Abstract
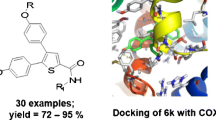
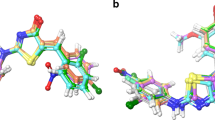
Although nonsteroidal anti-inflammatory drugs (NSAIDs) are medicines that are widely used to relieve pain, reduce inflammation, and bring down high temperature, literature confirmed that they still have harmful side effects. Most of their side effects are in the digestive system due to the carboxylic group. As naproxen is one of the NSAIDs, in this work, we try to mask the carboxylic group in naproxen with a relatively safe functional group. So, herein, we report the synthesis of new naproxen-based hydrazones derivatives, namely, (E)-N'-1-(4-chlorophenyl)ethylidene)-2-(6-methoxynaphthalen-2-yl)propane hydrazide (4a) and (E)-N'-(4-hydroxybenzylidene)-2-(6-methoxynaphthalen-2-yl)propane hydrazide ethanol solvate (4b). The compounds were confirmed by X-ray diffraction studies. Hirshfeld surface analyses and energy frameworks of 4a and 4b have been carried out and blind molecular docking studies of them to the COX-2 enzyme were undertaken to obtain binding affinities for judging whether the compounds could act as anti-inflammatory agents. The compounds interact with the key residues: Arg120, Val349, Leu352, Tyr355, Val523, Ala527, Ser530, and Leu531 of the active enzyme pocket. Molecular dynamics studies predicted that the complexes of 4a and 4b with COX-2 are structurally stable and no major conformational changes were observed. Confirmation of the docking and simulation data was achieved by a binding free energies analysis that indicated the dominance of van der Waals energy. The compounds are drug-like molecules as they obey all prominent drug-like rules and have acceptable pharmacokinetic profiles. To investigate the relationship between their intrinsic electronic properties and their possible similarities to actual drugs, the gas-phase DFT optimizations and NBO analyses were also performed in this study.














Similar content being viewed by others
Data Availability
All data and materials that were used in the article are available.
References
Camacho-Muñoz, D., Martín, J., Santos, J. L., Aparicio, I., & Alonso, E. (2012). Effectiveness of conventional and low-cost wastewater treatments in the removal of pharmaceutically active compounds. Water, Air, & Soil Pollution, 223(5), 2611–2621.
Zorita, S., Mårtensson, L., & Mathiasson, L. (2009). Occurrence and removal of pharmaceuticals in a municipal sewage treatment system in the south of Sweden. Science of the total environment, 407(8), 2760–2770.
Abbas, A. H., Elias, A. N., & Fadhil, A. A. (2015). Synthesis, characterization and biological evaluation of new potentially active hydrazones of naproxen hydrazide. Der Pharma Chemica, 7(10), 93–101.
Sharma, S., Srivastava, V. K., & Kumar, A. (2002). Newer N-substituted anthranilic acid derivatives as potent anti-inflammatory agents. European journal of medicinal chemistry, 37(8), 689–697.
Guinan, M., Benckendorff, C., Smith, M., & Miller, G. J. (2020). Recent advances in the chemical synthesis and evaluation of anticancer nucleoside analogues. Molecules, 25(9), 2050.
Vane, J. R. (1971). Inhibition of prostaglandin synthesis as a mechanism of action for aspirin-like drugs. Nature new biology, 231(25), 232–235.
Han, M. İ., Bekçi, H., Uba, A. I., Yıldırım, Y., Karasulu, E., Cumaoğlu, A., … Küçükgüzel, Ş. G. (2019). Synthesis, molecular modeling, in vivo study, and anticancer activity of 1, 2, 4‐triazole containing hydrazide–hydrazones derived from (S)‐naproxen. Archiv der Pharmazie, 352(6), 1800365.
Han, M. İ, & Küçükgüzel, ŞG. (2020). Anticancer and antimicrobial activities of naproxen and naproxen derivatives. Mini reviews in medicinal chemistry, 20(13), 1300–1310.
Han, M. İ, Atalay, P., Tunç, C. Ü., Ünal, G., Dayan, S., Aydın, Ö., & Küçükgüzel, ŞG. (2021). Design and synthesis of novel (S)-Naproxen hydrazide-hydrazones as potent VEGFR-2 inhibitors and their evaluation in vitro/in vivo breast cancer models. Bioorganic & Medicinal Chemistry, 37, 116097.
Han, M. I., Bekci, H., Cumaoğlu, A., & Küçükgüzel, ŞG. (2018). Synthesis and characterization of 1, 2, 4- triazole containing hydrazide-hydrazones derived from (S)-Naproxen as anticancer agents. Marmara Pharmaceutical Journal, 22(4), 559–569.
Türköz Acar, E., Helvacioğlu, S., Charehsaz, M., & Aydin, A. (2018). Determination and safety evaluation of furfural and hydroxymethylfurfural in some honey samples by using a validated HPLC-DAD method. Marmara Pharmaceutical Journal, 22(4), 519–527.
Morris, G. M., & Lim-Wilby, M. (2008). Molecular docking. Molecular Modeling of Proteins, 443, 365–382.
Sun, H., Li, Y., Tian, S., Xu, L., & Hou, T. (2014). Assessing the performance of MM/PBSA and MM/GBSA methods. 4. Accuracies of MM/PBSA and MM/GBSA methodologies evaluated by various simulation protocols using PDBbind data set. Physical Chemistry Chemical Physics, 16(31), 16719–16729.
Daina, A., Michielin, O., & Zoete, V. (2017). SwissADME: A free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Scientific Reports, 7(January), 1–13. https://doi.org/10.1038/srep42717
Nunes, A. M. V., de Andrade, F. das C. P., Filgueiras, L. A., de Carvalho Maia, O. A., Cunha, R. L. O. R., Rodezno, S. V. A., … Mendes, A. N. (2020). preADMET analysis and clinical aspects of dogs treated with the Organotellurium compound RF07: A possible control for canine visceral leishmaniasis? Environmental Toxicology and Pharmacology, 80, 103470.
Sheldrick, G. M. (2015). SHELXT - Integrated space-group and crystal-structure determination. Acta Crystallographica Section A: Foundations of Crystallography, 71(1), 3–8. https://doi.org/10.1107/S2053273314026370
Spackman, M. A., & Byrom, P. G. (1997). A novel definition of a molecule in a crystal. Chemical physics letters, 267(3–4), 215–220.
McKinnon, J. J., Spackman, M. A., & Mitchell, A. S. (2004). Novel tools for visualizing and exploring intermolecular interactions in molecular crystals. Acta Crystallographica Section B: Structural Science, 60(6), 627–668.
Spackman, M. A., & Jayatilaka, D. (2009). Hirshfeld surface analysis. CrystEngComm, 11(1), 19–32.
Turner, M. J., McKinnon, J. J., Wolff, S. K., Grimwood, D. J., Spackman, P. R., Jayatilaka, D., & Spackman, M. A. (2017). CrystalExplorer17. The University of Western Australia Australia.
Mackenzie, C. F., Spackman, P. R., Jayatilaka, D., & Spackman, M. A. (2017). CrystalExplorer model energies and energy frameworks: Extension to metal coordination compounds, organic salts, solvates and open-shell systems. IUCrJ, 4(5), 575–587.
Beck, A. D. (1993). Density-functional thermochemistry. III. The role of exact exchange. Journal of Chemical Physics, 98(7), 5646–5648.
Lee, C., Yang, W., & Parr, R. G. (1988). Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Physical review B, 37(2), 785.
Stephens, P. J., Devlin, F. J., Chabalowski, C. F., & Frisch, M. J. (1994). Ab initio calculation of vibrational absorption and circular dichroism spectra using density functional force fields. The Journal of physical chemistry, 98(45), 11623–11627.
McLean, A. D., & Chandler, G. S. (1980). Contracted Gaussian basis sets for molecular calculations. I. Second row atoms, Z=11–18. The Journal of Chemical Physics, 72(10), 5639–5648. https://doi.org/10.1063/1.438980
Frisch, M. J., Trucks, G. W., Schlegel, H. B., Scuseria, G. E., Robb, M. A., Cheeseman, J. R., Scalmani, G., Barone, V., Petersson, G. A., Nakatsuji, H. (2016). Gaussian 16.
Duggan, K. C., Walters, M. J., Musee, J., Harp, J. M., Kiefer, J. R., Oates, J. A., & Marnett, L. J. (2010). Molecular basis for cyclooxygenase inhibition by the non-steroidal anti-inflammatory drug naproxen. Journal of Biological Chemistry, 285(45), 34950–34959.
Sussman, J. L., Lin, D., Jiang, J., Manning, N. O., Prilusky, J., Ritter, O., & Abola, E. E. (1998). Protein Data Bank (PDB): Database of three-dimensional structural information of biological macromolecules. Acta Crystallographica Section D: Biological Crystallography, 54(6), 1078–1084.
Pettersen, E. F., Goddard, T. D., Huang, C. C., Couch, G. S., Greenblatt, D. M., Meng, E. C., & Ferrin, T. E. (2004). UCSF Chimera—A visualization system for exploratory research and analysis. Journal of computational chemistry, 25(13), 1605–1612.
Maier, J. A., Martinez, C., Kasavajhala, K., Wickstrom, L., Hauser, K. E., & Simmerling, C. (2015). ff14SB: Improving the accuracy of protein side chain and backbone parameters from ff99SB. Journal of chemical theory and computation, 11(8), 3696–3713.
Cousins, K. R. (2011). Computer review of ChemDraw ultra 12.0. ACS Publications.
Trott, O., & Olson, A. J. (2010). AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. Journal of computational chemistry, 31(2), 455–461.
Verdonk, M. L., Cole, J. C., Hartshorn, M. J., Murray, C. W., & Taylor, R. D. (2003). Improved protein-ligand docking using GOLD. Proteins Structure, Function, and Bioinformatics, 52(4), 609–623.
Case, D. A., Walker, R. C., Cheatham, T. E., Simmerling, C., Roitberg, A., Merz, K. M., … Darden, T. (2018). Amber 2018. University of California, San Francisco., 2018, 1–923.
Abro, A., & Azam, S. S. (2016). Binding free energy based analysis of arsenic (+ 3 oxidation state) methyltransferase with S-adenosylmethionine. Journal of Molecular Liquids, 220, 375–382. https://doi.org/10.1016/J.MOLLIQ.2016.04.109
Khan, J., Ali, G., Rashid, U., Khan, R., Jan, M. S., Ullah, R., … Sewell, R. E. (2021). Mechanistic evaluation of a novel cyclohexenone derivative’s functionality against nociception and inflammation: An in-vitro, in-vivo and in-silico approach. European Journal of Pharmacology, 902, 174091.
Wang, J., Wolf, R. M., Caldwell, J. W., Kollman, P. A., & Case, D. A. (2004). Development and testing of a general amber force field. Journal of computational chemistry, 25(9), 1157–1174.
Case, D. A., Babin, V., Berryman, J. T., Betz, R. M., Cai, Q., Cerutti, D. S., … others. (2014). The FF14SB force field.Amber, 14, 29–31.
Kräutler, V., Van Gunsteren, W. F., & Hünenberger, P. H. (2001). A fast SHAKE algorithm to solve distance constraint equations for small molecules in molecular dynamics simulations. Journal of computational chemistry, 22(5), 501–508.
Izaguirre, J. A., Catarello, D. P., Wozniak, J. M., & Skeel, R. D. (2001). Langevin stabilization of molecular dynamics. The Journal of Chemical Physics, 114(5), 2090–2098.
Roe, D. R., & Cheatham, T. E. (2013). PTRAJ and CPPTRAJ: Software for processing and analysis of molecular. Dynamics Trajectory Data, 9(7).
Genheden, S., & Ryde, U. (2015). The MM/PBSA and MM/GBSA methods to estimate ligand-binding affinities. Expert Opinion on Drug Discovery, 10(5), 449–461. https://doi.org/10.1517/17460441.2015.1032936
Miller, B. R., III., McGee, T. D., Jr., Swails, J. M., Homeyer, N., Gohlke, H., & Roitberg, A. E. (2012). MMPBSA. Py: An efficient program for end-state free energy calculations. Journal of Chemical Theory and Computation, 8(9), 3314–3321.
Lee, S. K., Lee, I. H., Kim, H. J., Chang, G. S., Chung, J. E., & No, K. T. (2003). The PreADME Approach: Web-based program for rapid prediction of physico-chemical, drug absorption and drug-like properties. EuroQSAR designing drugs and crop protectants: processes, problems and solutions, 418–420.
Author information
Authors and Affiliations
Contributions
All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Ethical Approval
All ethical norms were maintained by the authors during the article preparation process.
Consent to Participate
All authors agree to participate in the process.
Consent for Publication
All authors agree to publish the article.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mohamed, S.K., Ahmad, S., Albayati, M.R. et al. Insight into the Crystal Structures and Potential of Two Newly Synthesized Naproxen-Based Hydrazide Derivatives as Potent COX-2 Inhibitors. Appl Biochem Biotechnol 194, 5781–5807 (2022). https://doi.org/10.1007/s12010-022-04077-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-022-04077-2