Abstract
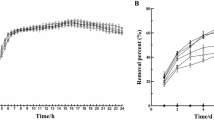
Microbial remediation has become one of the promising ways to eliminate polycyclic aromatic hydrocarbons (PAHs) pollution due to its efficient enzyme metabolism system. Catechol 1,2-dioxygenase (C12O) is a crucial rate-limiting enzyme in the degradation pathway of PAHs in Achromobacter xylosoxidans DN002 that opens the benzene ring through the ortho-cleavage pathway. However, little attention has been given to explore the interaction mechanism of relevant enzyme–substrate. This study aims to investigate the binding interaction between C12O of strain DN002 and catechol by means of a molecular biological approach combined with homology modeling, molecular docking, and multiple spectroscopies. The removal rate of catechol in the mutant strain of cat A deletion was only 12.03%, compared to the wild-type strain (54.21%). A Ramachandran plot of active site regions of the primary amino acid sequences in the native enzyme showed that 93.5% sequences were in the most favored regions on account of the results of homology modeling, while an additional 6.2% amino acid sequences were found in conditionally allowed regions, and 0.4% in generously allowed regions. The binding pocket of C12O with catechol was analyzed to obtain that the catalytic trimeric group of Tyr164-His224-His226 was proven to be great vital for the ring-opening reaction of catechol by molecular docking. In the native enzyme, binding complexes were spontaneously formed by hydrophobic interactions. Binding constants and thermodynamic potentials from fluorescence spectra indicated that catechol effectively quenched the intrinsic fluorescence of C12O in the C12O/catechol complex via conventional static and dynamic quenching mechanisms of C12O. The results of ultraviolet and visible (UV) spectra, synchronous fluorescence, and circular dichroism (CD) spectra revealed conspicuous changes in the local conformation, and site-directed mutagenesis confirmed the role of predicted key residues during catalysis, wherein His226 had a significant effect on catechol utilization by C12O. This is the first report to reveal interactions of C12O with substrate from the molecular docking results, providing the mechanistic understanding of representative dioxygenases involved in aromatic compound degradation, and a solid foundation for further site modifications as well as strategies for the directed evolution of this enzyme.






Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article (and its supplementary information files).
References
Kosnar, Z., et al. (2019). Comparing the removal of polycyclic aromatic hydrocarbons in soil after different bioremediation approaches in relation to the extracellular enzyme activities. Journal of Environmental Sciences (China), 76, 249–258.
Haritash, A. K., & Kaushik, C. P. (2009). Biodegradation aspects of polycyclic aromatic hydrocarbons (PAHs): A review. Journal of Hazardous Materials, 169(1–3), 1–15.
Imam, A., et al. (2021). Biological machinery for polycyclic aromatic hydrocarbons degradation: A review. Bioresource Technology, 343, 126121.
Trivedi, P., et al. (2016). Microbial regulation of the soil carbon cycle: Evidence from gene-enzyme relationships. ISME Journal, 10(11), 2593–2604.
Kusmierz, M., et al. (2016). Persistence of polycyclic aromatic hydrocarbons (PAHs) in biochar-amended soil. Chemosphere, 146, 272–279.
Da, S. J. F., et al. (2021). A look beyond the priority: A systematic review of the genotoxic, mutagenic, and carcinogenic endpoints of non-priority PAHs. Environmental Pollution, 278, 116838.
Ghosh, P., & Mukherji, S. (2021). Environmental contamination by heterocyclic polynuclear aromatic hydrocarbons and their microbial degradation. Bioresource Technology, 341, 125860.
Lu, C., et al. (2019). A PAH-degrading bacterial community enriched with contaminated agricultural soil and its utility for microbial bioremediation. Environmental Pollution, 251, 773–782.
Obi, C. C., et al. (2017). Structural dynamics of microbial communities in polycyclic aromatic hydrocarbon-contaminated tropical estuarine sediments undergoing simulated aerobic biotreatment. Applied Microbiology and Biotechnology, 101(10), 4299–4314.
Baboshin, M. A., & Golovleva, L. A. (2012). Biodegradation of polycyclic aromatic hydrocarbons (PAHs) by aerobic bacteria and its kinetics aspects. Mikrobiologiia, 81(6), 695–706.
Sainsbury, P. D., et al. (2015). Chemical intervention in bacterial lignin degradation pathways: Development of selective inhibitors for intradiol and extradiol catechol dioxygenases. Bioorganic Chemistry, 60, 102–109.
Reddig, N., et al. (2004). Mononuclear manganese (III) catechol compounds as substrate adduct complexes for manganese-substituted intradiol cleaving catechol dioxygenases. Inorganica Chimica Acta, 357(9), 2703–2712.
Matera, I., et al. (2010). Catechol 1,2-dioxygenase from the Gram-positive Rhodococcus opacus 1CP: Quantitative structure/activity relationship and the crystal structures of native enzyme and catechols adducts. Journal of Structural Biology, 170(3), 548–564.
Li, J., et al. (2021). Expression and characterization of catechol 1,2-dioxygenase from Oceanimonas marisflavi 102-Na3. Protein Expression and Purification, 188, 105964.
Guzik, U., et al. (2011). Catechol 1,2-dioxygenase from the new aromatic compounds – Degrading Pseudomonas putida strain N6. International Biodeterioration & Biodegradation, 65(3), 504–512.
Nakai, C., et al. (1995). Cloning, DNA sequencing, and amino acid sequencing of catechol 1,2-dioxygenases (pyrocatechase) from Pseudomonas putida mt-2 and Pseudomonas arvilla C-1. Archives of Biochemistry and Biophysics, 321(2), 353–362.
Micalella, C., et al. (2011). X-ray crystallography, mass spectrometry and single crystal micro spectrophotometry: A multidisciplinary characterization of catechol 1,2 dioxygenase. Biochimica et Biophysica Acta, 1814(6), 817–823.
Li, Y., et al. (2020). Complete genome sequence of the aromatic-hydrocarbon-degrading bacterium Achromobacter xylosoxidans DN002. Archives of Microbiology, 202(10), 2849–2853.
Ma, Y. L., et al. (2015). Elucidation of fluoranthene degradative characteristics in a newly isolated Achromobacter xylosoxidans DN002. Applied Biochemistry and Biotechnology, 175(3), 1294–1305.
Romero-Arroyo, C. E., et al. (1995). catM encodes a LysR-type transcriptional activator regulating catechol degradation in Acinetobacter calcoaceticus. Journal of Bacteriology, 177(20), 5891–5898.
Kuntal, B. K., Aparoy, P., & Reddanna, P. (2010). EasyModeller: A graphical interface to MODELLER. BMC Research Notes, 3, 226.
Trott, O., & Olson, A. J. (2010). AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. Journal of Computational Chemistry, 31(2), 455–461.
Lu, H., et al. (2022). Machine learning-aided engineering of hydrolases for PET depolymerization. Nature, 604(7907), 662–667.
Bhatt, P., et al. (2021). Binding interaction of glyphosate with glyphosate oxidoreductase and C-P lyase: Molecular docking and molecular dynamics simulation studies. Journal of Hazardous Materials, 409, 124927.
Lohning, A. E., et al. (2017). A practical guide to molecular docking and homology modelling for medicinal chemists. Current Topics in Medicinal Chemistry, 17(18), 2023–2040.
Rigsby, R. E., & Parker, A. B. (2016). Using the PyMOL application to reinforce visual understanding of protein structure. Biochemistry and Molecular Biology Education, 44(5), 433–437.
Laskowski, R. A., & Swindells, M. B. (2011). LigPlot+: Multiple ligand-protein interaction diagrams for drug discovery. Journal of Chemical Information and Modeling, 51(10), 2778–2786.
Zhang, J., et al. (2020). Study on the molecular interactions of hydroxylated polycyclic aromatic hydrocarbons with catalase using multi-spectral methods combined with molecular docking. Food Chemistry, 309, 125743.
Zhang, J., Zhu, Y., & Zhang, Y. (2021). Potential ability of different types of cyclodextrins to modulate the interaction between bovine serum albumin and 1-hydroxypyrene. Food Chemistry, 343, 128516.
Alvarez, R., et al. (2003). (9Z)- and (11Z)-8-methylretinals for artificial visual pigment studies: Stereoselective synthesis, structure, and binding models. Chemistry, 9(23), 5821–5831.
Micalella, C., et al. (2011). X-ray crystallography, mass spectrometry and single crystal microspectrophotometry: A multidisciplinary characterization of catechol 1,2 dioxygenase. Biochimica et Biophysica Acta, 1814(6), 817–823.
Varadi, T., et al. (2013). Iron (III) complexes with meridional ligands as functional models of intradiol-cleaving catechol dioxygenases. Inorganic Chemistry, 52(3), 1559–1569.
Pushpam, S., Kottaisamy, M., & Ramakrishnan, V. (2013). Dynamic quenching study of 2-amino-3-bromo-1,4-naphthoquinone by titanium dioxide nano particles in solution (methanol). Spectrochim Acta A Mol Biomol Spectrosc, 114, 272–276.
AlAjmi, M. F., et al. (2020). Understanding the interaction between alpha-1-acid glycoprotein (AGP) and potential Cu/Zn metallo-drugs of benzimidazole derived organic motifs: A multi-spectroscopic and molecular docking study. Spectrochim Acta A Mol Biomol Spectrosc, 225, 117457.
Blundell, T. L. (2021). The first resolution revolution in protein structure analysis: X-ray diffraction of polypeptide conformations and globular protein folds in 1950s and 1960s. Progress in Biophysics and Molecular Biology, 167, 32–40.
Li, J., et al. (2021). Mechanistic insights into the success of xenobiotic degraders resolved from metagenomes of microbial enrichment cultures. Journal of Hazardous Materials, 418, 126384.
Li, J., et al. (2021) Genomics-informed insights into microbial degradation of N, N-dimethylformamide. International Biodeterioration & Biodegradation 163.
Tournier, V., et al. (2020). An engineered PET depolymerase to break down and recycle plastic bottles. Nature, 580(7802), 216–219.
Igor, V. K., Steven, H. S., Olga, V. B. (2012). Solution-phase perfluoro alkylation of C 60 leads to efficient and selective synthesis of bis-perfluoro alkylated fullerenes. Journal of Fluorine Chemistry, 143.
Han, L., et al. (2015). Engineering catechol 1, 2-dioxygenase by design for improving the performance of the cis, cis-muconic acid synthetic pathway in Escherichia coli.
Bugg, T. D., & Ramaswamy, S. (2008). Non-heme iron-dependent dioxygenases: Unravelling catalytic mechanisms for complex enzymatic oxidations. Current Opinion in Chemical Biology, 12(2), 134–140.
Zeng, X. H., et al. (2020). Insights into the binding interaction of substrate with catechol 2,3-dioxygenase from biophysics point of view. Journal of Hazardous Materials, 391, 122211.
Du, H., et al. (2021). Mechanistic insight into esterase-catalyzed hydrolysis of phthalate esters (PAEs) based on integrated multi-spectroscopic analyses and docking simulation. Journal of Hazardous Materials, 408, 124901.
Bilen, E., et al. (2022). Bioactive sulfonyl hydrazones with alkyl derivative: Characterization, ADME properties, molecular docking studies and investigation of inhibition on choline esterase enzymes for the diagnosis of Alzheimer's disease. Chem Biol Interact p. 109956.
Mansouri, N., Benslama, O. (2022). Molecular docking exploration of the degradation activity of some synthetic hydro carbons polymers by the laccase enzyme of Streptomyces. Materials Today: Proceedings. 54(P3).
Sahin, S., et al. (2021). Investigation of binding interaction behavior between antiemetic drugs and Trypsin by spectroscopy and molecular docking. Spectrochim Acta A Mol Biomol Spectrosc, 258, 119817.
Mayilmurugan, R., et al. (2010). Novel square pyramidal iron (III) complexes of linear tetradentate bis(phenolate) ligands as structural and reactive models for intradiol-cleaving 3,4-PCD enzymes: Quinone formation vs. intradiol cleavage. Dalton Trans 39(40): p. 9611–25.
Bruijnincx, P. C., et al. (2007). Iron (III)-catecholato complexes as structural and functional models of the intradiol-cleaving catechol dioxygenases. Inorganic Chemistry, 46(20), 8391–8402.
Funding
The authors would like to acknowledge the support received from the National Natural Science Foundation of China (31000069), and the Key Project on Social Development of Science and Technology in Shaanxi Province (grant no. 2017ZDXM-SF-105).
Author information
Authors and Affiliations
Contributions
YL, FW, and YM conceived and designed the study. YL, RX, and TC performed the experiments and bioinformatics analysis. YL, FW, and RX analyzed the data and prepared all of figures. YL, RX, and YM wrote the manuscript. YL and YM revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent to Publish
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, Y., Wei, F., Xu, R. et al. Insights into the Binding Interaction of Catechol 1,2-Dioxygenase with Catechol in Achromobacter xylosoxidans DN002. Appl Biochem Biotechnol 195, 298–313 (2023). https://doi.org/10.1007/s12010-022-04129-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-022-04129-7