/2413197
Toker, A. (2012). Achieving specificity in Akt signaling in cancer. Advances in Biological Regulation, 52(1), 78–87. https://doi.org/10.1016/j.advenzreg.2011.09.020
Trachootham, D., Zhou, Y., Zhang, H., Demizu, Y., Chen, Z., Pelicano, H., Chiao, P. J., Achanta, G., Arlinghaus, R. B., Liu, J., & Huang, P. (2006). Selective killing of oncogenically transformed cells through a ROS-mediated mechanism by beta-phenylethyl isothiocyanate. Cancer Cell, 10(3), 241–252. https://doi.org/10.1016/j.ccr.2006.08.009
Vandamme, M., Robert, E., Lerondel, S., Sarron, V., Ries, D., Dozias, S., Sobilo, J., Gosset, D., Kieda, C., Legrain, B., Pouvesle, J. M., & Pape, A. L. (2012). ROS implication in a new antitumor strategy based on non-thermal plasma. International Journal of Cancer, 130(9), 2185–2194. https://doi.org/10.1002/ijc.26252
Vilchez, D., Saez, I., & Dillin, A. (2014). The role of protein clearance mechanisms in organismal ageing and age-related diseases. Nature Communications, 5, 5659. https://doi.org/10.1038/ncomms6659
Wee, P., & Wang, Z. (2017). Epidermal growth factor receptor cell proliferation signaling pathways. Cancers, 9(5), 52. https://doi.org/10.3390/cancers9050052
Wen, S., Zhu, D., & Huang, P. (2013). Targeting cancer cell mitochondria as a therapeutic approach. Future Medicinal Chemistry, 5(1), 53–67. https://doi.org/10.4155/fmc.12.190
Whitesell, L., & Lindquist, S. L. (2005). HSP90 and the chaperoning of cancer. Nature Reviews Cancer, 5(10), 761–772. https://doi.org/10.1038/nrc1716
Willimott, S., Merriam, T., & Wagner, S. D. (2011). Apoptosis induces Bcl-XS and cleaved Bcl-XL in chronic lymphocytic leukaemia. Biochemical and Biophysical Research Communications, 405(3), 480–485. https://doi.org/10.1016/j.bbrc.2011.01.057
Wu, G. S., & Ding, Z. (2002). Caspase 9 is required for p53-dependent apoptosis and chemosensitivity in a human ovarian cancer cell line. Oncogene, 21(1), 1–8. https://doi.org/10.1038/sj.onc.1205020
Wu, H., Chen, S., Ammar, A. B., Xu, J., Wu, Q., Pan, K., Zhang, J., & Hong, Y. (2015). Crosstalk between macroautophagy and chaperone-mediated autophagy: Implications for the treatment of neurological diseases. Molecular Neurobiology, 52(3), 1284–1296. https://doi.org/10.1007/s12035-014-8933-0
Xiang, X., Saha, A. K., Wen, R., Ruderman, N. B., & Luo, Z. (2004). AMP-activated protein kinase activators can inhibit the growth of prostate cancer cells by multiple mechanisms. Biochemical and Biophysical Research Communications, 321(1), 161–167. https://doi.org/10.1016/j.bbrc.2004.06.133
Xu, C., Liu, J., Hsu, L. C., Luo, Y., Xiang, R., & Chuang, T. H. (2011). Functional interaction of heat shock protein 90 and Beclin 1 modulates Toll-like receptor-mediated autophagy. FASEB journal: Official publication of the Federation of American Societies for Experimental Biology, 25(8), 2700–2710. https://doi.org/10.1096/fj.10-167676
Yang, H., Lee, M. H., Park, I., Jeon, H., Choi, J., Seo, S., Kim, S. W., Koh, G. Y., Park, K. S., & Lee, D. H. (2017). HSP90 inhibitor (NVP-AUY922) enhances the anti-cancer effect of BCL-2 inhibitor (ABT-737) in small cell lung cancer expressing BCL-2. Cancer Letters, 411, 19–26. https://doi.org/10.1016/j.canlet.2017.09.040
Yousefi, S., Perozzo, R., Schmid, I., Ziemiecki, A., Schaffner, T., Scapozza, L., Brunner, T., & Simon, H. U. (2006). Calpain-mediated cleavage of Atg5 switches autophagy to apoptosis. Nature Cell Biology, 8(10), 1124–1132. https://doi.org/10.1038/ncb1482
Yu, C., Jiao, Y., Xue, J., Zhang, Q., Yang, H., Xing, L., Chen, G., Wu, J., Zhang, S., Zhu, W., & Cao, J. (2017). Metformin sensitizes non-small cell lung cancer cells to an epigallocatechin-3-gallate (EGCG) treatment by suppressing the Nrf2/HO-1 signaling pathway. International Journal of Biological Sciences, 13(12), 1560–1569. https://doi.org/10.7150/ijbs.18830
Zhang, L., Yi, Y., Guo, Q., Sun, Y., Ma, S., Xiao, S., Geng, J., Zheng, Z., & Song, S. (2012). Hsp90 interacts with AMPK and mediates acetyl-CoA carboxylase phosphorylation. Cellular Signalling, 24(4), 859–865. https://doi.org/10.1016/j.cellsig.2011.12.001
Zhou, G., Myers, R., Li, Y., Chen, Y., Shen, X., Fenyk-Melody, J., Wu, M., Ventre, J., Doebber, T., Fujii, N., Musi, N., Hirshman, M. F., Goodyear, L. J., & Moller, D. E. (2001). Role of AMP-activated protein kinase in mechanism of metformin action. The Journal of Clinical Investigation, 108(8), 1167–1174. https://doi.org/10.1172/JCI13505
Zhou, H., Li, X. M., Meinkoth, J., & Pittman, R. N. (2000). Akt regulates cell survival and apoptosis at a postmitochondrial level. The Journal of Cell Biology, 151(3), 483–494. https://doi.org/10.1083/jcb.151.3.483
Zhou, J., Yang, J., Fan, X., Hu, S., Zhou, F., Dong, J., Zhang, S., Shang, Y., Jiang, X., Guo, H., Chen, N., Xiao, X., Sheng, J., Wu, K., Nie, Y., & Fan, D. (2016). Chaperone-mediated autophagy regulates proliferation by targeting RND3 in gastric cancer. Autophagy, 12(3), 515–528. https://doi.org/10.1080/15548627.2015.1136770
Zong, W. X., & Moll, U. (2008). p53 in autophagy control. Cell Cycle (Georgetown, Tex.), 7(19), 2947. https://doi.org/10.4161/cc.7.19.7008
Zorov, D. B., Filburn, C. R., Klotz, L. O., Zweier, J. L., & Sollott, S. J. (2000). Reactive oxygen species (ROS)-induced ROS release: A new phenomenon accompanying induction of the mitochondrial permeability transition in cardiac myocytes. The Journal of Experimental Medicine, 192(7), 1001–1014. https://doi.org/10.1084/jem.192.7.1001
Zou, Z., Tao, T., Li, H., & Zhu, X. (2020). mTOR signaling pathway and mTOR inhibitors in cancer: Progress and challenges. Cell & Bioscience, 10, 31. https://doi.org/10.1186/s13578-020-00396-1
Acknowledgements
The authors are thankful to Hon’ble Vice-Chancellor, Integral University, Lucknow, for providing infrastructural support and Dean Office, R&D, Integral University, for providing manuscript communication number (IU/R&D/2021-MCN0001317). The author, A.H., is thankful to SERB-DST for providing the fellowship.
Funding
This work was supported by the Science & Engineering Research Board (SERB-DST) (grant number: YSS/2015/001902, awarded to SSM).
Author information
Authors and Affiliations
Contributions
Conceptualization: AH, SSM; experiment performance: AH; material preparation, data collection, and analysis: AH and SSM; writing—original draft preparation: AH and SSM; writing—review and editing: AH, SSM, NK, ML; funding acquisition: SSM; resources: SSM; supervision: SSM.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
Yes. All authors agreed to participate in this research.
Consent for Publication
Yes. All authors have approved the last version of the manuscript for its submission.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hasan, A., Khamjan, N., Lohani, M. et al. Targeted Inhibition of Hsp90 in Combination with Metformin Modulates Programmed Cell Death Pathways in A549 Lung Cancer Cells. Appl Biochem Biotechnol 195, 7338–7378 (2023). https://doi.org/10.1007/s12010-023-04424-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-023-04424-x