Abstract
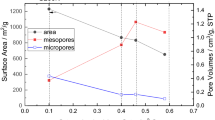
Recently, metal–organic frameworks (MOFs) are considered to be the moderate hosts for the bio-enzymes owing to their unique 3D pores and controllable surface affinity to the target molecules. In this work, the benzoic acid (BA)–modulated UiO-66-NO2 was introduced, and cytochrome c (Cyt C) was chosen as the target enzyme to evaluate the immobilization efficiency of the resulting UiO-66-NO2-BA. The immobilization conditions including pH, adsorption time, and temperature and the initial concentrations of BA were optimized. The adsorption kinetics and thermodynamics were analyzed to further explore the enhanced adsorption mechanism. It is worth noted that all the UiO-66-NO2-BA exhibited evidently enhanced adsorption capacities in comparison with the unmodified UiO-66-NO2 due to the formation of the chemical bonds between the UiO-66-NO2-BA and cytochrome C, indicating the positive roles of BA modification. Finally, the activities of the immobilized cytochrome C were assessed by using the catalytic oxidation of ABTS in the presence of H2O2, which reactions were also conducted over the free cytochrome C for comparison. The evidently improved stability under definite pH range, prolonged durability against the organic solvents, and the good reusability of the immobilized cytochrome C highlight the prospect applications of functional MOF immobilized enzymes in the practical catalytic reactions.
Graphical abstract












Similar content being viewed by others
References
Parvizpour, S., Hussin, N., Shamsir, M. S., & Razmara, J. (2021). Psychrophilic enzymes: Structural adaptation, pharmaceutical and industrial applications. Applied Microbiology and Biotechnology, 105, 899–907.
Dhakshinamoorthy, A., Asiri, A. M., & Garcia, H. (2020). Integration of metal organic frameworks with enzymes as multifunctional solids for cascade catalysis. Dalton Transactions, 49, 11059–11072.
Ying, T., Wang, Z. H., Lin, Y. W., Xie, J., Tan, X., & Huang, Z. X. (2009). Tyrosine-67 in cytochrome C is a possible apoptotic trigger controlled by hydrogen bonds via a conformational transition. Chemical Communications, 30, 4512–4514.
Kao, K. C., Lee, C. H., Lin, T. S., & Mou, C. Y. (2010). Cytochrome C covalently immobilized on mesoporous silicas as a peroxidase: Orientation effect. Journal of Materials Chemistry, 20, 4653–4662.
Kato, K., Suzuki, M., Tanemura, M., & Saito, T. (2010). Preparation and catalytic evaluation of cytochrome c immobilized on mesoporous silica materials. Journal of the Ceramic Society of Japan, 118, 410–416.
Kim, H. J., Lee, K. S., Won, M. S., & Shim, Y. B. (2008). Characterization of protein-attached conducting polymer monolayer. Langmuir, 24, 1087–1093.
Percin, I., Karakoc, V., Ergun, B., & Denizli, A. (2016). Metal-immobilized magnetic nanoparticles for cytochrome C purification from rat liver. Biotechnology and Applied Biochemistry, 63, 31–40.
Orhan, H., & Uygun, D. (2020). Immobilization of L-asparaginase on magnetic nanoparticles for cancer treatment. Applied Biochemistry and Biotechnology, 191, 1432–1443.
Yang, X., Zhao, C., Ju, E., Ren, J., & Qu, X. (2013). Contrasting modulation of enzyme activity exhibited by graphene oxide and reduced graphene. Chemical Communications, 49, 8611–8613.
Zhou, L., Jiang, Y., Ma, L., He, Y., & Gao, J. (2015). Immobilization of glucose oxidase on polydopamine-functionalized graphene oxide. Applied Biochemistry and Biotechnology, 175, 1007–1017.
Bulmus, V., Ayhan, H., & Piskin, E. (1997). Modified PMMA monosize microbeads for glucose oxidase immobilization. Chemical Engineering Journal, 65, 71–76.
Atiroglu, V., Atiroglu, A., & Ozacar, M. (2021). Immobilization of alpha-amylase enzyme on a protein @metal-organic framework nanocomposite: A new strategy to develop the reusability and stability of the enzyme. Food Chemistry, 349, 129127.
Chen, X., & Zhang, Q. (2019). Recent advances in mesoporous metal-organic frameworks. Particuology, 45, 20–34.
Wang, D., Zheng, P., Chen, P., & Wu, D. (2021). Immobilization of alpha-L-rhamnosidase on a magnetic metal-organic framework to effectively improve its reusability in the hydrolysis of rutin. Bioresource Technology, 323, 124611.
Liang, W., Wied, P., Carraro, F., Sumby, C. J., Nidetzky, B., Tsung, C. K., Falcaro, P., & Doonan, C. J. (2021). Metal-organic framework-based enzyme biocomposites. Chemical Reviews, 121, 1077–1129.
Pei, X., Wu, Y., Wang, J., Chen, Z., Liu, W., Su, W., & Liu, F. (2020). Biomimetic mineralization of nitrile hydratase into a mesoporous cobalt-based metal-organic framework for efficient biocatalysis. Nanoscale, 12, 967–972.
Ogata, A. F., Rakowski, A. M., Carpenter, B. P., Fishman, D. A., Merham, J. G., Hurst, P. J., & Patterson, J. P. (2020). Direct observation of amorphous precursor phases in the nucleation of protein-metal-organic frameworks. Journal of the American Chemical Society, 142, 1433–1442.
Marshall, R. J., Hobday, C. L., Murphie, C. F., Griffin, S. L., Morrison, C. A., Moggach, S. A., & Forgan, R. S. (2016). Amino acids as highly efficient modulators for single crystals of zirconium and hafnium metal-organic frameworks. Journal of Materials Chemistry A, 4, 6955–6963.
Lin, C., Xu, K., Zheng, R., & Zheng, Y. (2019). Immobilization of amidase into a magnetic hierarchically porous metal-organic framework for efficient biocatalysis. Chemical Communications, 55, 5697–5700.
Liu, X., Qi, W., Wang, Y., Lin, D., Yang, X., Su, R., & He, Z. (2018). Rational design of mimic multienzyme systems in hierarchically porous biomimetic metal−organic frameworks. ACS Applied Materials & Interfaces, 10, 33407–33415.
Liu, G., Xu, Y., Han, Y., Wu, J., Xu, J., Meng, H., & Zhang, X. (2017). Immobilization of lysozyme proteins on a hierarchical zeolitic imidazolate framework (ZIF-8). Dalton Transactions, 46, 2114–2121.
Zou, B., Zhang, L., Xia, J., Wang, P., Yan, Y., Wang, X., & Adesanya, I. (2020). Metal-organic frameworks conjugated lipase with enhanced bio-catalytic activity and stability. Applied Biochemistry and Biotechnology, 192, 132–145.
Xiong, J., Cai, X., & Ge, J. (2021). Enzyme–metal nanocomposites for antibacterial applications. Particuology, 02, 003.
Li, P., Chen, Q., Wang, T. C., Vermeulen, N. A., Mehdi, B. L., Dohnalkova, A., Browning, N. D., Shen, D., Anderson, R., & Gómez-Gualdrón, D. A. (2018). Hierarchically engineered mesoporous metal-organic frameworks toward cell-free immobilized enzyme systems. Chem, 4, 1022–1034.
Chen, Y., Angeles, F., Qiao, B., Krzyaniak, M., Sha, F., Kato, S., Gong, X., Buru, C., Chen, Z., Zhang, X., Gianneschi, N., Wasielewski, M., Cruz, M., & Farha, O. (2020). Insights into the enhanced catalytic activity of cytochrome c when encapsulated in a metal−organic framework. Journal of the American Chemical Society, 142, 18576–18582.
Chen, Y., Lykourinou, V., Vetromile, C., Hoang, T., Ming, L., Larsen, R., & Ma, S. (2012). How can proteins enter the interior of a MOF? Investigation of cytochrome c translocation into a MOF consisting of mesoporous cages with microporous windows. Journal of the American Chemical Society, 134, 13188–13191.
Lyu, F., Zhang, Y., Zare, R., Ge, J., & Liu, Z. (2014). One-pot synthesis of protein-embedded metal−organic frameworks with enhanced biological activities. Nano Letters, 14, 5761–5765.
Hu, Y., Dai, L., Liu, D., & Du, W. (2018). Rationally designing hydrophobic UiO-66 support for the enhanced enzymatic performance of immobilized lipase. Green Chemistry, 20, 4500–4506.
Cao, S., Yue, D., Li, X., Smith, T. J., Li, N., Zong, M., Wu, H., Ma, Y., & Lou, W. (2016). Novel nano-/micro-biocatalyst: Soybean epoxide hydrolase immobilized on UiO-66-NH2 MOF for efficient biosynthesis of enantiopure (R)-1, 2-octanediol in deep eutectic solvents. ACS Sustainable Chemistry Engineering, 4, 3586–3595.
Zhu, Q., Zhuang, W., Chen, Y., Wang, Z., Villacorta Hernandez, B., Wu, J., Yang, P., Liu, D., Zhu, C., Ying, H., & Zhu, Z. (2018). Nano-biocatalysts of Cyt c@ZIF-8/GO composites with high recyclability via a de novo approach. ACS Applied Materials & Interfaces, 10, 16066–16076.
Kumar, V., Misra, N., Goel, N. K., Thakar, R., Gupta, J., & Varshney, L. (2016). A horseradish peroxidase immobilized radiation grafted polymer matrix: A biocatalytic system for dye waste water treatment. RSC Advances, 6, 2974–2981.
Zhong, X., Xia, H., Huang, W., Li, Z., & Jiang, Y. (2020). Biomimetic metal-organic frameworks mediated hybrid multi-enzyme mimic for tandem catalysis. Chemical Engineering Journal, 381, 122758.
Niu, N., Zhao, W., Xiao, B., Liang, Y., Meng, X., Song, X., Li, D., Hong, J., & Movahedi, A. (2022). Quench treatment cytochrome c: Transformation from a classical redox protein to a peroxidase like enzyme. Journal of the Iranian Chemical Society, 19, 2347–2357.
Zhu, F., Xu, W., Li, X., Meng, H., Han, Y., Wu, J., Xu, J., Xu, Y., & Zhang, X. (2020). Lipase immobilization on UiO-66/poly(vinylidene fluoride) hybrid membranes and active catalysis in the vegetable oil hydrolysis. New Journal of Chemistry, 44, 14379–14388.
Tang, X., Luo, Y., Zhang, Z., Ding, W., Liu, D., Wang, J., Guo, L., & Wen, M. (2021). Effects of functional groups of -NH2 and -NO2 on water adsorption ability of Zr-based MOFs (UiO-66). Chemical Physics, 543, 111093.
Ramezanzadeh, M., Tati, A., Bahlakeh, G., & Ramezanzadeh, B. (2021). Construction of an epoxy composite coating with exceptional thermo-mechanical properties using Zr-based NH2-UiO-66 metal-organic framework (MOF): Experimental and DFT-D theoretical explorations. Chemical Engineering Journal, 408, 127366.
Wang, Y., Zhang, W., Guo, J., Duan, W., & Liu, B. (2019). Synthesis of well-defined internal-space-controllable UiO-66 spherical nanostructures used as advanced nanoreactor. ACS Applied Materials & Interfaces, 11, 38016–38022.
He, Z., Wu, F., Guan, S., Liu, L., Li, J., & Huang, Y. (2021). Polyamide amine/aramid nanofiber composite aerogels as an ultra-high capacity adsorbent for Congo red removal. Journal of Materials Chemistry A, 9, 13320–13331.
Quan, X., Sun, Z., Xu, J., Liu, S., Han, Y., Xu, Y., Meng, H., Wu, J., & Zhang, X. (2020). Construction of an aminated MIL-53(Al)-functionalized carbon nanotube for the efficient removal of bisphenol AF and metribuzin. Inorganic Chemistry, 59, 2667–2679.
Chen, X., Xue, S., Lin, Y., Luo, J., & Kong, L. (2020). Immobilization of porcine pancreatic lipase onto a metal-organic framework, PPL@MOF: A new platform for efficient ligand discovery from natural herbs. Analytica Chimica Acta, 1099, 94–102.
Yuan, X., Ou, J., Zhang, P., Xu, W., Jiang, B., & Tang, K. (2020). PEG-modified lipase immobilized onto NH2-MIL-53 MOF for efficient resolution of 4-fluoromandelic acid enantiomers. International Journal of Biological Macromolecules, 165, 1793–1802.
Kaba, O. B., Filippov, L. O., Filippova, I. V., & Badawi, M. (2021). Interaction between fine particles of fluorapatite and phosphoric acid unraveled by surface spectroscopies. Powder Technology, 382, 368–377.
Li, Y., Zhang, X., Yang, R., Li, G., & Hu, C. (2015). The role of H3PO4 in the preparation of activated carbon from NaOH-treated rice husk residue. RSC Advances, 5, 32626–32636.
Hausdorf, S., Wagler, J., Mossig, R., & Mertens, F. (2008). Proton and water activity-controlled structure formation in zinc carboxylate-based metal organic frameworks. Journal of Physical Chemistry A, 112, 7567–7576.
Gao, X., Ding, Y., Sheng, Y., Hu, M., Zhai, Q., Li, S., Jiang, Y., & Chen, Y. (2019). Enzyme immobilization in MOF-derived porous NiO with hierarchical structure: An efficient and stable enzymatic reactor. ChemCatChem, 11, 2828–2836.
Li, R., Liu, S., Zhou, X., Liu, H., Zhou, H., Wang, C., Liu, Y., & Zhang, X. (2022). Efficient immobilization of catalase on mesoporous MIL-101 (Cr) and its catalytic activity assay. Enzyme and Microbial Technology, 156, 110005.
Lage, F., Bassi, J., Corradini, M., Todero, L., Luiz, J., & Mendes, A. (2016). Preparation of a biocatalyst via physical adsorption of lipase from Thermomyces lanuginosus on hydrophobic support to catalyze biolubricant synthesis by esterification reaction in a solvent-free system. Enzyme and Microbial Technology, 84, 56–67.
Funding
The authors are very grateful for the financial support of LiaoNing Science and Technology Development Foundation Guided by Central government (2021JH6/10500141).
Author information
Authors and Affiliations
Contributions
Fenfen Zhu conceived and designed research. Peide An performed software analyses and the most writing work. All the authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
All authors read and approved for publication.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
1. It is the first time to use BA-functional UiO-66-NO2 in cytochrome C immobilization.
2. UiO-66-NO2-BA showed distinct enhanced immobilization efficiency for cytochrome C.
3. BA modification endows UiO-66-NO2 hierarchical pores and diminished particle size.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
An, P., Zhu, F., Liu, S. et al. Immobilization of Cytochrome C by Benzoic Acid (BA)-Functional UiO-66-NO2 and the Enzyme Activity Assay. Appl Biochem Biotechnol 194, 5167–5184 (2022). https://doi.org/10.1007/s12010-022-04018-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-022-04018-z