Abstract
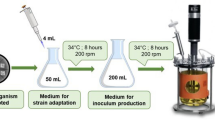
In this study, a newly isolated strain Filobasidium magnum JD1025 was investigated for its production of sclareolide, which was verified to be a valuable raw material in various industrial fields. Together with a comprehensive analysis of the genome sequence, effective fermentation method to convert sclareol to sclareolide via the isolated strain was explored and optimized by taking the selected co-solvent and nitrogen source into account. The results showed that the final conversion rate could be achieved at 88.79 ± 1.06% with the initial sclareol concentration of 30 g·L-1 after 72 h in baffled flask. The corresponding yield concentration of sclareolide was 21.62 ± 0.26 g·L-1 and the conversion rate per unit thallus attained to 6.11 ± 0.06 % g-1·L-1. Overall, the current study suggested a valid method for the application of Filobasidium magnum JD1025 as bio-transformer to produce sclareolide from sclareol.








Similar content being viewed by others
References
Upar, K. B., Mishra, S. J., Nalawade, S. P., Singh, S. A., & Bhat, S. V. (2009). Efficient enantioselective synthesis of (+)-sclareolide and (+)-tetrahydroactinidiolide: chiral LBA-induced biomimetic cyclization. Tetrahedron Asymmetry, 20(14), 1637–1640.
Martins, M. P., Ouazzani, J., Arcile, G., Jeller, A. H., Lima, J. P. F. D., Seleghim, M. H. R., Oliveira, A. L. L., Debonsi, H. M., Venancio, T., & Yokoya, N. S. (2015). Biohydroxylation of (-)-ambrox®, (-)-sclareol, and (+)-sclareolide by whole cells of brazilian marine-derived fungi. Marine Biotechnology, 17(2), 211–218.
Castro, J. M., Salido, S., Sánchez, A., & Altarejos, J. (2010). Synthesis of (+)-sclareolide based on a cyclic enol ether ring contraction induced by peroxy acids. Synlett, 2010(18), 2747–2750.
Frija, Luís. M. . T., Frade, R., & Afonso, C. (2011). Isolation, chemical, and biotransformation routes of labdane-type diterpenes. Chemical Reviews, 111(8), 4418–4452.
Duca, G., Aricu, A., Kuchkova, K., Secara, E., Barba, A., Dragalin, I., et al. (2018). Synthesis, structural elucidation and biological evaluations of new guanidine-containing terpenoids as anticancer agents. Natural Product Research, 33(21), 3052–3056.
Chen, S., Wang, Y., Zhang, W., Dong, M., & Zhang, J. (2017). Sclareolide enhances gemcitabineinduced cell death through mediating the nicd and gli1 pathways in gemcitabineresistant human pancreatic cancer. Molecular Medicine Reports, 15(4), 1461–1470.
Sultana, Ni., & Saify, Z. S. (2013). Enzymatic biotransformation of terpenes as bioactive agents. Journal of Enzyme Inhibition & Medicinal Chemistry, 28(6), 1113–1128.
Chen, Q., Tang, K., & Guo, Y. (2019). Discovery of sclareol and sclareolide as filovirus entry inhibitors. Journal of Asian Natural Products Research, 22(5), 1–10.
Krutmann, J., Meyer, I., Johncock, W., Schmaus, G., & Le Maire, M. (2017) Cosmetic compositions comprising sclareolide. World Intellectual Property Organization, WO 2017/215729 A1.
Völler, J. S. (2020). Expedient route to meroterpenoids. Nature Catalysis, 3(2), 92.
Farooq, A., & Tahara, S. (2000). Oxidative metabolism of ambrox and sclareolide by botrytis cinerea. Zeitschrift Fur Naturforschung C A Journal of Biosciences, 55(5–6), 341–346.
Serra, S., Fuganti, C., & Brenna, E. (2005). Biocatalytic preparation of natural flavours and fragrances. Trends in Biotechnology, 23(4), 193–198.
Choudhary, M. I., Musharraf, S. G., Sami, A., & Atta-ur-Rahman. (2004). Microbial transformation of sesquiterpenes, (-)-Ambrox and (+) -sclareolide. Helvetica Chimica Acta, 87(10), 2685–2694.
Barrero, A. F., Alvarez-Manzaneda, E. J., Chahboun, R., & Arteaga, A. F. (2004). Degradation of the side chain of (−) -sclareol: a very short synthesis of nor-ambreinolide and ambrox. Synthetic Communications, 34(19), 3631–3643.
Koga, T., Aoki, Y., Hirose, T., & Nohira, H. (1998). Resolution of sclareolide as a key intermediate for the synthesis of ambrox®. Tetrahedron Asymmetry, 9(21), 3819–3823.
Xiang, L., Chen, K., Wu, B., & Min, Z. (2016). Recycling of sclareolide in the crystallization mother liquid of sclareolide by adsorption and chromatography. International Journal of Chemical and Molecular Engineering, 10(2), 187–192.
Barton, D., Parekh, S. I., Taylor, D. K., & Tse, C. L. (1994). An efficient synthesis of (-)-dodecahydro-3a,6,6,9a-tetramethylnaphtho[2,1-b] furan from (-)-sclareol. Cheminform, 35(32), 5801–5804.
Schneider, M., Stalberg, T., & Gerke, T. (1996). Process for the production of sclareolide, United States Patent, US 5,525,728A.
Gerke, T., & Bruns, K. (1993). Process for the production of sclareolide. United States Patent, US 5,247,100 A.
Maurey, H. W., Schumacher, J. N., & Teague, J. (1962). Two stage oxidation of sclareol. United States Patent, US3050532 A.
Farbood, M. I., Morris, J. A., & Downey, A. E. (1990). Process for producing diol and lactone and microorganisms capable of same. United States Patent, US04970163 A.
Farbood, M. I., Morris, J. A., & Downey, A. E. (1991). Process and means for producing diol and lactone. European Patent, EP0419026A1.
Arturo, C., Teresa, R., & Guillermo, D. (2011). Biotransformation of sclareolide by filamentous fungi: cytotoxic evaluations of the derivatives. Journal of the Brazilian Chemical Society, 22(6), 1177–1182.
Chitsazan, M. H., Bina, E., & Asgarpanah, J. (2014). Essential oil composition of the endemic species pycnocycla bashagardiana mozaff. Journal of Essential Oil Research, 26(2), 141–145.
Zafar, I., Fatima, A., Khan, S. J., Rehman, Z., & Mehmud, S. (2010). GC-MS studies of needles essential oil of pinus roxburghaii and their antimicrobial activity from. Electronic Journal of Environmental Agricultural & Food Chemistry, 9(3), 468–473.
Zhou, P., Fang, Y. K., Yao, H. K., Li, H., Wang, G., & Liu, Y. P. (2018). Efficient biotransformation of phytosterols to dehydroepiandrosterone by Mycobacterium sp. Applied Biochemistry and Biotechnology, 186(4), 1–11.
Ma, M., Feng, J., Wang, D., Chen, S. W., & Xu, H. (2018). Synthesis and antifungal activity evaluation of drimane-amide derivatives from sclareol. Combinatorial Chemistry & High Throughput Screening, 21(7), 501–509.
Birney, E., Clamp, M., & Durbin, R. (2004). GeneWise and Genomewise. Genome Res, 14(5), 988–95.
Kuznetsov, A., & Bollin, C. J. (2021). NCBI Genome Workbench: desktop software for comparative genomics, visualization, and GenBank data submission. In Multiple Sequence Alignment. Humana, New York, NY 261-295
Kyrikou, I., Georgopoulos, A., Hatziantoniou, S., Mavromoustakos, T., & Demetzos, C. (2005). A comparative study of the effects of cholesterol and sclareol, a bioactive labdane type diterpene, on phospholipid bilayers. Chemistry and Physics of Lipids, 133(2), 125–134.
Ruan, Y., Zhu, L., & Qi, L. (2015). Improving the electro-transformation efficiency of corynebacterium glutamicum by weakening its cell wall and increasing the cytoplasmic membrane fluidity. Biotechnology Letters, 37(12), 2445–2452.
Feng, L., & Zhu, L. (2014). Surfactant-modified fatty acid composition of Citrobacter sp. SA01 and its effect on phenanthrene transmembrane transport. Chemosphere, 107, 58–64.
Funding
This work was supported by the National Key Research & Developmental Program of China (2018YFA0900300 and 2021YFC2100300).
Author information
Authors and Affiliations
Contributions
Y. K. Fang: investigation, formal analysis, and writing—original draft. Z. L. Wang: methodology, validation, and investigation. Y. Shi: writing—review and editing. L. Liu: methodology, validation, and investigation. J. H. Wang: data curation and visualization. T. Yang: visualization and software. X. Yu: resources and conceptualization. Z. H. Gu: methodology and software. L. Zhang: funding acquisition, project administration, and methodology. We confirm that this manuscript has been read and approved by all named authors and that there are no other persons who satisfied the criteria for authorship but are not listed. We further confirm that the order of authors listed in this manuscript has been approved by all of us.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fang, Y., Wang, Z., Shi, Y. et al. Efficient Biotransformation of Sclareol to Sclareolide by Filobasidium magnum JD1025. Appl Biochem Biotechnol 195, 1184–1196 (2023). https://doi.org/10.1007/s12010-022-04225-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-022-04225-8